MOJ
eISSN: 2576-4519


Research Article Volume 2 Issue 1
1Department of Hepatobiliary Surgery1, China
2Surgical Intensive Care Unit, First Affiliated Hospital of Zhejiang University, China
3Department of Anesthesiology, China
4Department of General Surgery, The Affiliated Wuxi No.2 People?s Hospital of Nanjing Medical University, China
Correspondence: Yigang Chen, Department of General Surgery, The Affiliated Wuxi No.2 Peoples Hospital of Nanjing Medical University, China, Tel 8651 0666 81222
Received: December 28, 2017 | Published: January 5, 2018
Citation: Dong L, Tan X, Fu M, et al. Experimental study on bone marrow mesenchymal stem cells for the treatment of acute pancreatitis in rats. MOJ App Bio Biomech. 2018;2(1):1–7. DOI: 10.15406/mojabb.2018.02.00036
Explored the appropriate way for bone marrow mesenchymal stem cells (bmMSCs) infuse in rat models of severe acute pancreatitis. Severe AP was induced in Sprage-Dawley rats by the sodium taurocholate bile duct retrograde injection. Rats were randomly divided into five groups: control group, SAP group, SAP+bmMSCs (tail vein) group, SAP+bmMSCs (intraperitoneal) group and SAP+bmMSCs (tail vein + intraperitoneal) group. In bmMSCs infused groups, bmMSCs labeled with DAPI were injected via the tail vein, intraperitoneal and via tail vein + intraperitoneal respectively 3h after SAP. Rats were sacrificed on days 1, 2 and 3, and pancreatic tissues and the blood were collected. The levels of serum anti-inflammatory cytokines IL-10 and inflammatory cytokines (IL-1β, TNF-α, IL-6) were determined. Pathological changes of the pancreas (HE staining) were observed under light microscope. Positioning of DAPI labeled bmMSCs In vivo were detected under fluorescence microscope at the same time. The pancreas in SAP group showed significantly massive hemorrhage, edema, inflammation, and necrosis compared with control group. The inflammation, edema, hemorrhage and necrosis in each model of pancreatitis were reduced significantly in SAP+bmMSCs group as compared to SAP group (P<0.05). In bmMSCs transplanted group, bmMSCs remarkably reduced the levels of pro-inflammatory cytokines (TNF-α, IL-6, and IL-1β), on the contrary increased the levels of anti-inflammatory cytokines (IL-10) in SAP rats. Infused bmMSCs through tail vein + intraperitoneal indicate a better recovery than through tail vein or intraperitoneal alone. Compared with intraperitoneal injection method, more bmMSCs were observed to be presented in pancreas when bmMSCs were injected through tail vein + intraperitoneal or tail vein injection. In a rat model of SAP, transplantation of bm-MSCs through tail vein + intraperitoneal significantly inhibits inflammation and decreases pancreatic injury secondary to SAP. It was a better method for pancreatitis.
Keywords: acute pancreatitis, bone marrow mesenchymal stem cells, endothelin, nitric oxide
Acute pancreatitis (AP) is a rapid onset and disease progression, and approximately 15-20% of patients with AP may develop severe acute pancreatitis (SAP) which has the high mortality rate.1 It is now generally accepted that the pathogenesis of AP start from the activation of pancreatin in pancreatic acinar cells and then induces both local and systemic inflammation.2 Tumor necrosis factor (TNF), C-reactive protein, interleukin (IL)-1, IL-8, IL-6, endothelin, nitric oxide (NO) and other inflammatory factors are thought to be involved in both the genesis and progression of AP. Inflammatory factors play a crucial role in the progression from slight acute pancreatitis to severe acute pancreatitis. Although efforts began to implement the appropriate method for the treatment of acute pancreatitis, mortality rate of SAP has not substantially decreased during the past two decades.3 For this reason, adult stem cells especially mesenchymal stromal cells (MSCs) have been used in the experimental studies in the treatment of SAP. MSCs are multipotent adult stem cells that can be obtained from many different tissues shown to low or no immunogenicity. MSCs can differentiated into many cells types by providing the suitable microenvironment similar to In vivo.4,5 Plenty of studies confirmed that MSCs are able to ameliorate pancreatic injury, inhibit systemic inflammation and promote tissue repair in AP.6,7 Other studies revealed that MSCs could relieve pancreatitis-associated kidney damage8,9 or pancreatitis-associated lung injury.10,11 Unfortunately, these studies did not highlight the appropriate way for the stem cells infuse In vivo. To refine these approaches, we transplanted bmMSCs through tail vein, intraperitoneal and tail vein combined with intraperitoneal respectly and then position of bmMSCs, compared pancrearic injury and inflammatory response.
