Journal of
eISSN: 2473-0831


Research Article Volume 6 Issue 4
Correspondence:
Received: November 18, 2017 | Published: December 5, 2017
Citation: Getaye M, Hagos S, Alemu Y, Tamene Z, Yadav OP (2017) Removal of Malachite Green from Contaminated Water using Electro-Coagulation Technique. J Anal Pharm Res 6(4): 00184. DOI: 10.15406/japlr.2017.06.00184
Electro-coagulation analytical technique has been used for the removal of malachite green - a dyestuff used as antimicrobial in aquaculture. Effects of parameters such as current density, pH, dye initial concentration, inter-electrode separation and amount of electrolyte on the dye color- removal have been investigated using Al and Fe as scarifying anodes. It was observed that whereas increase in dye initial concentration or inter-electrode separation, the rate of color removal continuously falls, however, on raising of electrolyte concentration, color removal rate enhances up to a definite electrolyte level, beyond that there was no further improvement in the color removal. Further, for achieving maximum color removal of malachite green aqueous solution optimum current density (76.5A/m2) and pH (8.0) of solution, is required. Complete color removal of 100mg/L malachite green aqueous solution could be achieved in 30min using electrolyte (NaCl) concentration 0.4g/L, inter-electrode distance: 1cm and optimum pH and current density. Performance of Al and Fe as sacrificial electrodes, in terms of their color removal efficiency, was almost similar except in their response to inter-electrode separation, as variable, where in case of Fe as anode, color removal rate declined more rapidly. The outcome of the present research work may be useful for an effective large scale treatment of industrial effluents contaminated with malachite green dye.
Keywords: Electro-coagulation; Malachite green; Electrolyte; Current density
COD: Chemical Oxygen Demand; BOD: Biochemical Oxygen Demand; EC: Electro-Coagulation; CD: Current Density
Considerable amount of unconsumed dyes from textile, paper and printing industries are being discharged into water bodies, every day. The presence of dyes and pigments in water causes considerable damage to the aquatic environment [1]. These contaminants result in high chemical oxygen demand (COD), high biochemical oxygen demand (BOD), toxicity, bad smell, and are mainly responsible for the coloration of wastewaters. The presence of color in water is undesirable since it blocks the sunlight access to aquatic flora and fauna thus reducing the photosynthetic action within the ecosystem [2]. Malachite green has been used as a food coloring additive, dyeing silk, jute, leather, wool, cotton and paper [3]. Moreover, this compound has also been used as a disinfectant and anti-helminthic, in aquaculture and as a fungicide and antiseptic [4].
The use of malachite green has been contested due to its effects on the reproductive and immune systems and its potential genotoxic and carcinogenic effects. A number of processes such as: incineration, advanced oxidation, ozonation, adsorption on solid phase and biological treatment, and have extensively been applied for the treatment of dye-containing wastewater [5-8]. But these are inefficient, involve high cost, environment unfriendly and generate large amount of sludge as a solid waste that requires secondary treatment. Electro-coagulation (EC) process being a clean, easy to operate and cost-effective due to lower equipment costs, has emerged as an alternative for the treatment of wastewater contaminated with pollutants [9,10].
Electro-coagulation (EC) is a complex process with a multitude of mechanisms operating synergistically for the removal of diverse pollutants from waste water. It involves electro-dissolution of metallic anodes followed by the formation of readily separable metallic hydroxides-pollutant coagulates. Compared with traditional flocculation and coagulation, electro-coagulation has the advantage of removing even the smallest colloidal particles [11,12]. Aluminum and iron are most commonly used anodic material in electro-coagulation due to their low cost and ready availability [13,14].
There are a number of reports on the use of electro-coagulation process applied for the treatment of waste water discharged from laundry [15], hospitals [16] and textile [17] industry. Mansoorian et al. [18] removed lead and zinc from battery industry wastewater using electro-coagulation process. They also studied the Influence of direct and alternating current by using iron and stainless steel rod electrodes. Recently, Takdastan et al. [19] have used electrocoagulation process for the treatment of wastewater contaminated with detergents and phosphates. In the present study, for further improving the efficacy and cost-effectiveness of electro-coagulation process, effects of variables viz current density, inter-electrode separation, the substrate as well as electrolyte concentrations, on the color removal of aqueous solution contaminated with model pollutant malachite green dye, has been investigated.
