International Journal of
eISSN: 2475-5559


Research Article Volume 4 Issue 4
1Department of Chemical and Biological Engineering, University of Saskatchewan, Saskatoon, SK, Canada
2Corning Reactor Technology Center (China), Changzhou, Jiangsu, China
Correspondence: Hui Wang, Department of Chemical and Biological Engineering, University of Saskatchewan, Saskatoon, SK, Canada S7N 5A9, Tel +1-306-966-2685, Fax +1-306-966-4777
Received: July 19, 2019 | Published: July 31, 2019
Citation: Moniri A, Wang H, Wu X. Application of corrosion-resistant corning advanced-flow reactors for multiphase Bunsen reaction-part one: investigation on SO2 absorption. Int J Petrochem Sci Eng. 2019;4(4):123-136. DOI: 10.15406/ipcse.2019.04.00112
Bunsen reaction () is a center reaction 2H2O + I2+ SO2→ H2SO4+ 2HI of two important chemical cycles for hydrogen production: the H2S splitting cycle and the sulfur-iodine (S-I) water-splitting cycles. Introducing I2-tolune solution to the reaction can ease challenges such as side reactions and corrosion hazard. The glass-made Corning® Advanced-FlowTM Reactor (AFR) can completely avoid corrosion and also improve the mass transfer among the multiphase, i.e., gas SO2-N2, water, and toluene-I2, to produce HI/H2SO4. In this paper, the absorption behavior of SO2 in water, toluene and water-toluene mixture was investigated in the absence of iodine. The influences of mole fraction of SO2 in gas phase and the flow rates of gas and liquid on SO2 absorption rate and liquid-side volumetric mass transfer coefficients showed different absorption behaviors of SO2 molecules in water and toluene. The differences also appeared between the microscale and milliscale AFR. However, the plot of the liquid-side volumetric mass transfer coefficient vs. residence time indicated straightforward and seamless scalability of the AFR from microscale to milliscale for SO2-water and SO2-toluene absorption. This absorption study was necessary to provide preliminary data for the multiphase Bunsen reaction study in which iodine is present.
Keywords: bunsen reaction, H2S splitting cycle, hydrogen production, corning® glass advanced-flowTM reactor, liquid-side volumetric mass transfer coefficient
To upgrade bitumen from Canadian oil sands into synthetic crude oil, a tremendous amount of hydrogen (H2) is needed mostly for hydrotreating, where nearly the same amount of hydrogen sulfide (H2S) is produced on molar basis. Traditionally, the H2S is converted into elemental sulfur (S) in sulfur recovery units through the Claus process.1 Although the Claus process is very well established, it has drawbacks. For instance, about half of the H2S handling costs are related to the tail gas treatment. More importantly, the conversion of hydrogen into low-value water reduces plant efficiency. An H2S chemical splitting cycle2was developed in our research group in order to directly decompose hydrogen sulfide with simultaneous production of hydrogen, which could be reused in oil sands upgrading as a partial substitute for steam reforming of methane (SRM). Similar to the S-I water-splitting cycle,3 this cycle consists of three chemical reactions:
H2S + H2SO4→ S + SO2+ 2 H2O (120–150oC)
2H2O + I2+ SO2→ H2SO4+ 2HI (25oC) (2)
2HI → H2+ I2(25oC)(3)
The overall reaction is:
H2S → H2+ S (4)
By further oxidizing the elemental sulfur produced from reaction (1) into SO2, the cycle can produce two moles of H2 from one mole of H2S, leading to the production of one mole of sulfuric acid.2 Reaction (2), known as the Bunsen reaction, and reaction (3), known as HI decomposition, are two of the three reactions in the S-I water-splitting cycle. These reactions have been widely studied since the S-I cycle was proposed, in particular, the Bunsen reaction, which links the sulfur and iodine loops in the S-I water-splitting cycle, has received remarkable attention recently through massive investments in research.4–6 Initially, General Atomics (GA) proposed the following stoichiometry:
SO2+ 16H2O + 9I2→ (2HI + 10H2O + 8I2)HIx phase+ (H2SO4+ 4H2O)sulfuric acid phase (5)
