Journal of
eISSN: 2377-4282


Research Note Volume 7 Issue 3
Department of Chemistry, Amirkabir University of Technology, Iran
Correspondence: Majid Abdouss, Department of Chemistry, Amirkabir University of Technology, Tehran, Iran
Received: February 15, 2018 | Published: June 13, 2018
Citation: Norouzi Z, Abdouss M, Tajiki A, et al. Application of chitosan/PVA nanofibrous composite for molecular capture. J Nanomed Res. 2018;7(3):221 ? 226. DOI: 10.15406/jnmr.2018.07.00190
Nanofiberous membrane with average diameters of 86 nm have been prepared by electrospinning of chitosan (CS) mixed with poly (vinyl alcohol) (PVA) in 2% (v/v) aqueous acetic acid. The formation of nanofibers was feasible with 3% concentration of solution containing up to an equal mass of chitosan. Finer fibers, fewer beaded structures and more efficient fiber formation was observed with increasing PVA contents. The interaction between CS and PVA in the solution was studied by FTIR spectra. The spectroscopic measurement showed that chitosan/PVA nanofiberous membrane (CS/PVAnfm) could recognize some organic molecules such as Acid Blue 26 by electrostatic interactions. The molecular capturing ability of nanofibers was increased with the amount of CS/PVAnfm in solution. The results suggested that the nanofibrous membrane was potentially applied to purification/separation processes.
Keywords: acid blue 26, chitosan, dye removal, electrospinning, molecular capture, nanofiberous membrane
Chitosan is N-deacetylated form of chitin which is obtained by alkaline treatment of chitin (50% of aqueous NaOH) at high temperatures. Chitosan has a repeated structure of (1,4)-linked β-D-glucosamine and an apparent pK of 6.5. Commercial products are traditionally composed of 80% β-D-glucosamine and 20% N-acetyl-β-D-glucosamine.1 Chitosan is a cationic polymer. Its key properties are biocompatibility, non-toxicity and solubility in moderated acidic aqueous solutions. Chitosan has been widely used in food, cosmetic, biomedical, wastewater treatment, and pharmaceutical applications.2,3 A large variety of useful forms, including beads, films, sponges, tubes, powders, and fibers, can be obtained from chitosan.4 Electrospinning is a versatile technique for producing multifunctional nanofibers from various polymers, polymer blends and composites.5 Electrospinning applies high voltages to a capillary droplet of polymer solution or a melt to overcome liquid surface tension and thus enables the formation of much finer fibers than conventional fiber spinning methods.6
Electrospun nanofibers have many unique properties such as high surface area-to-volume, pore size within a nano range, high porosity and flexibility for chemical/physical functionalization.7 Therefore, it has been shown that the very fascinating properties of these electrospun nanofibers make them applicable in numerous areas, including biotechnology membranes/filters, electroanalysis, power source, and so on.8 Naturally, proteins and polysaccharides are attractive materials for electrospinning. However, CS is difficult to electrospin in to a nanofibrous structure because it has polyionic characteristics in (acidic) aqueous solutions due to the many amino groups in the CS backbone. This polyionic nature of CS increases the surface tension of the solution considerably. Therefore, a strong electrical force is needed to produce electrospun nanofibers, and beads are often formed during the electrospinning process, which is likely due to the repulsive forces between the ionic groups in the backbone in an acidic or aqueous solution.9 However, because of the strong molecular interaction, generating smooth and uniform electrospun chitosan nanofibers still remains a great challenge. Poly (vinyl alcohol) (PVA) is a non-toxic, water-soluble, biocompatible, and biodegradable synthetic polymer, which is widely used in biomedical field. PVA has better fiber forming and highly hydrophilic properties, and it has been selected as suitable partner for fabrication of nanofibers.10
In current work nanofibers of CS/PVAnfm were synthesized through Electrospinning method. Then the obtained nanofibers were used for efficient removal of an acid dye (Acid Blue 26) from aqueous media. The concentration of dye molecules were measured by uv-visible spectroscopy before and after using nanofiber and the difference was referred as nanofiber capacity. Different parameters such as nanofiber dosage and contact time were investigated. It has shown that the nanofibers have great potential for removal of organic molecules. Acid Blue 26 used as a model of organic molecule in this work.
