International Journal of
eISSN: 2475-5559


Research Article Volume 2 Issue 7
1Egyptian Company for plastic industries, Egypt
1Egyptian Company for plastic industries, Egypt
2Department of Chemistry, Ain Shams University Cairo, Egypt
2Department of Chemistry, Ain Shams University Cairo, Egypt
3Department of Polymers and Pigments, National Research Centre, Egypt
3Department of Polymers and Pigments, National Research Centre, Egypt
Correspondence: MF Afify, Egyptian Company for plastic Industries, Egypt
Received: July 20, 2017 | Published: September 15, 2017
Citation: Afify MF, Mekewi MA, Ghaffar MAA, et al. Halogen free, acrylate based/nano bentonite composite functionally validated insulator for electric conductors. Int J Petrochem Sci Eng. 2017;2(7):238-245. DOI: 10.15406/ipcse.2017.02.00062
Poly (methyl methacrylate-butyl acrylate-acrylic acid)/nano bentonite 34 and Cloisite 20 A composites were prepared through bulk polymerization in presence of BPO (benzoyl peroxide) as initiator. The polymer/nano bentonite composite sheets were molded at 180˚C prior subjecting to the specific mechanical and electrical characterization. Structural, physical and thermal validations were conducted through GPC, FT-IR, XRD and HRTEM and TGA classified techniques. The results of the electrical resistivity and mechanical properties aptitudes signpost step towards a new generation of halogen free insulators for electrical cables according to IEC 60502 and BS7655 standards.
Keywords: Poly (methyl methacrylate-butyl acrylate-acrylic acid), bentonite 34, cloisite 20 A, halogen free insulators for electrical cables
GPC; gel permeation chromatography; FTIR, fourier transform infra red; XRD, X ray diffraction; HRTEM, High Resolution Transmission Electron Microscopy; TGA, thermo gravimetric analysis; IEC 60520; international electronic commission number 60520; BS 7655; british standard number 7655, PMMA, poly methyl methacrylate; PC, Poly Carbonate; PBA, poly butyl acrylate; PP, poly propylene; PVC, poly vinyl chloride; NBR, nitrile butadiene rubber; BA, butyl acrylate; MMA, methyl methacrylate; AA, acrylic acid; EPDM; ethylene propylene diene monomer, BPO; benzoyl peroxide; Tg, glass transition temperature; Wt. %, weight percent; ASTMD1248-98, american society for testing and materials number 1248-98
Electric power cables are made of stranded copper or aluminum conductors. As copper metal acquires higher conductivity than aluminum it is more commonly employed for the cables make being more ductile and of relatively high tensile strength. Rather, copper is more expensive and denser than aluminum but still favored.1 Halogen free cables are becoming increasingly necessary to protect against the risk of toxic gas emissions during combustion. This type of cabling is increasingly of relevance in public sector housing and major new developments.2
Poly (methyl methacrylate), PMMA, is one of the most widely used types of acrylate polymers due to its favorable clarity and outstanding outdoor weathering resistance, easy handling, processing, and low cost. These impressive properties made PMMA preferred economically to polycarbonates (PC) when a high strength quality is not critical. However, PMMA usually behaves in a brittle manner when loaded, resulting in its restricted application. To overcome functional risking aspects, copolymerization of MMA with other specified monomers that aid glass transition temperature (Tg) alleviation to below room temperature, e.g., butyl acrylate polymers (PBA) are exploited. Generally, the PBA is considered as a colorless transparent rubbery polymer at ambient temperature, thus, it is commonly used in copolymer systems to lighten, also, the brittleness of the final product.3 Polymer/silicate nanocomposites are proposed as a novel class of composites due to their higher mechanical advantages, proved gas barrier, flame retardation etc.4 Among such validated silicates is the bentonite which was exercised with the PMMA matrix5,6 acrylate polymers,7,8 NBR/PVC,9 PP/EPDM10 and PVC.11 The efficiency of bentonite to modify the properties of the polymer depends primarily on the degree of its dispersion in the polymeric matrix and its compatibility.12 The objective of this work is to improve the physical, electrical, thermal and mechanical properties of PMMA copolymers/nano bentonite composites to design a valid, compatible and opportune halogen free electrical insulation that satisfies measurable specified properties of cable insulators compared to currently used formulations.
