Journal of
eISSN: 2373-437X


Mini Review Volume 5 Issue 7
1Oregon Institute of Technology, USA
2University of Strathclyde, UK
Correspondence: May Al Doori, Oregon Renewable Energy Centre, Oregon Institute of Technology, Klamath Falls, Oregon 97601, USA
Received: December 07, 2017 | Published: December 19, 2017
Citation: Doori MA, Hunter IS (2017) Theoretical and Practical Studies on the Metabolic Engineering of Streptomyces for Production of Butanols. J Microbiol Exp 5(7): 00177. DOI: 10.15406/jmen.2017.05.00177
Production of biofuels is a significant technical and commercial challenge. To compete effectively with fossil fuels, the efficiency of processes that convert sustainable feedstocks to biofuel has to be improved. Ideally, the ultimate aim is a single process that degrades the renewable feedstock quickly and delivers high titers of biofuel at a price that is competitive with equivalent fuel from fossil sources. However, the microbes that are currently being investigated for biofuel production fall into two categories: those that degrade cellulosic feedstock but cannot make biofuels effectively and those that do make biofuels but are poor at degrading the cellulosic feedstock. When biofuel production is engineered into cellulose degraders, or cellulose degradation engineered into microbes with capability to produce biofuel, another factor, lignin content (a toxic component in most sustainable feedstocks), comes into play. This review considers the latent opportunity to harness Streptomyces bacteria to address this issue.
Keywords: Feedstocks, Production, Butanol, Decarbonisation, Streptomyces
Biofuels have emerged as one of the important sustainable fuel sources and are considered as a promising way to minimize Greenhouse Gas emissions and provide new energy sources.1‒3 Different countries have developed different strategies to introduce biofuels. The main concern is the economic drivers for each fuel, which varies with location and feedstocks. In addition, strategic and environmental aspects play a crucial role in the production and utilization of biofuels.4 Feedstocks, i.e. lignocelluloses, in the form of wood waste and straw are attractive alternative substrates, being available in large quantities in many geographic locations. Lignocelluloses contain carbohydrates in the form of cellulose (a polymer formed exclusively of glucose) and hemicelluloses (hetero polymers of various hexoses and pentoses in a branched structure). Invariably, they also contain lignin, a polymer of toxic aromatic compounds.5 To act as a feedstock, the cellulose and hemi‒cellulose must first be digested to low molecular weight sugars, and these are fermented to alcohols (Figure 1).
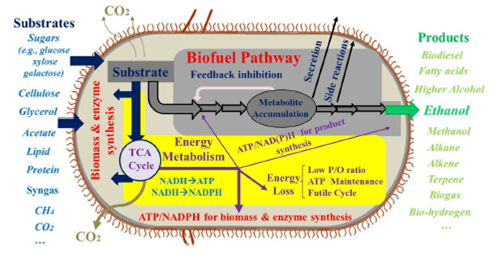
Figure 1 Carbon and energy limitation for biosynthesis. Reproduced from,12 with permission.
Microbes make a significant contribution to the production of biofuels.6,7 However, the yield of product by native microbes is uneconomic, which makes it necessary to develop and improve them through strategies of systems and synthetic biology.8,9 Alcohol producers, such as Saccharomyces cervisiae which are excellent at fermenting sugars to alcohols, cannot digest lignocelluloses. A combination of pretreatment10,11 of the feedstock and genetic engineering of the yeast, results in utilization of the cellulose and hemi‒cellulose, but hydrolysis products of lignin are toxic to yeast. Other natural alcohol producers, e,g. the bacterium Zymomonas mobilis, have similar problems of lignin toxicity.
Therefore, the choice of organism(s) should be based on those that have a proven ability to degrade lignocelluloses, be impervious to toxicity of lignin and be comparatively fast growing. They should also be amenable to genetic manipulation and ideally have been used industrially for large scale fermentation. This mini‒review considers the latent potential of Streptomyces bacteria to make a significant contribution to biofuel production, using biobutanol as an example. Although notable for antibiotic production, these resilient bacteria seem to have been overlooked in the context of biofuel production.