Animals
Healthy Sprague Dawley (SD) rats weighing 200-250g were provided by Shandong Lukang by the Experimental Animal Room of pharmaceutical companies, animal certificate (SLXK Lu 20080002), the experiment disposal of animals found in the animal ethics standards. All the animals were fed under 12-h light-dark cycle conditions with temperature maintained at 20±2°C. All experiments protocols were approved by the Animal Ethical Committee of institute.
Materials and reagents
Fetal bovine serum (Gibco Company), Dulbecco’s modified Eagle’s medium (DMEM; Hyclone Corporation, United States), 4,6-diamidino-2-phenylindole (DAPI; Roche, Switzerland), IL-10 ELISA for rats, and TNF-α ELISA kit for rats (all ELISAs from GBD company, USA). 0.25% trypsin +0.02% EDTA (Wuhan Boster Company); rabbit anti-rat CD45, CD90 antibody (biolegend company); Histopaque-1077 separation medium (Sigma); flow cytometry (Beckman Coulter Inc.; Type: Epics XL • MCL).
Culture and identification of mesenchymal stem cells
About 140g SD rats were sacrificed and the primary bmMSCs were collected from the femurs and tibias. Following flushing the bone marrow cavity with DMEM-LG medium and plated the cells in basal medium supplemented with 20% fetal bovine serum, and penicillin (100U/ml)/streptomycin (100U/ml) in culture flasks at 37°C in 5% CO2, 95% air in a humidified incubator. When cells confluence reached >80% they were harvested by multiple digestions and passages. The surface markers of bmMSCs (CD45 and CD90) were detected by flow cytometry.
Preparation of rat SAP model
Before surgery, rats were fasted for 12h but given water. SAP group: SD rats, weighing 200-250g, were selected for the experiment. A midline incision was made at the abdomen (2cm in length) and the duodenum was taken out of the abdominal cavity. The pancreas was identified and the duodenal papilla, common bile duct and pancreatic duct were located. Clamp the common bile duct by small clamp, and the shared outlet of common bile duct and pancreatic duct on the duodenum was clamped by bending clamp. Then, 3% sodium taurocholate (1ml/kg) was slowly injected at a speed of 5 min/ml.
Cell transplantation
Rats (n=90) were randomly divided into control group (n=18), SAP group (n=18), SAP+bmMSCs (tail vein) group (n=18), SAP+bmMSCs (intraperitoneal) group (n=18) and SAP+bmMSCs (tail vein + intraperitoneal) group (n=18). In the SAP+bmMSCs (tail vein) group and SAP+bmMSCs (intraperitoneal)) group, 1mL of the bmMSCs suspension (containing approximately 1×106 cells labeled with DAPI) were injected into the tail vein or intraperitoneal respectively. In the SAP + bmMSCs (tail vein + intraperitoneal) group, 0.5mL of the bmMSCs suspension was injected into the caudal vein and at the same time 0.5mL of the bmMSCs suspension was injected into intraperitoneal.
Cell marking
Third generation bmMSCs were used for subsequent experimentation. bmMSCs were labeled with DAPI(final concentration 50g/l) and incubated at 37˚C for 12h and observed under fluorescence microscope. Then repeated washing the cells with PBS to remove DAPI, the cells were collected after one more centrifugation. DMEM-F12 medium (1ml) supplemented containing 1x106 MSCs were injected into the rats with SAP through the caudal vein at 3h.
Morphological examination of the pancreas
Detection of serum pro-inflammatory cytokines and anti-inflammatory cytokines Six rats were collected from all groups at 24h, 48h and 72h after injection bmMSCs, and pathological scoring was done according to the pathological criteria for pancreatitis (Table 1). Blood samples were collected from the apex of the hearts. The concentrations of serum IL-10, IL-1, IL-6 and TNF-αwere determined using ELISA. The pancreatic tissues were flash frozen, and the number of DAPI positive cells were observed under fluorescence microscopy. The body of pancreas was obtained at the indicated time, fixed in 10% formalin containing phosphate-buffered saline, and embedded in paraffin. Sectioned at 4 mm thickness sections from each group were analyzed. Samples were stained with H&E.