Materials
Acrylic acid was used without further purification was supplied by Fine-Chemicals LTD Company, butyl acrylate monomer was provided by Sigma Aldrich, and methyl methacrylate by Alfa Aesar. The methyl methacrylate and butyl acrylate monomers were activated prior use by shaking several times with 5% NaOH solution, to remove the stabilizer and then washed by distilled water several times until neutrality. The freshly inhibitor-free monomer was then distilled under vacuum and finally kept under Na2SO4 anhydrous. The refractive index of the inhibitor-free monomers was measured to affirm its purity prior use. Benzoyl peroxide as initiator was provided by Merck-schuchardt.
Cloisite 20 A (organically modified layered magnesium aluminum silicate platelets in nano scale) was provided by BYK for additives and instruments, Germany and Bentonite 34 (organically modified clay) was provided by ELEMENTIS specialties, USA were used at different ratio as fillers.
The polymerization process
Different ratio of inhibitor-free methyl methacrylate, butyl acrylate, acrylic acid monomers were mixed in dry glass ampoules.BPO, as initiator, was then added at 0.6% by weight in addition to different wt. percent of Cloisite 20 or bentonite 34, as filling material, and as given in Table 1. The glass ampoules were sealed under flow of purified and dry nitrogen gas, then placed in water bath at 60-70°C for 4 hours, then left to cool to ambient temperature and finally broken to release its contents and molded at 180°C.
Monomer % (By weight) |
Filler % (By Weight) |
||
|---|---|---|---|
Butyl acrylate |
Methyl methacrylate |
Acrylic acid |
|
45% |
50% |
5% |
Zero |
45% |
50% |
5% |
1% Cloisite 20 A |
45% |
50% |
5% |
3% Cloisite 20 A |
45% |
50% |
5% |
5% Cloisite 20 A |
45% |
50% |
5% |
7% Cloisite 20 A |
45% |
50% |
5% |
10% Cloisite 20 A |
45% |
50% |
5% |
1% bentonite 34 |
45% |
50% |
5% |
3% bentonite 34 |
45% |
50% |
5% |
5% bentonite 34 |
45% |
50% |
5% |
7% bentonite 34 |
45% |
50% |
5% |
10% bentonite 34 |
Table 1 Formulation of copolymer samples
Molecular weight determination
The molecular weights of polymer blend samples using 45% butyl acrylate, 50% methyl methacrylate and 5% acrylic acid monomers in absence and presence of bentonite 34 or Cloisite 20 A, as fillers, were determined using Gel Permeation Chromatography using tetrahydrofuran as the mobile phase Table 2, shows that, the weight average molecular weight (Mw) and number average molecular weight (Mn) in absence of the filling material are 3.5206×105 and 6.0933×104 respectively with a degree of Polydispersity (PD) @5.78 which confirms an obvious disorder due to a free radical mechanism influence. In the presence of bentonite 34, as a filling material, both the weight average and number average molecular weights fluctuate up and down accompanied by a lower value of dispersity index depending on the percentage of bentonite 34 used, Figures 1a, 1b & 1c, affirming an intervening role played by the filler to an interfering ionic polymerization mechanism that aids a more stereo-regular polymeric chains compacts.
