Journal of
eISSN: 2373-4396


Review Article Volume 14 Issue 3
1Department of internal Medicine, University of Campania Luigi Vanvitelli, Italy
2Department of Cardiology, Monaldi Hospital, Italy
Correspondence: Prof. Federico Cacciapuoti, Department of Internal Medicine, Campania University “Luigi Vanvitelli” Piazza L.Miraglia, 2 80131-Naples-Italy, Tel 081/5665022
Received: May 06, 2021 | Published: May 20, 2021
Citation: Cacciapuoti F, Cacciapuoti F. Heart failure in patients with high homocysteine levels. J Cardiol Curr Res. 2021;14(3):50-54. DOI: 10.15406/jccr.2021.14.00509
Increased levels of homocysteine (HHcy) can induce both systolic and diastolic left ventricular (LV) dysfunction that may evolve until chronic heart failure (HF). It is known that HHcy acts as an independent risk factor for atherosclerosis and hypercoagulation. This condition may cause coronary artery disease (CAD. Chronic reduction of oxygen supply to the myocardium can induce a reduction in LV contractility. These conditions can further evolve towards HF with reduced ejection fraction (HFrEF). But, HHcy can be also responsible for adverse cardiac remodeling for accumulation and proliferation of interstitial collagen, increased fibrosis and myocardial stiffness and preserved LV ejection fraction (HFpEF). Both HF can be further favoured by the advanced age, hypertension, diabetes and others. The administration of some nutrients, such as folic acid and B12 vitamin can delay or avoid these cardiac complications. However, the cellular mechanisms behind the adverse effects of Hcy on cardiac remodeling and pump function are not understood.
Keywords: homocysteine, left ventricle, systolic heart failure, diastolic heart failure, ejection fraction, micronutrients
HF, heart failure; HfrEF, heart failure with reduced ejection fraction; HfpEF, heart failure with preserved ejection fraction; HfmEF, heart failure with mid-range ejection fraction; Hcy, homocysteine; HHcy, hyperHomocysteine; SAM, S-adenosyl-methionine; MAT, methionine adenosyl transferase; CH3, methyl group; SAH, S-adenosyl-homocysteine; SAHH, S-adenosyl homocysteine hydrolase; CBS, cystationine-beta-synthase; CAD, coronary artery disease; MI, Myocardial Infarction; ED, endothelial dysfunction; NO, nitric oxide; NOS, nitric oxide synthase; ADMA, asymmetric-d-methyl arginine; LV, left ventricle; LVH, left ventricular hypertrophy; SH, systemic hypertension; DM, diabetes mellitus; NF-kB, nuclear factor-kB
Heart Failure (HF) is a major public health issue worldwide, responsible for considerable rate of morbidity and mortality. Its incidence wanders about 2% in adults and increases to 6-10% in persons>65years.1 In the population>55years, chronic HF was found in about 4%, reaching 13% in subjects aged 75-84years.2 In the United States, HF affects about 6,5millions adults. Its incidence increases with advancing age, and HF prevalence in elderly people will reach to 45%-46% between 2012 and 2030. Older adults with HF have a 5-year mortality about 50%.3,4 In Italian people, an HF-incidence of 1.44% was reported in a recent study.5 This value increases with advancing age.6 Referring to the ontractility, two types of left ventricular (LV) ejection fraction% (EF%) were identified: HF with reduced EF% (HfrEF), in which LVEF is below 40% and HF with preserved EF% (HfpEF), in which LVEF exceeds 50%.7 Recently, HF with borderline or mid-range EF% was also described (HfmEF).8
Many factors, including ischemic artery disease, systemic hypertension, diabetes mellitus, dyslipidemia, valvular heart disease can induce both HfrEF and HfpEF.9 But, an increase of homocysteine (Hcy) level may also act as a risk factor for HF.10
Homocysteine
Hcy is a sulfur amino acid produced during the metabolism of Methionine.11 An increase of Hcy serum concentration (HHcy) can happen for a defective metabolism of Methionine, resulting for either mutation in genes coding for the enzymes of Hcy metabolism or deficiencies of certain vitamin cofactors.12,13 Genetic alterations of some enzymes acting in the Hcy metabolism, some vitamin deficiencies that act as coenzymes14,15 or an increased intake of Methionine, certain drugs, some diseases, pregnancy and others can also induce HHcy.16–18 After the conversion of Hcy in Methionine, this is converted to S-Adenosyl-Methionine (SAM), an energetic compound, through the catalytic action of the enzyme methionine adenosyl-transferase (MAT), responsible for the transfer of a methyl group (-CH3) of SAM to an acceptor molecule. Consequently, SAM is converted in S-Adenosyl-Homocysteine (SAH). Finally, SAH is divided in Hcy and Adenosine through the enzyme SAH-hydrolase (SAHH).19–21
Hcy can be also further metabolized through the trans-sulfuration pathway. In the first step, the enzyme cystathionine ß-synthase (CBS) catalyzes the condensation of Hcy and serine in Cystathionine, using vitamin B6 as cofactor.22 Subsequently, Cystathionine is metabolized until to Cysteine. This compound favours the synthesis of Glutathione, the most important antioxidant of the body (Figure 1).23
HCY metabolic cycle

