Journal of
eISSN: 2373-4396


Research Article Volume 12 Issue 1
1Department of cardiology, Ain Shams University Hospital, Egypt
2Department of Chest Diseases, Ain Shams University Hospital, Egypt
Correspondence: Hazem M Khorshid, Department of cardiology, Ain Shams University Hospital, Egypt, Tel 20224821894
Received: January 03, 2019 | Published: January 10, 2019
Citation: Khorshid H, Fattah KHEA, Salem HM, et al. Effect of exercise based cardiac rehabilitation on patients with coronary artery disease and chronic obstructive pulmonary disease. J Cardiol Curr Res. 2019;12(1):21-26. DOI: 10.15406/jccr.2019.12.00423
Background: Coronary artery disease (CAD) and chronic obstructive pulmonary disease (COPD) are common treatable and preventable chronic diseases and it is not uncommon to co-exist. Rehabilitation is now considered an important part of the long term management in both diseases which include exercise, education and smoking cessation.
Objective: To study the effect of exercise based cardiac rehabilitation on quality of life, respiratory function and exercise tolerance in patients suffering from both CAD and COPD.
Methods: The study included 40 Patients suffering from CAD and COPD referred for cardiac rehabilitation unit at Ain shams university hospitals. Patients were subjected to clinical assessment, ECG, echocardiography, modified Bruce protocol, spirometry and St. George's Respiratory Questionnaire (SGRQ). All Patients were subjected to twice weekly exercise sessions of 12 weeks of Cardiac rehabilitation (CR) and were reassessed by modified Bruce protocol, spirometry and SGRQ after completion of the program.
Results: The study group showed statistically significant improvement regarding spirometric parameters (There was a statistically significant increase in FEV1% predicted and FEV1/FVC from 52.1+/- 8.6% to 56.3+/- 8% and 60.6+/-5.6 vs 63.9+/-5.5 respectively, P<0.001) There was also statistically significant improvement in the SGRQ score (77.1+/-13.1 vs 69.9+/-13.4, p<0.001) and exercise parameters on symptom limited exercise test by modified Bruce protocol (There was statistically significant increase in the METs from 8.3+/-1.9 to 10.4+/-1.7 p<0.001, HR reserve from 56.8+/-13.3 to 68.1+/-12.8 beats, p<0.001 and HR recovery at 1 minute post exercise from 13.7+/-6.1 to 16.3+/-6 beats, p=0.014) after completion of the exercise based CR program.
Conclusion: Cardiac rehabilitation has favourable impact on respiratory function, exercise tolerance and quality of life in CAD patients who also suffer from COPD.
Keywords: cardiac rehabilitation, CAD, COPD, respiratory function test
CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CR, cardiac rehabilitation; ECG, electrocardiogram; SGRQ, St. george's respiratory questionnaire; FEV1, forced expiratory volume in one second; FVC, forced vital capacity
Atherosclerotic cardiovascular and chronic pulmonary diseases are leading causes of mortality and morbidity worldwide.1 Coronary artery disease (CAD) and chronic obstructive pulmonary disease (COPD) share many risk factors, which include ageing, smoking, sedentary life style, obesity and metabolic diseases2 and the prevalence of respiratory comorbidity (eg, COPD, asthma) in patients with cardiac disease has varied in the literature between 9% to 39%.3,4 Comorbid cardiovascular disease (CVD) in patients with COPD is associated with more symptoms, higher rate of hospitalizations and increased mortality irrespective of the degree of airflow obstruction.5,6 On the other hand, multiple studies have shown a strong correlation between low forced expiratory volume in the first second (FEV1) and cardiovascular mortality.7 Patients with CAD and concomitant COPD have a higher incidence of major adverse cardiovascular events (myocardial infarction (MI), heart failure (HF) and mortality) compared to patients without COPD.8 Cardiac patients with COPD have higher mortality rates than those without COPD after percutaneous coronary intervention8,9 worse in hospital mortality and following discharge after an MI10,11 and long-term mortality post coronary artery bypass grafting (CABG) surgery as compared to patients without COPD.12−14
Exercise-based cardiac rehabilitation (CR) has been well-established in the effective management of patients with CVD with a positive impact on cardiovascular risk factors, quality of life, all-cause mortality and cardiac mortality in cardiac patients.15,16 Hence, the present study aimed at studying the impact of exercise based cardiac rehabilitation on quality of life, respiratory function and functional capacity in patients with CAD and COPD.