Chitosan, Poly Vinyl Alcohol and glutaraldehyde was purchased from Sigma Aldrich, Acetic Acid and other solvents were from analytical grade and obtained from Merck Company. Acid blue 26 with C.I.27070 was obtained from textile department, Amirkabir University of Technology.
Viscosity measurements
Interestingly low molecular weight chitosan is readily soluble in water due to free amino group in D-glucosamine and shows excellent biological activities.[11] however high and medium molecular weight chitosan is poorly soluble which makes them difficult to adsorb anions due to absence of free amino groups. As molecular weight and viscosity have a direct relationship, we are aimed to measure viscosity of chitosan solutions. All polymers increase the viscosity of the solvent in which they are dissolved. Several important viscosity functions are used in viscosity studies. The relative viscosity, , is the dimensionless ratio of solution viscosity, η, to solvent viscosity, η0. The specific viscosity, , is related to the fluid viscosity increase due to all polymer solute molecules. The reduced viscosity, , is the fluid viscosity increase per unit of polymer solute concentration, c, expressed in g/mL. The intrinsic viscosity, [η] (in mL/g), is the limit of the reduced viscosity as the polymer solute concentration approaches zero following the Huggins relationship:
(1)
The viscosity-average molecular weight (Mv) of chitosan was determined by dilute solution viscosity measurement using Ubbelohde capillary viscometer (Schott, Germany) at 25°C. A mixture of 0.1M HOAc- 0.2M NaCl was used as the solvent to prepare chitosan solutions with concentrations from to g/mL. The viscosity-average molecular weight (Mv, in Daltons or Da) was derived from the following Mark–Houwink equation:
(2)
Electrospinning procedure
First of all, chitosan was dissolved in acetic acid 2% (v/v), to obtain chitosan 3% (w/w). The solution was stirred overnight at room temperature. Poly Vinyl alcohol was dissolved in water and heated till 90°C for 2h to obtain PVA 8% (w/w) and added to above solution to obtain CS/PVAnfm in mass ratios of 20/80. The above solution was electrospun to obtain nanofibers.
Crosslinking of nanofibers
The electrospun CS/PVAnfm were crosslinked via a glutaraldehyde (GA) vapor crosslinking method. Briefly, a Petri dish containing GA solution (20% aqueous solution, 10mL) was placed at the bottom of a desiccator. Then the electrospun nanofibrous mats deposited onto the supporting aluminum foils were put onto the top ceramic plate of the desiccator. Vacuum was applied for 48h. After that, the mats were exposed in a fume hood for 2h, followed by a heating treatment at 100°C in an electric oven for 1h to remove the residual GA and to partially enhance the crosslinking. The nanofibrous mats were rinsed with water 3 times to remove the excess GA.12
Characterization of nanofibers
The morphology of electrospun CS/PVAnfm was observed with a scanning electron microscope (SEM) (XL30 Philips) with gold coating. The average fiber diameter and diameter distribution were determined by randomly measuring the diameters of the nanofibers at 100 different points from SEM images. FT-IR spectra of chitosan, PVA and CS/PVAnfm were recorded by a Nicolet Nexus 670 USA. All samples were prepared as KBr pellets and scanned over the wave number range of 4000–400 cm−1 at a resolution of 4.0cm−1.
Molecular recognition of CS/PVAnfm
The filtration performance of the CS/PVAnfm was tested using Acid Blue 26 (with C.I.27070) as a model organic molecule. Uptake was determined by measuring the reductions in absorbance or depletion from solution using UV–vis spectrophotometry (JENWAY 6505) in the wavelength of 635nm. The weight of fiber mass and the amount of Acid Blue 26 used were calculated under the assumption that 20% CS(w/w) is present on the fiber surface and thus available for complexation and 1:1 complexation between CS and Acid Blue 26 molecules occur. A 10ppm of Acid Blue 26 solution was prepared in water. The pH of the solution was adjusted to neutral (pH 7) by adding drops of pH buffer solution. The pH of the solutions was measured with a pH meter (pH meter 827, Metrohm) before and after the UV–vis experiments; the pH of the solutions was unchanged during the course of the UV–vis experiments. 5mg of CS/PVAnfm were placed separately in the bottom of the flask and were filled with 10ml of Acid Blue 26 solution. The absorbance spectra of the Acid Blue 26 solutions were recorded initially (time=0, right after adding the fibers) and every 1h for 24hours.