Figure 1 Metabolic cycle of homocysteine with remethylation, transsulfuration and transmethylation routes.
HHcy and HfrEF%
HHcy is a cause of early and massive atherosclerosis, that may involve all arterial vessels, mainly coronary arteries.24 In the long run, coronary artery disease (CAD) can induce or not acute myocardial infarction (AMI).25,26 In turn, CAD (with or without AMI) causes a reduction of myocardial contractility for defective oxygen supply to the myocites and subsequent left ventricular (LV) dilation. These changes achieve LV dysfunction that may evolve subsequently towards HF with reduced EF% (HfrEF%).27,28 Endothelial dysfunction and thrombosis (hypercoagulability) are the main causes of CAD (Figure 2).
HHcy and HfrEF%
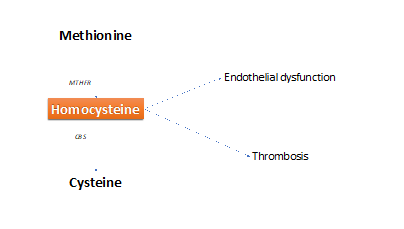
Figure 2 Two main effects (endothelial dysfunction and thrombosis) induced by homocysteine, that favor atherosclerosis.
Endothelial dysfunction
In detail, endothelial dysfunction (ED) can induce CAD for reduced nitric oxide (NO) bioavailability.29 The reduced NO bioavailability depends from reduced activity of NO synthase (NOS). On the other hand, the reduced NOS activity is dependent from the activity of Asymmetric-D-Methyl-Arginine (ADMA), present in excess when plasma Hcy concentration increased.30,31 The reduced NO biovailability is responsible of excessive systemic vasoconstriction, with reduced peripheral tissue perfusion.32 In addition, ED can induce water and sodium retention, and increased neurohormonal activation (activation of the sympathetic nervous system and the renin-angiotensin system). The reduced NO bioavailability also favours the production of some vasoconstrictors, mainly endothelin-1 (that further deteriorates vascular resistance), and causes smooth muscle cell growth and matrix metalloproteinase production.33
Role of thrombosis
Another mechanism inducing CAD are the platelets’ activation, their increased aggregation and adhesion,34 with consequent hypercoagulability. In turn, this condition promotes coronary atherosclerosis, for the imbalance of procoagulant/anticoagulant ratio, with reduced oxygen supply to the myocardial cells.35 But, HHcy also activates Nuclear Factor-kB (NF-kB) responsible for chemokines’ expression and subsequent inflammation of vascular wall (early stage of atherosclerosis). That a can evolve towards LV dysfunction and HfrEF%.36
HHcy and HfpEF%
Elevated Hcy levels can also promote HfpEF, mainly in patients simultaneously suffering for systemic hypertension (SH) with or without left ventricular hypertrophy (LVH) and/or diabetes mellitus (DM). That happens via promoting increased LV mass37 through growth of myocardial cells and collagen production.38 In addition, HHcy can induce cardiac fibrosis by increasing transforming growth factor ß1,39 and stimulates matrix metalloproteinase (MMP) production(Figure 3).40 In particular, MMP-2 was found significantly high in HfpEF-patients before the occurrence of symptoms, even if its pathogenic role is unknown.41 Finally, referring to the relationship between HHcy and HfpEF%, the Framingham Heart Study reports that plasma HHcy is directly related to left ventricular mass and wall thickness.42 A previous study of Cesari et al. confirmed a strong association between HHcy and HfpEF%.43
Conclusive remarks
HHcy can favor both systolic and diastolic dysfunction with different mechanisms.7 In turn; LV systolic dysfunction may further evolve in chronic HfrEF. That mainly happens in CAD induced by HHcy, with or without MI.25,26 Concerning that, Gueant-Rodriguez et al. found a prevalence of HHcy both in patients with CAD and in those without.44 On the contrary, LV diastolic dysfunction dependent of interstitial vascular fibrosis and increased myocardial stiffness, may subsequently evolve in HfpEF marked by preserved or lightly reduced LV contractility. As previously affirmed this is commonly observed in elderly HHcy-patients with co-existence of other diseases (diabetes mellitus, dyslipidemia, systemic hypertension, etc.) and is associated with an increased risk of death at 5years.45 It must also added that the prognosis is better in HfpEF% in patients than in those with HfrEF.46
Referring to the prevention of chronic HF, it is known that deficiencies of folic acid and vitamins B6-12 are the most common causes for mild HHcy. But, Hermann et al. affirmed that HHcy “per se”, and not B vitamins, acts in HfpEF.28 On the contrary, these micronutrients could help to r chron iceduce HHcy and thus, on the beginning and the progression of HfrEF.47 In this regard, Witte et al. reported a 13% reduction in LV volumes and a 5% increase EF% in patients with HfrEF% after 9-months of folates, vitamin B6, vitamin B12.48 Thus, the relationship between mild HHcy and HfpEF remains largely unexplored too, while the connection between HHcy, and HfrEF seems to be more evident. However, the vitamins-B supplementation in HHcy-patients both with HfrEF% and HfpEF% ameliorates the quality of life.48
Conclusively, the relationship between HHcy and HF is insufficiently known too, and future studies needed to clarify the definite role of vitamins B6-9-12 supplementation to prevent or minimize especially HfpEF, in these patients.
None.
The rest of the authors declare do not have conflicts of interest.
None.
The Authors have no conflict of interests.

©2021 Cacciapuoti, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.