Bentonite 34 filler ratio |
|
|
PD |
||
- |
3.5206× 105 |
6.0933 × 104 |
5.7778 |
||
1% |
2.3759× 105 |
6.9852 × 104 |
3.4014 |
||
3% |
1.9584× 105 |
3.8487 × 104 |
5.0884 |
||
5% |
4.8036 × 105 |
1.4202 × 105 |
3.3823 |
||
7% |
2.9605× 105 |
7.8717 × 104 |
3.7609 |
||
10% |
3.4329× 105 |
7.7850 × 104 |
4.4096 |
Table 2 Effect of bentonite 34, on the weight average molecular weight, the number average molecular weight and the Polydispersity of BA/MMA/AA polymer blends
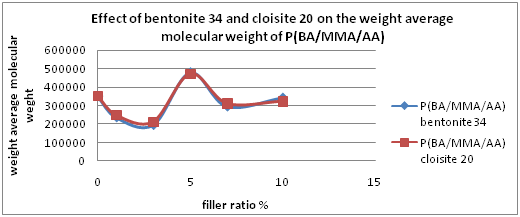


The same trend was observed when using Cloisite 20 instead of bentonite 34 with nearly the same molecular weight values, Table 3 and Figures 1a, 1b & 1c. The highest value of weight average and number average molecular weights were obtained when using 5% of bentonite 34 (4.8036×105 and 1.4202×105) or Cloisite 20 (4.7335×105 and 1.5281×105) with a much lower in the degree of dispersity (@ 3.4 and 3.1) respectively.
Cloisite 20 filler ratio |
|
|
PD |
||
- |
3.5206× 105 |
6.0933 × 104 |
5.7778 |
||
1% |
2.4799× 105 |
6.8832 × 104 |
3.6028 |
||
3% |
2.1120× 105 |
3.9567 × 104 |
5.3357 |
||
5% |
4.7335× 105 |
1.5281 × 105 |
3.0976 |
||
7% |
3.1155× 105 |
7.9927 × 104 |
3.8979 |
||
10% |
3.2338× 105 |
7.8860 × 104 |
4.1006 |
Table 3 Effect of Cloisite 20 on the weight average molecular weight, the number average molecular weight, and the Polydispersity of BA/MMA/AA polymer blends
This is indicating that both Bentonite 34 and Cloisite 20 A acquire the same role through the polymerization process by acceleration of propagation step through a probable cationic mechanism that aids improving molecular weight distribution (lower PD values).Two parallel mechanisms were suggested by D Njopwouo et al.13 For polymerization reaction in the presence of bentonite and acrylic acid who could explain the PD improvement values.
Chemical structural justification (FT-IR spectroscopy)
The accomplishment of the polymerization process in absence and presence of Bentonite 34 or Cloisite 20, as a filling material, was declared through infrared spectroscopy. The FT-IR spectra of Bentonite 34 and Cloisite 20 are shown in Figures 2a, Figure 2b. The absorption bands prevailed at 3630 cm−1 and 916 cm−1 are attributed to Al-OH stretching and bending vibration. The band at 625 cm−1 ascribing the Si-O-Al bond, two absorption bands at 463cm−1 and 522 cm−1 are assigned to low frequency Si-O bending vibration. For the quaternary ammonium salt, two characteristic absorption bands at 2850 and 2920 cm-1 are assigned to the C-H stretching vibration of -CH3 and -CH2 groups respectively and the C-N stretching vibration band at 1045 cm-1 is also assigned. This is in addition to the absorption corresponding to ;band. FT-IR spectra of butyl acrylate/ methyl methacrylate/acrylic acid polymer blend is exhibited in Figure 2c.
The copolymer blend characteristic absorption bands are shown at 1730 cm-1, which is attributed to -C=O stretching vibration, while bands at 2875, 2960 cm-1 can be assigned to the -C-H stretching vibration of -CH3 and -CH2 groups.
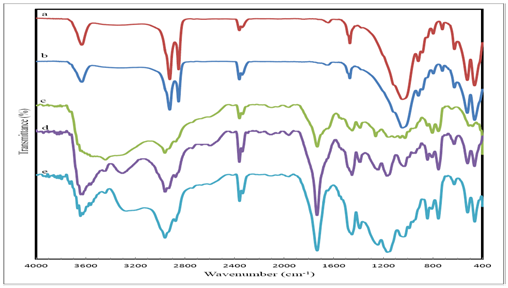
The –C-H in-plane bending vibration of -CH3 and -CH2 groups appear at 1392 and 1450 cm-1. The-C-O-C single bond stretching vibration and single bond deformation vibration bands appear at 1260 cm-1and 844 cm-1 respectively which is the characteristic bands for poly (methyl methacrylate) and poly (butyl acrylate). The indicative band of the -OH stretching vibration (broad) as characteristic for the polyacrylic acid is developed at 3400 cm-1. The FT-IR spectra of the same polymer blends in presence of bentonite 34 or Cloisite 20, are represented in Figures 2d, Figures 2e. The spectra profile proves the disappearance of the-C-N stretching vibration band at 1045 cm-1, while the disappearance of broad band of -OH stretching vibration is clearly observed. The instinct absorption of-C=O stretching vibration at 1730 cm-1, the -C-H in-plane bending vibration of -CH3 and -CH2 groups appear distinctively at 1387, 1450 cm-1 which affirm polymer blend prevalence.
