Journal of
eISSN: 2471-1381


Case Report Volume 1 Issue 3
1Department of Gastroenterology, Indira Gandhi Institute of Medical Science, India
2Apollo Gleneagles Hospital, India
3Department of Gastroenterology, Sawai Man Singh Medical College, India
Correspondence: Ashish Kumar Jha, Department of Gastroenterology, Indira Gandhi Institute of Medical Science, Patna-14, India, Tel 9161 2229 7631, Fax 916122297225
Received: August 20, 2015 | Published: December 16, 2015
Citation: Jha K, Goenka, Goenka MK, et al. Endoscopic treatment of portal hypertensive biliopathy: intervene with caution!. J Liver Res Disord Ther. 2015;1(3):58-61. DOI: 10.15406/jlrdt.2015.01.00012
Portal hypertensive biliopathy (PHB) is defined as biliary abnormalities associated with portal hypertension, particularly caused by extra hepatic portal venous obstruction. Clinical spectrum ranges from no symptoms to biliary obstruction and life threatening cholangitis. PHB is an unusual cause of hemobilia. We present three cases of PHB presented to us with varied clinical features and managed with endoscopic interventions. Here, we highlight the outcome of endotherapy in patients of PHB. We also review the literature regarding the pathogenesis, clinical features and management of PHB.
PHB, portal hypertensive biliopathy; TLC, total leukocyte count; PT-INR, prothrombin time; US, ultra sonography; MRC, magnetic resonance cholangiography; CBD, common bile duct; ERCP, endoscopic retrograde cholangio pancreatography; LFT, liver function tests; TIPS, transjugular intrahepatic portosystemic shunt
The term ‘‘portal hypertensive biliopathy’’ (PHB) also known as portal biliopathy refers to abnormalities of the entire biliary tract including Intrahepatic and extrahepatic bile ducts, cystic duct and gallbladder in patients with portal hypertension.1 This disease is also referred as pseudosclerosing cholangitis and portal hypertensive cholangiopathy. PHB is an unusual cause of biliary obstruction, cholangitis and hemobilia. Here, we present three cases of PHB and review the literature regarding the pathogenesis, clinical features and management of PHB.
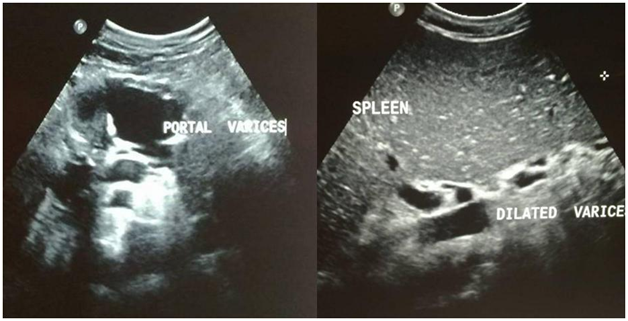
A 32-year- old man presented with jaundice. Eight-months ago, he had history of esophageal variceal bleeding due to extra hepatic portal venous obstruction (EHPVO) treated with band ligation. Examination showed icterus, pallor and splenomegaly. The blood investigations revealed haemoglobin 10.7 gm%, total leukocyte count (TLC) 2700 cells/cumm, platelet count 0.45 lac/cumm, serum bilirubin 23.0 (direct 12.0) gm%, serum albumin 4.0 gm%, Prothrombin time (PT-INR) 1.26, alanine aminotransferase 46 IU/L, aspartate aminotransferase 91 IU/L, gamma glutamyl transferase 443 IU/L, and alkaline phosphatase 148 IU/L. Abdominal ultrasonography (US) revealed dilated intrahepatic biliary radicals (IHBR), dilated portal vein (PV), collaterals at peri pancreatic and splenic hilar location; and splenomegaly with calcific foci (Figure 1). Magnetic resonance cholangiography (MRC) showed cholelithiasis, suspected choledocholithiasis, and a tiny filling defect in left hepatic duct and lower common bile duct (CBD) stricture. Endoscopic retrograde cholangiopancreatography (ERCP) showed irregular lower CBD stricture with dilated IHBR. Biliary sphincterotomy, bile duct clearance and placement of two 10 FG plastic stents was done. The follow-up laboratory parameters were improving. Patient was discharged with advice to follow-up for stent removal/exchange.