The present study included 40 patients presenting to the CR unit in Ain Shams University. It included both genders with age ranging between 30–60 years. The study was approved by the Ethics committee of our institute. Informed consent was obtained from the patients after explaining the objectives and study design ensuring privacy of their data.
The study included:
The following patients were excluded from the study: patients with significant arrhythmias or heart block, fixed rate pacemaker, decompensated heart failure, unstable coronary syndromes, Active pericarditis or myocarditis, significant valvular regurgitation, valvular stenosis, prosthetic valves, patients in acute infective exacerbation of COPD, patients with hypoxia at rest or exercise, other respiratory disease, neuromuscular disease or disability that result in inability to perform physical activity. Also patients refusing to participate were excluded.
Initial patients’ assessment
All patients were subjected to baseline thorough history taking, full clinical examination, 12 lead resting ECG and baseline echocardiographic examination. Outcome measures were assessed at baseline and after completion of the CR program in the form of:
Exercise based cardiac rehabilitation protocol
All the patients were subjected to formal CR program in our institute which included medical assessment, risk factor modification, psychosocial management, nutritional and physical activity counselling as well as treadmill exercise training. The patients were subjected to moderate intensity exercise training twice/week for 3 months achieving target heart rate of 40-60% of HR reserve calculated from pre-exercise symptom limited exercise stress test by modified Bruce protocol. The duration of each session was 30-45 minutes. The progression of exercise intensity was monitored by continuous ECG monitoring, recording of heart rate, blood pressure and symptoms as well as the rate of perceived exertion (RPE) on Borg scale where the patients were exercised at an RPE of 11–13 in the absence of symptoms. Only the patients who completed the 3 month course of the CR program in our unit attending > 80% of the sessions were included in the study.
Statistical analysis was done using IBM SPSS® Statistics version 22 (IBM® Corp., Armonk, NY, USA). Numerical data were expressed as mean and standard deviation or median and range as appropriate. Qualitative data were expressed as frequency and percentage. Paired t-test (for normally distributed quantitative data) or Wilcoxon-signed ranks test (for not normally distributed quantitative data) were used to compare two consecutive measures of numerical variables. A p-value<0.05 was considered significant.
Baseline demographic data, clinical risk factors and EF:
Table 1 summarizes baseline demographic data, clinical risk factors and baseline EF. The mean age of the study group was 47+/-8.7 years, 34 patients (85%) were males, 18(45%) were diabetic, 12(30%) were hypertensive, 27(67.5%) were smokers and 7(17.5%) had positive family history for premature CAD. All patients were in sinus rhythm, the mean ejection fraction (EF) was 48.8+/-8.1%.
Variable |
N = 40 |
|
Age (Years) |
47 +/- 8.7 |
|
Gender |
Males |
34 (85%) |
Females |
6 (15%) |
|
DM |
18 (45%) |
|
HTN |
12 (30%) |
|
Smoking |
27 (67.5%) |
|
+ve FH for CAD |
7 (17.5%) |
|
EF% |
48.8 +/- 8.1%. |
|
Table 1 Baseline demographic data, prevalence of Risk factors and EF
DM, diabetes mellitus; HTN, hypertension; FH, family history; CAD, coronary artery disease; EF, ejection fraction
Baseline SGRQ, exercise and respiratory function data
The baseline mean SGRQ score of the patients was 77.1+/-13.1. Regarding the baseline Exercise parameters, the mean resting HR was 77.9+/-6.3 bpm and the peak HR achieved during exercise was 134.7 +/- 11.5 bpm with HR reserve of 56.8+/-13.3 beats and mean METs achieved during exercise of 8.3+/-1.9. Heart rate recovery at 1 minute was 13.7 +/-6.1 beats. Regarding the baseline respiratory function test, the baseline FEV1 (% predicted) was 52.1+/-8.6% and FEV1/FVC was 60.6+/- 5.6 (Table 1).
Impact of cardiac rehabilitation on exercise parameters
After completion of the CR program there was statistically significant increase in the METs (8.3+/-1.9 vs 10.4+/-1.7 p<0.001) (Figure 1), HR reserve (56.8+/-13.3 vs 68.1+/-12.8 beats, p<0.001) and HR recovery at 1 minute post exercise (13.7+/-6.1 vs 16.3+/-6 beats, p=0.014). There was also statistically significant decrease in resting HR after completion of the CR program (77.9+/-6.3 vs 67+/-6 bpm, p <0.001). There was no statistically significant change in the peak heart rate achieved during exercise testing (Table 2).