Chemicals
Aluminum and iron plates with dimension 15x7x1cm were obtained from the local market in Addis Ababa, Ethiopia. Malachite green (IUPAC name: 4-{[4-(dimethylamino)phenyl](phenyl)methylidene}-N,N-dimethylcyclohexa-2,5-dien-1-iminium chloride); molecular formula: C23H25ClN2 ; MW; 364.911g/mol, H.P. Chemicals, Ahmedabad, India, Analytical Grade; λmax: 619nm) ). Chemical structure of malachite green is presented in Figure 1.
Malachite green aqueous solution (1L) of known concentration was taken in a 2 L glass beaker at ambient temperature. The anode (Al or Fe) and cathode (mild Steel) plates, each 15x7x1cm in size and pre-cleaned using dilute HCl and distilled water, were immersed in the dye solution up to 7cm depth thus providing an effective surface area of each electrode as 98 cm2. The two electrodes were kept vertically and run parallel to each other separated by a wooden block. The desired distance between the electrodes was achieved by using the wooden block of appropriate width for maintaining constant current density, the electrodes were connected to a DC power supply using galvanostatic operational option. Schematic of electro-coagulation cell used, is shown in Figure 1. The desired conductivity level of dye solution was obtained by dissolving in it appropriate amount of NaCl and its pH was adjusted by adding required volume of 1M NaOH or 1M H2SO4 solution.
The current density (CD) was calculatedusing the following equation:
(1)
Where, I is the current (in amperes) passing through the dye solution and S is the area (in m2) of the electrode.
Absorbance of dye solution at regular interval was recorded at λmax = 619nm using a UV/Vis spectrophotometer (SP 65) (Figure 2). Percent color removal, CR(%), of dye was obtained using the relation -
(2)
Where A0 and At are the absorbance values at the initial stage and at time t, respectively.
Effect of current density
The current density determines the flocculation as well as bubble production rates thus impacting the removal efficiency of pollutants such as organic dyes [13]. Plots of percent malachite green removal as a function of current density at 30 minutes using Fe and Al as scarifying anodes, are shown in Figure 3.

Figure 3: Plots of percent MG removal as a function of current density, at 30 minutes using Fe or Al as scarifying anodes. [Malachite green initial concentration: 50 mg/L; Electrolyte (NaCl) concentration: 0.2 g/L and initial pH: 4.0].
It was observed that dye removal increased upon raising current density till 76.5A/m2 and then became constant upon further increasing the current density. Maximum color removal at 30 minutes using malachite green initial concentration: 50mg/L; Electrolyte (NaCl) concentration: 0.2g/L and initial pH 4, were 99.73% and 99.65% in case of Al and Fe scarifying anodic electrodes, respectively. The initial increase in dye removal till the optimum level of current density (76.5 A/m2) is obvious since at higher current density dissolution of anode forming cations (Fe+3 or Al3+, in the present case) is enhanced facilitating higher rate of flocculation and hence the color removal rate [20,21]. However, on further raising current density above its optimum value, the advantage of enhanced ion formation or flocculation rate is counterbalanced by lowering of diffusion rate of flocculants due to enhanced inter-particle collision/hindrance. Similar effect of current density variation during C.I acid yellow dye removal, using electro-coagulation technique, was observed by Danshever et al. [22].
Effect of pH
As the nature as well as the magnitude of net charge on metal hydroxide and dye molecules are pH dependent, therefore, the later may have a significant influence on the dye removal efficiency during electro-coagulation [23,24]. There are diverse reports in the literature on the effect of pH on anodic oxidation process, sometimes even contradictory, due to different organic structures of substrate (pollutant) and electrode materials [25,26]. Plots of percent malachite green removal as a function of pH at 30 minutes, using Fe or Al as scarifying anodes are shown in Figure 4.