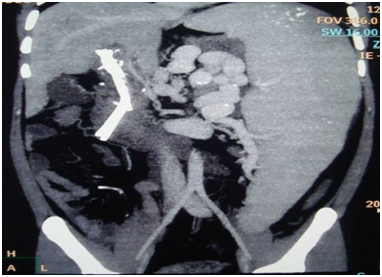
Four weeks later, the patient presented with malena. Haemoglobin level was 9.7 gm%. Esophagogastroduodenoscopy showed esophageal varices, portal hypertensive gastropathy and hemobilia (Figure 2). CT splenoportovenography revealed thrombosed PV with evidence of extensive cavernoma formation (Figure 3). Splenomegaly with dilated and tortuous splenic vein was also noted. Intravenous terlipressin was failed to stop hemobilia. Repeat ERCP performed and two more 10 FG plastic stent was placed (Figure 4). Idea behind placing more stents was to cause tamponade of bleeding venous plexus. Bleeding stopped and patient was discharged from the hospital.
Two weeks later, patient again presented with low grade fever. Blood investigation showed neutrophilia (TLC-5590/cumm with 87% polymorphs); and bile culture revealed growth of Klebsiella pneumonia and Enterococcus faecalis. There was no further deterioration of liver function tests (LFT). Fever was persisting, despite 72 hours of adequate antibiotic therapy. Therefore, a repeat ERCP was performed. All old four stents removed and three new biliary stents placed. The fever settled after four days of ERCP.
Five days later, patient again presented with hemobilia. Conservative therapy with intravenous terlipressin was successful in controlling the hemobilia. Later on, the porto-systemic shunting and cholecystectomy was performed. Patient was reevaluated after four months of surgery and biliary plastic stents were removed. No further complications were noted.
A 20- year- old man presented with jaundice and pruritus. Past history revealed recurrent low grade fever and growth retardation. Examination showed icterus, pallor, scratch mark and massive splenomegaly. The blood investigations revealed haemoglobin 11.0 gm%, total leukocyte count (TLC) 1580 cells /cumm, platelet count 1.25 lac /cumm, serum bilirubin 20.0 (direct 12.0) gm%, serum albumin 3.5 gm%, Prothrombin time (PT-INR) 1.2, alanine aminotransferase 112 IU/L, aspartate aminotransferase 114 IU/L, and alkaline phosphatase 208 IU/L. US showed hepatosplenomegaly, dilated IHBR, PV thrombosis, periportal cavernoma, and collaterals at splenic hilum. MRC showed dilated IHBR and stricture at upper bile duct involving bile ducts confluence (Figure 5). Diagnosis of EHPVO and portal biliopathy was made. ERCP showed irregular upper CBD stricture extending up to hilum. Biliary sphincterotomy followed by placement of a 10 FG plastic stent was done. The follow-up laboratory parameters were improving. Patient is advised for definitive shunt surgery, but he is not willing to go for surgery. Patient is on regular stent exchange.
A 39- years- old man with known cryptogenic cirrhosis of liver presented with pain abdomen. Examination revealed icterus, pallor, and splenomegaly. The investigations showed anaemia, pancytopenia, elevated serum bilirubin and alkaline phosphatase. US showed splenomegaly, periportal cavernoma, CBD stone, distal CBD stricture and dilated IHBR. ERCP showed small irregular distal CBD stricture and small CBD stone. Biliary sphincterotomy, stone extraction followed by placement of a 10 FG plastic stent was done. After 7 weeks, the patient presented with septic shock and cholangitis due to blocked stent. Endoscopic stent exchange was done. Patient recovered well. Six-months later, repeat session of stent exchange was done. Finally, we were able to remove stent without further development of biliary obstruction.
The frequency of PHB in patients with EHPVO (81-100%) is much higher as compared to patients with cirrhosis of liver (0-33%).2‒4 However, only a small proportion of these individuals are symptomatic. PHB is a relatively rare cause of biliary obstruction. As compared to Western Countries, PHB due to EHPVO is more common in India and other underdeveloped countries. But symptomatic PHB requiring endoscopic therapy is not common even in this part of World. In last 5year, we have experience of 3 cases of PHB managed by endoscopic methods. Clinical features of PHB include pain abdomen, jaundice, pruritus, hemobilia, fever and cholangitis. Long term biliary obstruction may lead to secondary biliary cirrhosis.