Statistical analysis
All data were expressed as means ± SEM. Data from experimental groups were compared by Student’s t test or analysis of variance. A value of p<0.05 was considered statistically significant and all analyses were performed using Graphpad Prism 5 and SPSS22.0.
Pathological |
Scoring |
||||
|---|---|---|---|---|---|
0 |
1 |
2 |
3 |
4 |
|
4Edema |
No |
Diffused expansion |
Diffused expansion of |
Diffused expansion of |
Diffused expansion |
Infiltration of inflammatory cells |
0-1 white blood |
2-10 white blood |
11-20 white blood |
21-30 white blood |
≥31 white blood |
Bleeding |
No |
Pancreatic parenchy- |
Pancreatic parenchy- |
Pancreatic-parenchy- |
Pancreatic parenchy- mal bleeding at |
Necrosis |
No |
1-4 necrotic cells/ |
5-10 necrotic cells/ |
11-16 necrotic cells/ |
≥17 necrotic cells/ |
Table 1 Criteria for pathological scoring of the pancreas
General morphology of mesenchymal stem cells and the expression of surface markers
Isolated the cells from bone marrow distributed sparsely on the culture flasks. During the early days of incubation the cells were spindle-shaped morphology. Within 3-5days, the cells displayed small colonies that called colony forming units (Figure 1A). Primary cells reached monolayer confluence and appeared mostly fibroblast like within 10-14 days (Figure 1B). In the later passages, bmMSCs exhibited large and fibroblast-like morphology (Figure 1C). Identification of bmMSCs was by Flow cytometry. The cells analysis data showed that bmMSCs expressed CD54, CD90 but not CD45, the positive rate of CD90 was 99.6%. In contrast, CD45 were negative (0.56% and 0.89%, respectively), demonstrating that the purity of the MSCs was >95% (Figure 1D).
Histologic Analysis of Pancreas after hcMSCs Infusion
At 24h, 48h and 72h points, edema, infiltration of inflammatory cells and necrosis were not observed in the pancreatic tissue in control group. Pancreatic tissue from the SAP group induced by sodium taurocholate showed significant edema and inflammation compared with the control (P<0.05). In bmMSCs infused group the edema formation and inflammatory cell infiltration were significantly reduced, and the improvement of pancreatic in SAP+bmMSCs (tail vein + intraperitoneal) group was better than in SAP+bmMSCs (tail vein) group and SAP+bmMSCs (intrapertoneal) (Figure 2). On the basis of pancreatic parenchymal necrosis, pathological scoring was performed (Figure 3). As showed in Figure 3 the pathological scores of pancreatic edema, necrosis and infiltration of inflammatory cells in SAP group were markedly higher than those in control group (P<0.05). In SAP+bmMSCs group, the pancreatic scores were reduced when compared with SAP group. In addition, in SAP+bmMSCs (tail vein + intraperitoneal) group, the pancreatic scores were reduced more significantly than in SAP+bmMSCs (tail vein) and in SAP+bmMSCs (intraperitoneal) group.
Regulation of bmMSCs on inflammatory factors
In sodium taurocholate -induced SAP models, levels of IL-1β, TNF-α, IL-6 and IL-10 in serum were detected by ELISA. Compared with control group, serum IL-1β, TNF-α and IL-6 were obvious increased in SAP group and tended to decrease after injection of bmMSCs treatment. On the contrary, bmMSC increased the level of the anti-inflammatory cytokines such as IL-10 in rats with SAP. Interestingly, bmMSC could better regulate inflammatory mediators and cytokines in rats receiving bmMSCs through tail vein + intraperitoneal compared with receiving bmMSC through tail vein or intraperitoneal alone.
Detection of MSCs in rat pancreas
DAPI was selected as the labeling system for bmMSCs tracking In vivo. We labeled bmMSCs with DAPI. We further investigated whether bmMSCs migrate into the injured pancreatic after bmMSC transplant by using fluorescence microscopy. DAPI–labeled cells in the bmMSC infused groups displayed blue fluorescence in both SAP + bmMSCs (tail vein + intraperitoneal) group and SAP + bmMSCs (tail vein) group. However there was no cell-specific blue fluorescence was observed in sections from rats that injected DAPI-labeled bmMSC through intraperitoneal.