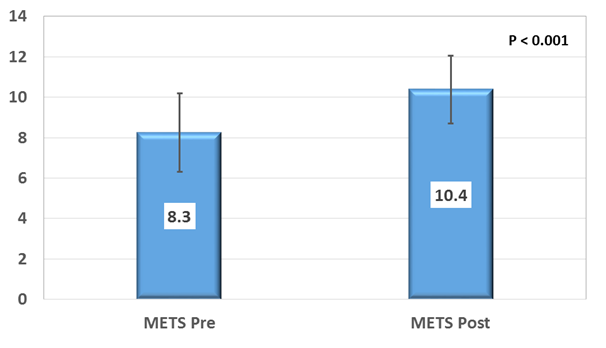
Figure 1 METs achieved on Symptom limited treadmill exercise stress test using Modified Bruce protocol: Baseline and after completion of CR program.
Baseline |
After 3 month |
P value |
|||
Mean |
SD |
Mean |
SD |
||
METs |
8.3 |
1.9 |
10.4 |
1.7 |
<0.001 |
RHR (bpm) |
77.9 |
6.3 |
67 |
6 |
<0.001 |
PHR (bpm) |
134.7 |
11.5 |
135.1 |
11.5 |
0.487 |
HR Reserve (beats) |
56.8 |
13.3 |
68.1 |
12.8 |
<0.001 |
HR1 (bpm) |
121 |
11.9 |
118.8 |
12.1 |
0.027 |
HRR1 (beats) |
13.7 |
6.1 |
16.3 |
6 |
0.014 |
FEV1 (% predicted) |
52.1 |
8.6 |
56.3 |
8 |
<0.001 |
FEV1/FVC |
60.6 |
5.6 |
63.9 |
5.5 |
<0.001 |
SGRQ |
77.1 |
13.1 |
69.9 |
13.4 |
<0.001 |
Table 2 SGRQ, Exercise and Respiratory function data: Baseline and after completion of the CR program
MET, metabolic equivalent of task; RHR, resting heart rate; PHR, peak heart rate; HR1, heart rate at one minute during recovery; HRR1, heart rate recovery at 1 minute into recovery; FEV1, forced expiratory volume in one second; FVC, forced vital capacity, SGRQ: St. george's respiratory questionnaire
Respiratory function tests and SGRQ after cardiac rehabilitation
There was a statistically highly significant increase in FEV1 and FEV1/FVC after completion of the CR program (52.1+/-8.6% vs 56.3+/-8% and 60.6+/-5.6 vs 63.9+/-5.5 respectively, P<0.001) There was also statistically highly significant improvement in the SGRQ score (77.1+/-13.1 vs 69.9+/-13.4, p< 0.001) (Table 2) (Figure 2).
Effect of exercise based CR on respiratory function
In the present study exercise based CR was associated with significant improvement of the respiratory function tests (FEV1% and FEV1/FVC) in patients with CAD and COPD. This association is supported by previous studies.20−24 The FIRST (FEV1 as an Index of Rehabilitation Success over Time) study prospectively compared the changes in FEV1 in 190 COPD patients undergoing supervised exercise training to those in 67 drug treated patients: in the exercise training group, FEV1 (% of predicted) increased from 57.3% to 60.8% after 3 years, whereas in the controls the values were decreased from 55% at baseline to 51% after 3 years(P<0.001).21 Improvement in FEV1 was also reported in patients with very severe COPD after a 3-week exercise training program (including endurance and strength exercise training; aiming at a workload of 60–80% of the personal maximum).22 Similar findings were also reported in patients hospitalised with acute exacerbation of COPD. Ali et al showed that 9 sessions of exercises in the form of walking, bicycle ergometry and resistance exercises (20 minutes each), thrice-weekly for three weeks resulted in statistically significant improvement in FEV1, FEV1 (% predicted) and FEV1/ FVC (%) as compared to the control group.23
The finding of the present study is also concordant with Elkhateeb et al.24 who showed that 6–8 weeks of aerobic exercise training program in patients with COPD (FEV1 < 80% predicated together with an FEV1/FVC <0.70), resulted in statistically significant improvement of FEV1 as compared to the control group.24 This is partially matching with Kaminsky DA et al, who showed that FEV1 had significantly improved after cardiac rehabilitation in patients with a baseline BMI > 30 kg/m.25 The hallmark of COPD is airflow obstruction caused by airway narrowing and loss of elastic recoil.26 The FEV1 declines over time during progression of the disease27 There is a general agreement on the key significance of FEV1 in evaluating the worsening of COPD over time21 and studies have shown value of FEV1 and its correlation with maximum aerobic capacity28 prognosis and mortality risk in the COPD patients.29 Aerobic exercise can decrease the airway resitance and hence increases the VC, TLC, FVC, and FEV1indices.30
Effect of exercise based CR on HRQL
In the present study, there was a significant improvement in HRQL as assessed by SGRQ. Cardiac rehabilitation has been shown to improve exercise capacity, quality of life, and functional status of CAD patients.31 There is also evidence of a positive effect of exercise on HRQL in COPD patients, as shown by previous studies despite the wide range of intensities (type, modality, duration, and frequency of exercise training) in the heterogeneous pulmonary rehabilitation programs included in these studies.32−37 This finding is supported by the systematic review of exercise based pulmonary rehabilitation in COPD patients by McCarthy et al which included 65 RCTs and showed statistically significant improvements in all domains of the SGRQ.38