Determination of intrinsic viscosities
Figure 1 shows the dependence of the precursor viscosity on its concentration. It is found that its viscosity increases linearly with increasing concentration. This result is essential for the optimization on the formation of nanofibers of this material. As shown in Figure 1, the curve should be linear and the y-axes intercept is the intrinsic viscosity. The intrinsic viscosity ([η]) for Seacure chitosan was found equal to 780mL/g.
FT-IR spectra
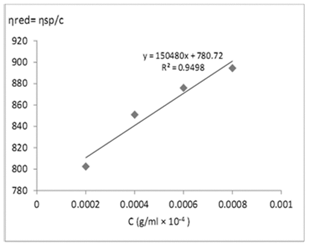
Figure 1 Huggins plot (ηred = ηsp/c vs. c) for Seacure chitosan with the intrinsic viscosity ([η]) calculated from the y-intercept.
Figure 2 gives FT-IR spectra of CS, PVA and CS/PVAnfm. CS membrane showed FT-IR absorption features around 905 and 1154cm-1 peaks assigned saccharine structure and a weaker amino characteristic peak at 1262cm-1 was the absorption of (O–H), and the peak at 1321 cm-1 was assigned to the CH3 symmetrical deformation mode. The CS exhibited characteristic broad bands of OH group at 3400-3500cm-1.The bands of NH2 group and O-C-NH2 group can be observed at 1652cm-1. The broad bands of CH3 group and CH3-O group can be observed at 1000–1200 cm-1. The FT-IR spectra of PVA showed the characteristic broad band at 2900-3000 cm-1 for CH2 group and CH3 group, respectively.
It was observed that the absorption peak at about 3420cm-1 concerned with –OH and –NH stretching vibrations shifted to a lower wave number with the adding of PVA in the nanofiberous membrane. Moreover, compared with FT-IR spectra of pure CS membrane, the absorption peak of CS/PVAnfm at 1262cm-1 disappeared. These results suggested the formation of hydrogen bond between CS and PVA molecule. Fourier Transform Infrared Spectroscopic measurement exhibited the existence of relevant functional groups of both PVA and CS in the nanofiberous membrane.
Electrospun nanofiberous membrane characterization
While electrospinning has proven to be a powerful and generally accepted means of fabricating polymer micro and nanofibers, its applicability to a new material system is not straightforward. Previous studies have shown that the system configuration and operation conditions differ vastly from one material system to another, depending on the material and the choice of solvent. Physical and chemical parameters of polymer solution such as viscosity, electric conductivity, and polymer concentration can definitely affect the formability and morphology of electrospun nanofibers. A set of requirements were defined to rate the success of electrospinnability of the solution. For success all of the following requirements need to be fulfilled:
Important observations that will determine the selection of the optimal values are uniformity of the deposition at nanolevel, diameter of the fibers, distribution of the fiber thickness and absence of bead formation.13 To select the most suitable composition of the chitosan containing solution (90% acetic acid), different chitosan solutions have been prepared and electrospun. Table 1 summarizes different experiments that have been performed for electrospinning of pure chitosan from different acetic acid solutions.
Conc. of chitosan (%) |
Conc. of acetic acid (%) |
Fiber formation |
6 |
2 |
No fibers |
6 |
20 |
No fibers |
6 |
90 |
No fibers |
5 |
2 |
No fibers |
5 |
20 |
No fibers |
5 |
90 |
No fibers |
4 |
2 |
No fibers |
4 |
20 |
Few fibers |
4 |
90 |
Few fibers |
3 |
2 |
No fibers |
3 |
20 |
Few fibers |
3 |
90 |
Few fibers |
Table 1 Electrospinning of pure chitosan from different acetic acid solutions
The major complication in electrospinning of chitosan is the poor solubility of chitosan and the high viscosity of its aqueous solution. At low polymer concentrations, the solutions do not contain sufficient material to produce stable solid fibers. With increasing polymer concentration, the number of direct interchain associations of chitosan molecules in the solution increases rapidly and reaches a critical value of forming a 3–D network structure a highly viscous gel, rendering the solution unspinnable. It was observed visually that for chitosan concentrations below 2% the requirements for electrospinnability were not fulfilled, for concentrations above 4% the viscosity of the solution was too high, which resulted in an unstable jet. Further research on solutions containing between 2 and 4% chitosan revealed that 3% is the optimal value.