Usually, probable polymers stereo-regularity elevation due to the involvement of an ionic mechanism could be argued from the FT-IR spectra but, unfortunately, could not be affirmed due to polymers elapsed absorption bands.
Microstructure relevance (HRTEM imaging)
The HRTEM microstructure of nano bentonites are exhibited in Figures 3a, Figures 3b where the regular sheet structure is clearly demonstrated.
Upon copolymerization and in presence of the bentonite, the polymeric matrix offered, at set weight ratios, tendency to allocate the bentonite nano particles in a simulated intercalated layer rather than an exfoliated pattern as clearly indicated by the pattern shown in Figures 3c. The intercalated structure deployed seems to have been encouraged by the presence of the basicity of the filler material that enhanced a selective polymerization mechanism. Such intercalation could be principally assigned to the improvements observed to both the mechanical and electrical properties of the polymer composite in addition to its thermal and anti-degradation aspects.
The black lines are described as the cross section of aluminosilicate platelets, the platelets seen as single and uniformly dispersed, which aids activating the polymerization process within the bentonite laminar forming a probable inorganic-organic hybrid.
XRD studies
The XRD patterns of Cloisite 20 A and Bentonite 34 are shown in Figures 4a, Figures 4b. The crystalline peak corresponded to 001 plane of Cloisite 20 A is at2θ = 6.99° and 2θ = 7.11° for Bentonite 34, indicating the basal spacing of 12.6 A and 12.4 A respectively.
The XRD patterns of Poly (BA/MMA/AA)/Bentonite 34 composite at 5 wt. %, however, is shown in Figures 4c. The crystalline peak of 001 plane was observed with disappearance of other small d001 peaks, implying that the intercalation of the P (BA/MMA/AA) into Bentonite 34 layers.
Copolymers/nanocomposites thermal studies
The thermal stability of the polymer blend in absence and presence of filling materials are done using TGA analysis Table 4, represent thermal parameters disclosure of the residual weight, degradation temperature at 10% weight loss (Td10)and degradation temperature at 50% weight loss (Td50)for the prepared copolymers and their composites with bentonite 34 and Cloisite 20. Figures 5a, 5b & 5c represent clearly the thermo grams of the prepared copolymers and those nano bentonites added that reveal the following;
Type |
Residual weight % Above 400˚C |
(Td10)˚C |
(Td50)˚C |
P(BA/MMA/AA) |
0.59 |
300 |
390 |
P(BA/MMA/AA)/Bentonite 34 composite |
3.81 |
310 |
390 |
P(BA/MMA/AA)/Cloisite 20 composite |
4.47 |
325 |
390 |
Table 4 Residual weight and thermal degradation parameters of pure copolymers and bentonite composites
Mechanical properties validation
The mechanical properties of polymeric materials are most important parameters that determine the capability of material mold in suitable shape which induce in turn suitable specific and related features. The tensile strength and elongation at break for all polymer blend samples and composites have been measured and are given in Table 5 and Figures 6A, Figure 6B.