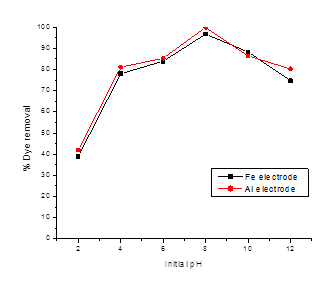
Figure 4: Plots of percent malachite green removal as a function of pH at 30 minutes, using Fe or Al as scarifying anodes.[Current density: 76.5A/m2; MG initial concentration: 100mg/L; Electrolyte (NaCl) concentration 0.2g/L].
It can be observed that there exists an optimum pH 8.0 at which maximum dye removal takes place. Using malachite green initial concentration: 100mg/L, current density: 76.5A/m2 and electrolyte (NaCl) concentration 0.2g/L, as high as 97% and 99.0% color removal could be possible with Fe or Al as anodes, respectively. The lower rate of color removal below optimum pH may be due to the availability of fewer hydroxyl (-OH) ions required for the formation of metal hydroxides flocculant. Further, on raising pH above the optimum value the color removal rate falls owing to diminishing positive charge at tertiary N of Malachite green molecules, minimizing its interaction with the metal ions released by anodic oxidation and hence the lower rate of flocculation [21,22].
Effect of dye initial concentration
Plots of percent removal. MG as a function of time for varying malachite green initial concentration, using current density: 76.53A/m2, electrolyte (NaCl) concentration: 0.2g/L, inter-electrode separation: 1 cm. and Fe and Al as scarifying anodes are presented in Figs. 4A and 4B, respectively. It can be seen that the dye removal efficiency, irrespective of anodic material, decreases upon increasing the dye initial concentration. In both of the anodic material (Fe and Al) used, the dye removal fell from 99.67% to 62.3% on raising the dye initial concentration from 50 to 200mg/L, respectively. The observed diminished rate of color removal with increasing malachite green initial concentration may be due to decrease in effective current density caused by the slow down of current carrier ionic species, hindered by increasing dye molecules, in aqueous solution (Figure 5).
Effect of inter-electrode distance
Plots of percent malachite green removal as a function of inter-electrode distance using Fe and Al as scarifying anodes and fixed current density as well as electrolyte and malachite green initial concentrations, are presented in Figure 5. The observed fall in color removal rate with increasing inter-electrode separation may be due to (a) decrease in effective current density and (b) diminished interaction rate between metal ions and the dye molecules caused by their enhanced degree of freedom inside larger available electrolyte volume between the two electrodes. Further, the observed less rapid color removal, with increasing inter-electrode distance using Al as anodic electrode, compared to Fe anodic electrode, may be due to a larger tendency of smaller Al3+ ions forming their undesired hydrates thus minimizing the probability of their interaction with hydroxide ions and dye molecules to generate the flocculants (Figure 6).

Figure 6: Plots of percent MG removal as a function of inter-electrode distance using Fe and Al as scarifying anodes. [Current density:76.5A/m2, Electrolyte (NaCl) concentration: 0.2g/L and malachite green initial concentration:. 100mg/L]
Effect of electrolyte concentration
Plots of percent dye removal as a function of electrolyte (NaCl) concentration using Fe and Al as anodic electrodes with other parameters invariable, are presented in Figure 7. The initial increase in color removal upon raising electrolyte concentration may be due to enhance current density. At low electrolyte load (0.1g/L), malachite green initial concentration: 100mg/L, current density: 76.53A/m2, inter-electrode separation; 1cm and pH; 8.0, dye removals using Fe and Al as electrodes were: 91% and 96%, respectively. However, such difference was leveled off exhibiting identical maximum color removal (99.0%) due to predominate similar current density effect at high electrolytic concentration, irrespective of the anodic material.
Electro-coagulation (EC) technique, using Al and Fe as scarifying electrodes, have proved an efficient alternative for a complete removal of color from water, contaminated with malachite green dye. For enhanced economy and maximum color removal from malachite green aqueous solution, optimum current density: 76.5A/m2; pH: 8.0 and electrolyte concentration: 0.4g/L, are required. The findings of the reported work would be useful for an effective large scale treatment of waste water contaminated with malachite green dye.
The authors would like to express their gratitude to Jigjiga University, Ethiopia for providing material and chemical support as well as financial assistance.
None.

©2017 Getaye, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.