Figure 3 shows SEM picture taken from a 3% pure chitosan in 90% acetic acid solution. The success rate for obtaining nanofibers in strongly concentrated acetic acid solutions is based on the relatively higher concentrations of chitosan that can be obtained. Such concentrations result in an optimal viscosity for electrospinning, but also contribute to the degree of entanglement of polymer chains in the jet. The latter results in the formation of fibers instead of the ‘‘explosion’’ of the jet and the Taylor cone. The solution is strongly acidic, thus the polymers are well aligned in the electrical field because of the high fraction of protonated (thus charged) amine functions. This also promotes entanglement of the polymer chains.13

Figure 3 SEM picture taken from a 3% pure chitosan in 90% acetic acid solution at 20kV applied over a nozzle to collector distance of 10cm and a solution flow rate of 0.1mLh–1.
our attempted to electrospin of solution of pure CS by regulating the concentration of CS failed to obtain fibers except for strong acetic acid solutions with minimum fiber yield. Pure CS itself was difficult to electrospun. To obtain nanofibers, PVA was selected as a suitable partner for fabrication of nanofibers. When a small portion of PVA was mixed with CS (CS/PVA=20/80), uniform ultrafine fibers with average diameter around 80–120 nm were deposited on the collector. Figure 4 shows SEM images of electrospun nanofibers prepared from 20/80 solution of CS/PVAnfm.
The reported weight ratio was selected based on two factors including ease of electrospinnability and the nanofiber mechanical stability during all procedure. PVA was introduced in this study to reduce the viscosity of chitosan solution by interacting with chitosan through hydrogen bonding, rendering the solution spinnable at higher polymer concentrations. The solution viscosity is a critical factor that affects solution spinnability and morphology of as-spun fibers.14 Viscosity of a polymer solution is the characteristics of intermolecular interactions between polymer chains. The high viscosity of chitosan solution is due to the strong hydrogen bonding between NH2 and OH groups of chitosan polymer chains. The decrease in viscosity with addition of PVA can be attributed to the change in inter and intramolecular interactions of chitosan chains. PVA molecules bound onto chitosan backbone disrupt the self-association of chitosan chains by forming new hydrogen bonding between its OH groups and water molecules. Physically, this modulation in associative forces by PVA is manifested as an increase in chitosan solubility and a decrease in its solution viscosity.
Different values for the electrospinning parameters used in the preliminary study towards electrospinnability of CS/PVAnfm include Concentration, mass ratio, applied voltage (kV), distance from nozzle to collector (cm) and solution flow rate (mLh–1) have been optimized. In Table 2 it has been summarized the optimal values for electrosinning of CS/PVAnfm from acetic acid solution.
Solution type |
Conc. (%) |
CS/PVA weight ratio |
Flow rate |
Nozzle to collector distance |
CS/PVA |
[PVA]= 8 |
20/80 |
0.1 (mL h–1) |
10cm |
Table 2 Optimal values for electrosinning of CS/PVAnfm from acetic acid solution
It usually took 5 h to obtain a sufficiently thick membrane that could be detached from the collector. However, for longer production time it seems that the nanofibers split and form short side arms on the main fiber. This can be explained by distortion of the electrical field during fiber deposition. At the start of the experiment the electrical field distribution between nozzle and collector surface is homogeneous. After some time of fiber deposition the field is distorted in the vicinity of the deposited nanofibrous structure (in its pores) because this structure behaves as an isolator that is brought into the system between nozzle and collector. During electrospinning more and more chitosan fibers are deposited, thus an isolator is building up at the collector surface. This isolation becomes so intensive after some hours that the electrical field lines are distorted towards the edge of the collector (which is still free of nanofibres) and thus the jet is aligned along these field lines.13
Uptake of acid blue 26
The trapping ability of CS/PVAnfm was tested using Acid Blue 26 as a model system. The CS/PVAnfm was immersed into Acid Blue 26 solution and the change in absorbance of Acid Blue 26 was recorded as a function of CS/PVAnfm as a molecular filter by UV–vis spectrometry (Figure 5). It was observed that the absorbance of Acid Blue 26 solution decreased significantly by increasing the amount of CS/PVAnfm and blue solution become colorless, due to the removal of Acid Blue 26 from solution by CS.