Patients with COPD have limited functional capacity due to impaired skeletal muscle power, hormonal changes (reduced levels of anabolic steroids), systemic inflammation and increased energy expenditure at rest.39,40 The peripheral skeletal muscle shows both structural and biochemical changes in the form of decrease in the muscle mass with decrease in type I (oxidative) fibres and increase in type II (glycolytic) fibres, decreased capillary density as well as reduction of number of aerobic enzymes.41 Exercise training results in significant increase in oxygen uptake capacity (VO2 max). Blood lactate levels, minute ventilation (VE), dyspnoea and HR are decreased for the same level of exercise.42−46 The oxidative enzymes and oxygen extraction of skeletal muscle also are improved after exercise training47,48 This results in significant increase in endurance capacity and maximal workload.49
Effect of exercise based CR on exercise parameters
Regarding the exercise parameters, there was significant improvement in all exercise parameters in the present study. There was statistically significant increase in the METs, HR reserve and HR recovery at 1 minute post exercise (HRR1). There was also statistically significant decrease in resting HR after completion of the CR program. Exercise reconditioning has long been considered an essential component of the rehabilitation process. CR has been shown to improve functional capacity as measured by exercise tolerance and peak oxygen consumption in patients with CAD.50 Similar finding were reported in patients with COPD. Adults with COPD randomly assigned to an 8-week comprehensive rehabilitation program including supervised exercise training showed significant improvement in exercise endurance as measured by treadmill walking time which was still maintained at 6 months as compared to the control group.51
Randomized, controlled trials have clearly shown that exercise training (treadmill, cycling, free walking, stair climbing, or a combination of these) increases exercise endurance and 10-25% increase in walking distance.52−57
In the present study the statistically significant increase in HRR1 after completion of the CR program is concordant with Rodríguez et al who studied the effects of exercise training on autonomic cardiac function in COPD patients, where both the CR (chronotropic Response) and heart rate recovery (HHR1) improved by 45% [53.1(25.5) to 77.4(13.8); P=0.001] and 26% [12.3(10.4) to 15.6(10.0) bpm; P=0.03], respectively with significant improvement in HRR1 in patients with baseline autonomic dysfunction as compared with patients without baseline autonomic dysfunction.58 Similarly a systematic review by Snoek et al that included 8 studies (449 patients) showed that aerobic training increases HRR in patients with established heart disease.59
The decrease in HR after exercise represents withdrawal of the sympathetic nervous system and, more importantly, reactivation of the parasympathetic nervous system.60,61 An attenuated HRR after exercise denotes reduced vagal tone and has been established as an independent prognostic marker of mortality.62−64 Patients with chronic complex diseases develop autonomic cardiac dysfunction throughout their natural course65,66 and is associated with higher mortality rates.66,67 In patients with CAD, HRR is predictive of mortality independent of the left ventricular systolic function, angiographic severity of CAD and exercise capacity.68 Similarly, COPD patients have a lower HRR than healthy subjects, and have a worse prognosis when presenting abnormal HR.69 A decrease in the sympathetic nervous system tone, and subsequent increase in the parasympathetic response could explain the improvement in functional capacity following exercise training.70
Cardiac rehabilitation, incorporating supervised exercise training as the main component, has shown to improve respiratory function, quality of life and functional exercise capacity and hence it is recommended as an effective treatment option for patients with CAD and COPD, and that the presence of comorbidity of COPD in CAD patients should not hinder them from participating in exercise based CR programs. Future larger randomized studies are recommended to confirm the results of the present study.
None.
No financial interest or any conflict of interest exists.

©2019 Khorshid, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.