Review of the literature and discussion
The soil Actinomycetes are notable as being adopted by industry, particularly for large‒scale production of antibiotics. Most actinomycetes do not normally produce significant quantities of alcohols. However, introduction of the key enzymes into strains can stimulate alcohol (ethanol) production. The critical enzymes are pyruvate decarboxylase and alcohol dehydrogenase. Corynebacterium glutamicum12,13 and several Streptomyces sp.14 have been manipulated successfully for ethanol production. Although ethanol has been the focus of much research in the past few decades, its shortcomings as a fuel have led to metabolic engineering of microbial hosts to produce fuels with more desirable properties such as butanol, isobutanol medium to long‒chain hydrocarbons and isoprenoids.15,16 Bioethanol has been used in automobiles with gasoline in different blending proportions.17,18 However, biobutanol is considered an advanced biofuel, because of its good properties of high heat value, high hydrophobicity, reduced corrosive activity, high viscosity and low volatility. Biobutanol has great potential to be adopted in the future.19 Biological fermentative production of the advanced biofuel, n‒butanol, was first discovered by Pasteur in 1861 and later attributed to the Gram‒positive strict anaerobic bacterium, Clostridium acetobutylicum.20 Several other Clostridia species were also found to contain the acetone‒butanol‒ethanol (ABE) pathway21 for butanol production. Due to the requirement for anaerobic growth (resulting in slow growth), underdeveloped genetic tools and the clostridial strain being fastidious such as in spore formation, the five key reactions of the clostridial n‒butanol pathway were expressed heterogeneously in Escherichia coli to develop high yielding systems.22,23 This strategy of introducing the whole pathway could also be applied to Streptomyces, for which gene manipulation methodologies is now highly developed (for example24). Recent strategies that have been successful for Clostridia can be applied immediately to the Streptomyces.25 Finally, the production of butanol by microbes is often limited by the inherent toxicity of the butanol to the microbe – a major stumbling block in strain development in other species. It has been demonstrated that ribosome engineering in Streptomyces can confer enhanced tolerance to butanol t and hence the productivity of biofuel production is not limited by its inherent toxicity to the producing organism.26
Given the complexity of the many types of biomass, biochemical conversion of plant material(s) to fuels or chemicals after pre‒treatment remains a complex challenge (Figure 2). Cellulose is the predominant component of sustainable feedstocks and significant progress has been made in development of assays for cellulases.27,28 To initiate the deconstruction of plant matter, a number of methods, collectively called ‘Pretreatment’ have been developed to initiate the breakdown process. This pretreatment step is a key process to enable efficient conversion of lignocellulosic feedstock to biofuel or other fermentation products. These methods range from alkaline, mildly acidic to strongly acidic conditions to attack of the biomass structure.29
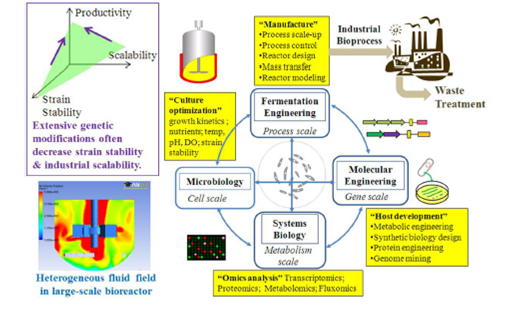
Figure 2 The design‒engineering‒analysis cycle for scale‒up of biofuel fermentation. Reproduced from,12 with permission.
Cellulases are needed to break down the complex components of cellulosic feedstocks while also processing the pathways to ferment the resulting sugars into alcohols but there are few, if any, naturally occurring microorganisms that combine these features. One approach is therefore to introduce the key genes encoding the desired cellulolytic enzymatic activities from other species into host organisms that are already optimized for ethanol fermentation.30 Here, the natural habitat and lifestyle of Streptomyces, which is soil‒based and often dependent on degradation of plant material, points to that inherent capability. The realization that some Streptomyces are prestigious at degradation of lignocelluloses31 and many have highly evolved enzymes, such as the laccases for lignin degradation,32 bodes well that Streptomyces could provide a robust organism chassis on which to build the capability to degrade lignocelluloses effectively. To enhance that approach, genetic engineering could be used to introduce to alcohol producing microbes the metabolic pathways needed to utilize sugar sources, such as the five carbon xyloses and other pentoses that are released from hydrolysis of woody biomass.33 Much of today’s commercial activity using advanced biotechnology for biofuels production processes on the ‘creation; selection or improvement of strains of desired microorganisms having enhanced properties of these traits’.34
Production of biofuels is expected to offer new opportunities to diversify income and fuel supply sources, to promote employment in rural areas, to develop long term replacement of fossil fuels, and to reduce greenhouse gas emissions ‒ boosting the decarbonisation of transportation fuels and increasing the security of energy supply. Biofuels contribute to lower combustion emissions than fossil fuels, per equivalent power output. They can be produced using existing technologies and be distributed through the available distribution system. For this reason, biofuels are currently pursued as a fuel alternative that can be easily applied in the short term, until other options that are harder to implement, such as hydrogen, are available. Despite biofuels being currently more expensive than fossil fuels, their production is increasing around the world.
Encouraged by policy measures and targets for use as transport fuel, global production of biofuels is estimated to be over 35 billion litres.35 Recent research efforts have concentrated on applying metabolic engineering and modern genetic methods in order to develop organisms optimized for high productivity and energy value, in order to achieve their full processing capabilities.36 It is for these reasons that Streptomyces bacteria should be considered as serious contenders for an integrated approach for production of biofuels from cellulosic feedstocks.
None.
None.

©2017 Doori, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.