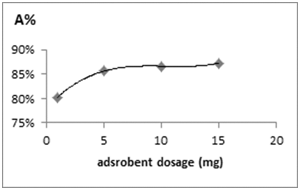
Figure 5 Effect of nanofiber dosage on sorption of Acid Blue 26 on CS/PVAnfm. Experimental conditions pH 7.8, Acid Bleu 26 concentration (10mg/L), reaction volume 10mL, contact time 4.5h, T=300C.
The CS/PVAnfm function as a molecular filter have been studied through electrostatic interaction of the Acid Blue 26 molecules with the CS molecules on the surface of the PVA fibers. It was observed that the higher the content of CS on the fibers, the more rapidly the Acid Blue 26 is removed from the solution. These findings are very promising and show the potential application for chitosan electrospun fibers to be used in filters for the removal of organic molecules. We expect that such chitosan electrospun nanofilters will be very applicable for waste treatments as long as the CS can interact with the target molecules.
Effect of nanofiber dosage
CS/PVAnfm dosage is particularly important because it determines the extent of de-colorization and may also be used to predict the cost of CS/PVAnfm per unit of solution to be treated. The effect of nanofiber dosage on the removal of Acid Blue 26 is presented in Figure 5. It was observed that the adsorption efficiency of Acid Blue 26 by CS/PVAnfm increases from 85.61% to 86.66% mg/g with the increase in nanofiber dose from 5 to 10mg in 10mL of 10mg/L Acid Blue 26 solution at 30°C, 150 rpm, 4.5h at a pH of 7.8. As expected, the adsorption density increases significantly as nanofiber dosage decreases. This is explainable because at a higher nanofiber dose extra adsorption sites are available for the adsorption. Also, by increasing nanofiber dose, the quantity of Acid Blue 26 adsorbed onto the unit weight of the nanofiber gets reduced, thus causing a decrease in qe value with increasing nanofiber dose. The CS/PVAnfm mass of 5mg was selected because an increase of the CS/PVAnfm mass to 10 mg did not provide significantly higher adsorption efficiencies (85.61%, 86.66% for 5mg and 10mg of CS/PVAnfm, respectively). Increasing CS/PVAnfm dose had a dramatic positive impact on color removal and there was an approximately linear relationship between CS/PVAnfm dose and color removal of the dye.15 Increase in adsorption with nanofiber dosage can be attributed to increased nanofiber surface and availability of more adsorption sites. However, if the adsorption capacity was expressed in mg adsorbed per gram of material, the capacity decreased with the increasing amount of sorbent. This may be attributed to overlapping or aggregation of adsorption sites resulting in a decrease in total nanofiber surface area available to the dye and an increase in diffusion path length. It was also indicated that the time required to reach equilibrium decreased at higher doses of the nanofiber.16,17
Sorption kinetics
In order to evaluate kinetic data, the agitation time was varied from 1 to 24h and the kinetic studies were done at 10 mg/L dye concentration and 5mg nanofiber dose. Dye removal was monitored with time. Kinetics of dye sorption governs the rate, which determines the residence time, and it is one of the important characteristics defining the efficiency of a nanofiber. Adsorption of dye by the polymer initially indicated rapid binding of dye and more than 50% adsorption takes place within 2 h, however with the passage of time adsorption rate slows down. Equilibrium was reached at 4.5 h and no change in the uptake capacity was observed up to 24h. The initial rapid binding increase is due to greater number of vacant sites available initially, resulting in a high concentration gradient between Acid Blue 26 in solution and Acid Blue 26 in the nanofiber.18 Figure 6 represents Kinetics of dye sorption on CS/PVAnfm.