Filler ratio |
Bentonite 34 |
Cloisite 20 A |
||
|---|---|---|---|---|
Tensile strength |
Elongation at break (%) |
Tensile strength |
Elongation at break (%) |
|
(Kg/cm2) |
(Kg/cm2) |
|||
- |
140 |
250 |
140 |
250 |
1% |
142 |
264 |
141 |
260 |
3% |
161 |
275 |
162 |
273 |
5% |
177 |
255 |
175 |
260 |
7% |
159 |
238 |
161 |
235 |
10% |
146 |
222 |
145 |
225 |
Table 5 Effect of Bentonite 34 and Cloisite 20 on the mechanical properties of BA/MMA/AA polymer blend
As detected, in absence of bentonite fillers the tensile strength of the copolymer BA/MMA/AA is 140 kg/cm2 with measured elongation at break of 250 %. This is indicating that this composition is suitable to produce a polymeric material that can be suitable for cable insulators or sheathing, according to IEC 60502 and BS7655 section 6.1. Upon loading 1-5 wt. % of bentonites filling material a considerable improvement in the mechanical properties was noticed which may be attributed to homogeneous dispersion of the filler within the polymeric matrix due to a probable conjugation and electrostatic attractions between polymer chains and the silicate layers. While at high filler concentration, poor dispersion and silicates particles agglomeration could have probably decreased the cohesive forces between polymer chains.
Shore hardness D
Shore hardness D is a measure of the resistance of hard polymer material to penetration of a spring-loaded needle-like indenter. Shore hardness value may vary in the range from 0 to 100. The maximum hardness value 100 corresponds to zero penetration while zero value corresponds to minimum shore hardness (2.5-2.54 mm penetration).
Upon measuring the shore hardness for all the samples, Table 6 and Figure 7, it is evident that the polymer blend offers a shore hardness of 53.5, which is nearly the same value observed when adding bentonite filling material. Such observation indicates that the additions of bentonite 34 or Cloisite 20 silicate particle were not permitted to penetrate within the polymeric chains to influence the alteration of its stiffness. Accordingly, the composite is then still applicable for sheathing materials according to the cable specification HD 620.
Filler ratio |
BA/MMA/AA blend |
|
|---|---|---|
Bentonite 34 |
Cloisite 20 |
|
- |
53.5 |
53.5 |
1% |
54 |
54 |
3% |
54 |
54 |
5% |
54 |
54 |
7% |
54 |
54 |
10% |
54 |
54 |
Table 6 Effect of filling materials on Shore Hardness D after 15 second of the polymer blend
Density measurements
The density of all polymer blend samples has been measured and given in Table 7 and Figure 8. As shown, the density increased gradually as the percentage of the filler material increased. So, according to cable specifications (ASTM D1248-98), these blends can be used as sheathing or insulators for electric cables.
Filler ratio |
BA/MMA/AA blend |
|
|---|---|---|
Bentonite 34 |
Cloisite 20 |
|
- |
1.12 |
1.12 |
1% |
1.125 |
1.126 |
3% |
1.142 |
1.14 |
5% |
1.145 |
1.144 |
7% |
1.151 |
1.15 |
10% |
1.156 |
1.155 |
Table 7 Effect of filling materials on the density (g/cm3) of the polymer blends
Electrical properties
The electrical property in term of volume resistivity and electric capacity is a one of the most important parameter for the polymeric material especially if it is used as an insulator or sheathing for the electrical cables.
The volume resistivity and electric capacity of different samples were measured and are given in Tables 8 & 9 and Figures 9A, Figure 9B. As shown, the volume resistivity and electric capacity of the three polymer blends are acceptable according to cable specifications data, IEC 60502 and ASTM D1248-98. Upon addition of Bentonite 34 or Cloisite 20 to the polymer blends an improvement to both the volume resistivity and the electric capacity properties were observed. This behavior can be attributed to the presence of nanofiller materials may affects the space charge accumulation in polymer matrix.
Filler ratio |
BA/MMA/AA blend |
|
|---|---|---|
Bentonite 34 |
Cloisite 20 |
|
- |
1.2 x 1014 |
1.2 x 1014 |
1% |
1.5 x 1014 |
1.44 x 1014 |
3% |
2.0 x 1014 |
1.9 x 1014 |
5% |
2.3 x 1014 |
2.25 x 1014 |
7% |
3.0 x 1014 |
2.85 x 1014 |
10% |
3.2 x 1014 |
3.13 x 1014 |
Table 8 Effect of filling material on the Volume Resistivity (Ω cm) of the polymer blends
Filler ratio |
BA/MMA/AA blend |
|
|---|---|---|
Bentonite 34 |
Cloisite 20 |
|
- |
100 |
100 |
1% |
134 |
135 |
3% |
144 |
143 |
5% |
147 |
150 |
7% |
162 |
161 |
10% |
123 |
120 |
Table 9 Effect of filling material on the Electrical Capacity (PF) of the polymer blends
Melt Flow Index
Melt flow index is a measure of how many grams of a polymer flow through the die in ten minutes. It is an important parameter that is considered crucial during the fabrication process especially if it is used as insulator or sheathing for the electric cables. The melt flow index was measured for all samples, Table 10 and Figure 10. According to ASTM D 1248-98 categories 3, 4, and 3 respectively, the three blends are considered feasible for producing sheathing or insulating materials even in presence of bentonite filling materials at the studied weight ratios.