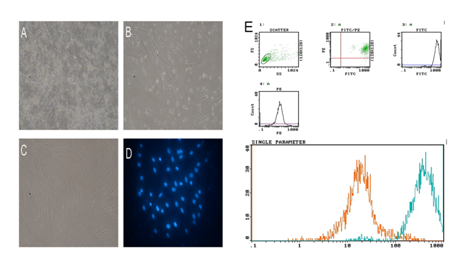
Figure 1 Isolated cells from bone marrow of rat distributed sparsely on the culture flasks. (A) During the early days of incubation the cells displayed mostly spindle shaped morphology. (B) The small colonies reached monolayer confluence within 10-14 days. (C) Most of these MSCs exhibited large and displayed mostly fibroblast-like morphology in the later passages. (D) labeled MSCs with DAPI. (E) Surface markers of MSCs CD90 was 99.6%. In contrast, CD45 were negative (0.56% and 0.89%, respectively)
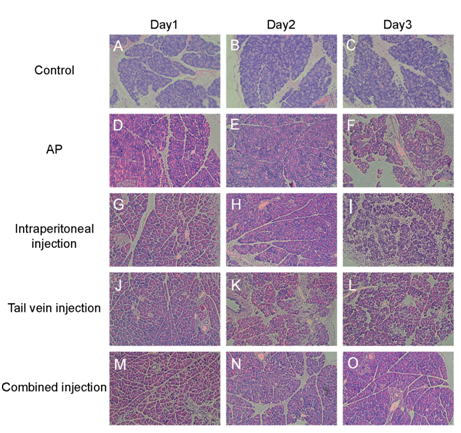
Figure 2Histological evaluation of pancreas in sodium taurocholate-induced SAP when treated with bmMSCs In vivo (×200). A-C, Pancreatic pathology on days 1, 2 and 3 in control group; D-F, SAP group on days 1, 2 and 3; G-I, SAP + BMSCs (intraperitoneal) group J-L, SAP + bmMSCs (tail vein) group; M-O, SAP + bmMSCs (tail vein + intraperitoneal) group
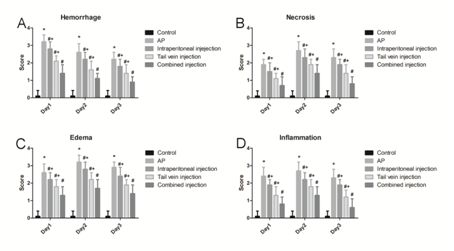
Figure 3 Scoring of pancreatic pathology. A, Score of pancreatic hemorrhage; B, Score of pancreatic necrosis; C, Score of pancreatic edema; D: Score of pancreatic inflammatory cell infiltration. * means P<0.05 when compared with Control group, # P means P<0.05 when compared with SAP group. + means P<0.05 when compared with SAP + combined injection group
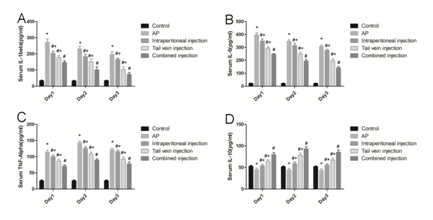
Figure 4 Inflammatory cytokines after infusion of bmMSCs. IL-1β (A), IL-6 (B), TNF-α (C), and IL-10 (D) serum levels after bmMSC infusion by enzyme-linked immunosorbent assay. *P means P<0.05 when compared with Control group, #P means P<0.05 when compared with SAP group. + P means P<0.05 when compared with SAP+combined injection group
AP is acute abdominal diseases and characterized elevated serum amylase and lipase, acinar cells necrosis, pancreatic edema, and inflammatory cells infiltration. Some patients with AP may develop SAP with lots of complications and high mortality, as yet has no satisfactory therapy. During the course of pancreatitis multiple inflammatory cytokines are reported involved in the pathogenesis of SAP including anti-inflammatory cytokines (IL-10) and pro-inflammatory cytokines (IL-1, IL-6, TNF-α).12,13 Mesenchymal stromal cells (MSCs) as a kind of stem cells can differentiat into various adult cells and tissue and can inhibit the inflammatory response.14 To date, accumulated evidence from preclinical and linical studies has confirmed that MSCs conferred biological and functional protections in diabetes, myocardium infarction, brain and spinal cord injury etc.15 In re-sent years some experiment have reported the protective roles of MSCs in AP16‒20 MSCs infusion through tail vein can protect the structural integration of acinar cells, promoted pancreatic angiogenesis, significantly inhibited inflammation and attenuate acinar cell apoptosis. Sun XC found that intraperitoneal injection of MSCs exerted protective effects on pancreas and small intestine injury.21 The goal of this study was to explore the appropriate way for the stem cells infuse In vivo. In the previous study of our group, bmMSCs were transplanted through tail vein, intraperitoneal and tail vein+ intraperitoneal respectivelyl 3h after SAP, and then the number of bmMSCs migrated into pancreatic and the therapeutic effect of bm-MSCs was compared. Sodium taurocholate bile duct retrograde injection was employed to induce acute pancreatitis. This animal model was feasible and easy to establish. The pancreatic enzyme detection and pathological examination reveal this animal model was stable. The pathological showed