The exact pathogenesis of PHB is still not well known. External pressure of portal cavernoma and biliary tract ischaemia are two postulated theories of pathogenesis of PHB.5 The venous drainage of biliary tract is accomplished through the epicholedochal and paracholedochal venous plexuses. The epicholedochal venous plexuses of Saint is a fine reticular network on the outer surface of CBD and hepatic ducts becomes dilated and causes fine irregularities in the biliary tract.6 The paracholedochal venous plexuses of Petren runs parallel to the CBD and is connected with the gastric, pancreaticoduodenal and portal vein, and to the liver directly; It’s dilatation causes extrinsic compression over CBD.7 The paracholedochal collaterals can protrude into the lumen of CBD (Intracholedocal plexus) by two possible mechanism of herniation or perforation.8 The dilated paracholedochal plexus can give rise to perforators that perforate the muscular layer of the CBD and travel toward the liver hilum in the sub endothelial layer of the CBD (known as subepithelial Varix).9 Vascular neogenesis and formation of connective tissue occurring with increasing duration of thrombosis, which results in tumour like solid tissue around ducts. An extension of the thrombotic process to small venules of the bile ducts causing ischaemia of the bile duct.5 The changes in biliary tract due to cavernoma formation are usually reversible; however, ischemic changes are irreversible after shunt surgery.
US abdomen is the initial screening tool for the diagnosis of PHB. US is able to identify a dilated PV, thrombosed PV, cavernoma, CBD stones, dilated IHBR gall stones and splenomegaly. Magnetic resonance imaging cholangiography and portography is non-invasive method of choice for diagnosis of PHB. MRC is especially useful in delineating biliary stricture and portal cavernoma. Shin et al. described biliary changes as varicoid (reversible), fibrotic (irreversible) and mixed type on the basis of pathogenesis. CT splenoportovenography is alternative non-invasive modality for the evaluation of PHB.10 Endoscopic ultrasound appears to be a promising tool in the demonstration of cavernoma and differentiating stone from choledochal varices.11 ERCP is modality of choice both for diagnosis and treatment. Because of the potential complications, ERCP is usually indicated for therapeutic interventions.12
Treatment is indicated only for the symptomatic patients. Treatment options for PHB include endoscopic, surgical, radiological and medical therapy.2,12,13 Endoscopic interventions include papillotomy, stone extraction, nasobiliary drainage and stenting to relieve biliary obstruction.12 It is successful in patients with CBD stones, or dominant biliary stricture. Balloon dilatation of the stricture may be required before stent insertion. Biliary stenting could also lead to permanent stricture resolution. Urgent ERCP is warranted in patients presented with cholangitis. Although, successful endoscopic therapy have been reported on multiple occasions, it may be dangerous if interventions such as papillotomy, dilatation or stone extraction are performed in the presence of collaterals in the region. Reports have showed higher risk of bleeding and cholangitis in patients of PHB treated with endoscopic methods.12 Blockage of plastic stents occurs more frequently in patients of PHB. Very tight strictures particularly located high in bile duct; hilum and hepatic ducts have higher chances of complication of endotherapy. Extra- carefulness is required to avoid serious complications of endotherapy. Surgery has been the usual therapy in case of failure of endoscopic therapy. Endoscopic therapy does not treat its underlying cause and its long term success is poor. Therefore, endoscopic treatment is not a definitive treatment.12,13,14
Porto-systemic shunt surgery with or without hepato-biliary bypass is the established treatment method in a fit patient.13,14 Surgical approaches to the biliary system in PHB are hazardous as a result of the presence of collaterals in the region. By decompressing these collaterals, porto-systemic shunt surgery usually relieves the choledochal obstruction in PHB and, makes access to the region possible for further bypass surgery. Varicoid type of stricture caused by compression of bile duct by choledochal venous plexus is reversible after shunt surgery. Endoscopic management is an option in patients with cholangitis and those who is surgically unfit. Shunt surgery is indicated in fit patients, since it could provide definitive treatment in a young patient. Shunt surgery usually relieves the biliary obstructive symptoms particularly in varicoid type. Medical therapy with terlipressin, somatostatin or octreotide has been shown successful in patients with hemobilia and during surgery to reduce blood loss.15 Transjugular intrahepatic portosystemic shunt (TIPS) have been used for reduction of compression caused by portal cavernoma.16 Liver transplantation is indicated for decompensated secondary biliary cirrhosis.
Portal biliopathy is an unusual cause of biliary obstruction in patient of portal hypertension. Endoscopic therapy is very effective therapeutic modality; however it carries high risk of haemobilia, stent blockage and cholangitis. Extra- carefulness is required to avoid serious complications of endotherapy.
None.
Author declares that there is no conflict of interest.

©2015 Jha, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.