Filler ratio |
BA/MMA/AA blend |
|
|---|---|---|
Bentonite 34 |
Cloisite 20 |
|
- |
1.4 |
1.4 |
1% |
1.39 |
1.4 |
3% |
1.38 |
1.37 |
5% |
1.2 |
1.22 |
7% |
1.2 |
1.21 |
10% |
1.17 |
1.19 |
Table 10 Effect of filling material on the Melt Flow Index (g/10 min) of the polymer blends
Sun light effect and aging
All samples were subjected to sun light radiation for different time periods up to 180 days. The physico-mechanical properties of the aged samples were determined and the obtained results are presented in Tables 11 & 12 and Figures 11A, Figures 11B.
Aging time (Days) |
Polymer |
Polymer + bentonite34 1% |
Polymer + bentonite34 3% |
Polymer + bentonite34 5% |
Polymer + bentonite34 7% |
Polymer + bentonite34 10% |
0 |
140 |
142 |
161 |
177 |
159 |
146 |
30 |
140 |
142 |
140 |
170 |
158 |
155 |
60 |
135 |
172 |
143 |
195 |
162 |
160 |
90 |
133 |
169 |
210 |
191 |
205 |
179 |
120 |
130 |
157 |
195 |
205 |
152 |
142 |
150 |
128 |
147 |
188 |
145 |
147 |
139 |
180 |
125 |
133 |
153 |
130 |
139 |
135 |
Table 11 Effect of sun light radiation on tensile strength of the BA/MMA/AA polymer and its bentonite 34 composites
Aging time (days) |
Polymer |
Polymer + bentonite34 (1%) |
Polymer + bentonite34 (3%) |
Polymer + bentonite34 (5%) |
Polymer + bentonite34 (7%) |
Polymer + bentonite34 (10%) |
0 |
250 |
264 |
275 |
260 |
242 |
230 |
30 |
245 |
260 |
250 |
260 |
240 |
227 |
60 |
230 |
230 |
240 |
260 |
234 |
225 |
90 |
210 |
223 |
238 |
258 |
228 |
219 |
120 |
160 |
159 |
210 |
216 |
201 |
207 |
150 |
155 |
150 |
210 |
175 |
190 |
201 |
180 |
150 |
149 |
165 |
162 |
185 |
190 |
Table 12 Amplitude in mill volts of the Lead-1 of electrocardiography in sheep
*Significant (P≤0.05); NSNot significant (P>0.05)
As observed from the above data, stiffening of the polymeric composite is noticed upon exposure to sun light up to 180 days especially at filler concentration of 5 %. Elevation of the mechanical parameters was observed up till a 100 day of exposure then depreciation started to be influenced due to probable polymeric chains degradation and volatilization of smaller molecules that leads to filler material wt. % increase comparatively and therefore affecting the mechanical parameters negatively.
The present work investigated the possibility of using the copolymerization of some acrylic monomers in presence of different ratio of organically treated bentonite and its applicability as a halogen free cables insulator. The following conclusions are derived:
Chemical structural and microstructure features have partially been supported by the central lab of the faculty of science, Ain-Shams University, Cairo, thanks also to the research and development department of the Egyptian company for plastic industries (El- Sewedy Electric), 10th of Ramadan city, Sharkia, Cairo while density, melt flow, mechanical and electrical measurements were all conducted.
The author declares no conflict of interest.

©2017 Afify, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.