pancreatic edema, pancreatic parenchymal bleeding, massive inflammatory cells infiltration, and acinar cell necrosis. The bone marrow derived MSCs were injected into SAP rats via the tail vein, intraperitoneal or tail vein+intraperitoneal respectively. Some studies showed that MSCs could migrate into pancreas during early phase in AP.16,17 Consistent with recent studies, bmMSCs presented in pancreas on days 1, 2 and 3 when we injected bmMSCs through tail vein or through tail vein + intraperitoneal. However we observed that bmMSCs were nearly could not be presented in pancreas on days 1, 2 and 3 when we transplanted bmMSCs through intraperitoneal. It was demonstrated that bmMSCs could not migrate into pancreatic when they infused into intraperitoneal. In this study, we also confirmed that bmMSCs infusion significantly decreased serum amylase and lipase, relieved pancreatic lesions (edema, necrosis and inflammatory cell infiltration) caused by injection of sodium taurocholate.16,18 And we observed the better recovery of damaged pancreas when we transplanted bmMSCs through the tail vein+ intraperitoneal compared with through tail vein or intraperitoneal alone. Consistently, inflammatory cytokines, including IL-1β, IL-6 and TNF-α of rats in the bmMSCs infusion groups were lower than the SAP group, whereas IL-10 (anti-inflammatory mediator) levels were higher in the bmMSCs infusion groups compared with SAP group. These results indicated that bmMSCs could regulate immune response by promoting the expression of anti-inflammatory mediators and reducing the expression of inflammatory factors in SAP rat modles. In addition, transplant bmMSC through tail vein combine with intraperitoneal could better inhibit inflammation than transplant bmMSC through tail vein or intraperitoneal alone. Previous studies have confirm that MSCs can repair the injured pancreas through migrate into the injured pancreas and differentiate into pancreatic cells. Interestingly, in this study the bmMSCs excerted the protective role in SAP althrough there was not enough bmMSCs homed and positioned in pancreas in SAP + bmMSCs (intraperitoneal) group. In recent years, MSCs are found to secret some cytokines such as interleukin 10, IL-1Reg, TGF-β, HGF, vascular endothelial growth factor and insulin-like growth factor IGF-1 in a paracrine dependent manner to exert the anti-inflammatory effect.22,23 It was indicated that the bmMSCs’ protective role in SAP+bmMSCs (intraperitoneal) groups was through a paracrine effect, without homing to pancreas. According to our results, the bmMSC infused through tail vein and intraperitoneal interplay to amplify the protective role in SAP.
In conclusion, our results demonstrated that transplanted bmMSCs can decrease serum amylase and lipase, alleviate pancreatic lesions (edema, inflammatory cell infiltration and necrosis) caused by injection of sodium taurocholate in a rat model of SAP. According to the results of this study, transplanted bmMSCs through tail vein+intraperitoneal could be considered the better method of choice for cell-based treatment of SAP. Of course, some problems have not yet been overcome in this study. The future research should be focused on why bmMSCs can play a better role when they were transplanted through tail vein+intraperitoneal, how to lengthen cell survival time and improve the safety of bmMSCs transplantation.
This study was supported by a National Natural Science Foundation of China 81171466.
The author declares that there is no conflict of interest.

©2018 Dong, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.