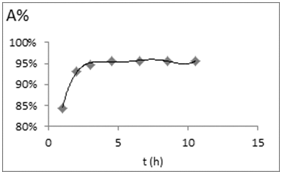
Figure 6 Kinetics of dye sorption on CS/PVAnfm. Experimental conditions pH 7.8, adsorbent dose (5,7,15mg), Acid Bleu 26 concentration (10mg/L), reaction volume 10mL, T=300C.
The rapid interaction between nanofibers and the dye molecule to be removed is desirable and beneficial for practical pollutant adsorption applications. During the process, the nanofiber surface is progressively blocked by the Acid Blue 26 molecules, becoming covered after some time. When this happens, the nanofiber cannot absorb any more dye molecules. As each particle purifies a certain volume of liquid, increasing the dosages rapidly promotes equilibrium between Acid Blue 26 and nanofiber because the number of particles to treat the same volume of liquid is increased. In general, the adsorption capacity increases with time and, at some point in time, reaches a constant value where no more dye is removed from the solution. At this point, the amount of dye being adsorbed onto the material is in a state of dynamic equilibrium with the amount of dye desorbed from the nanofiber. The time required to attain this state of equilibrium was termed the equilibrium time (te) and the amount of dye adsorbed at te reflected the maximum dye adsorption capacity of the nanofiber under these conditions. The process was initially very fast and then slowly reached equilibrium.19 Maximum accumulation occurs within 4–5 h for Acid Blue 26 dye on CS/PVAnfm and adsorption kinetics was relatively fast. Adsorption of dyes is fast at the initial stages of the treatment time, and thereafter, becomes slower near the equilibrium. It is obvious that a large number of vacant surface sites are available for adsorption during the initial stage, and after a lapse of time, it is difficult to occupy the remaining vacant surface sites due to repulsive forces between dye molecules adsorbed on the solid and those in the solution phase.
Sorption capacity
The amount of dye adsorbed per gram of the nanofiber was calculated by the difference between the initial and the final readings using the following equation:
(3)
where qe is the amount of dye adsorbed on the CS, Co, the initial dye concentration (mg/L), Ce, the equilibrium concentration of dye in solution (mg/L), V, the volume of dye solution used (L), and W is the weight of the CS/PVAnfm used (g) as nanofiber. Adsorption capacity is an important factor to evaluate, because it determines how much CS/PVAnfm is required to purify dye (analyte) from a given solution quantitatively.20 The maximum capacity of CS/PVAnfm for Acid Blue 26 was determined. 5mg of CS/PVA nanofiberous membrane were contacted with 10ml of dye solution containing 10mgL-1 dye molecules at an optimum adsorption pH of 7.8 and contact time of 4.5h. Then the concentration of the remaining dye in the solution was determined by uv-vis spectroscopy at the wave length of 635nm. The adsorption capacity (adsorption uptake rate) had been calculated and expressed in milligrams of dye adsorbed per gram of the (dry) nanofiber. The amount of dye adsorbed at equilibrium, qe, had been calculated from the mass balance equation given by Eq. (3). According to these results, the maximum amount of dye that can be absorbed by CS/PVAnfm has been found to be 392mgg-1 at a pH of 7.8.
In this study nanofibers based on CS blended with PVA were fabricated by electrospinning. Although electrospinning from aqueous solution of CS was unsuccessful, CS blended with a small amount of PVA could be electrospun into nanofibers without acids or organic solvents. FTIR results indicated the formation of hydrogen bond between CS and PVA molecule. According to spectroscopic methods, moreover, we demonstrated that the electrospun uniform CS/PVAnfm could capture some organic molecule readily and effectively. Since the composite nanofibrous membrane could be used as the attractive host matrix for the available loading of guest molecules and result in the enhanced spectrophotometric response. Thus, the specific property of CS functional nanofibrous membrane, in the case of super molecule interactions, would be exploited and combined with spectrophotometric detections.
None.
The author declares there is no conflict of interest.

©2018 Norouzi, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.