Where, a large amount of iodine and water is required to prevent side reactions3 and to separate the two acid products into two immiscible liquid phases: sulfuric acid (upper phase), mainly a mixture of H2SO4 and water, and hydroiodic acid (lower phase), including HI, I2, and water.7 Excessive use of water and iodine, according to the stoichiometry of reaction (5), results in increased capital and operating costs because of the excessive material circulation and reduced energy efficiency of the entire chemical cycle. For instance, high heat duty is required for the concentration of both acids prior to decomposition.6 In addition, the Bunsen reaction must be carried out at elevated temperatures above the melting point of iodine (117°C), leading to iodine vapor deposition in the setup and tube blockage. At the elevated temperatures, the severe corrosive of the Bunsen reaction products is also a major concern.8 Therefore, experimental and theoretical research activities have been carried out during the last decade to minimize Bunsen reaction challenges.9‒12 Our research group recently proposed a low-temperature process for the H2S splitting cycle where toluene is used to simply dissolve iodine and this dissolved iodine can be transported from I2-toluene solution to aqueous phase such that the Bunsen reaction can be carried out continuously at room temperature.13
This low-temperature process avoids the side reactions and iodine vapor deposition, and prevents corrosion. Subsequent electrolysis of the Bunsen reaction products relieves concerns about product acids separations as well. Despite all the benefits, the use of an I2-toluene solution leads to a multiphase reaction system involving gas, aqueous, and organic solutions, denoted as a gas-liquid-liquid system. In the multiphase reaction system, mass transfer across phases may become rate-limiting. The overall rate is proportional to the liquid-side volumetric or overall mass transfer coefficient (KLa). Usually, generation of high shear in the liquid near the interface and large interfacial areas are desirable in order to achieve high mass transfer rates.14 Therefore, the apparent reaction rate of the Bunsen reaction was studied in the gas-liquid-liquid multiphase system using a batch reactor.15 Comparisons of the rate of SO2 absorption in the liquids of different compositions (toluene, toluene and water, I2-toluene solution, water), indicated that SO2 dissolution in liquid phase was the rate-controlling step provided the gas phase resistance was negligible. This was confirmed by the small value of the activation energy (6.02kJ mol-1) obtained by fitting the reaction rate at various temperatures using the Arrhenius equation. It was concluded that improvement of cross-phase mass transfer is critical to increase process efficiency.
The emerging new reactor technology, Corning® Advanced-FlowTM Reactor, was claimed to be capable of improving the gas-liquid mass transfer coefficients compared to other gas-liquid contactors such as bubble columns and spray columns.16 The AFR are made of glass or ceramic materials that are highly corrosion-resistant for aqueous acid solutions. The modular design of these AFR allows scale-up from the lowest flow rate (10mL min-1) of the low-flow (LF) cells to highest flow rate (5000mL min-1) of the G4 cells. In addition, AFR can improve heat transfer effectively.17‒19 To investigate the gas-liquid-liquid multiphase system of the Bunsen reaction where I2 was provided for the Bunsen reaction through I2-toluene solution, two types of AFR, AFR-LF (maximum through-put: 5m3/year) and AFR-G1 (maximum through-put: 80m3/year), were chosen. The work is reported under one general title, divided into part one and part two. Part one, the present paper, deals with the absorption of SO2 in water, toluene and water-toluene mixture to study the behaviors of multiphase system in the absence of iodine (Bunsen reaction). By applying two-film theory, the liquid-side volumetric mass transfer coefficients were calculated for the binary systems. The results for the two scales of AFR (LF and G1) were also compared. With iodine added to SO2-water-toluene mixture, part two studied the effects of operating conditions such as gas and liquid flow rates, water-to-toluene ratio, and temperature on SO2 absorption and of I2 consumption in continuous mode in both LF and G1.
The experiment with the AFR-LF was conducted and analyzed at the University of Saskatchewan (UofS). The experiment using the AFR-G1, a larger commercially available module, was performed at the Corning Reactor Technology Center (CRTC) in Changzhou, China, while the analyses were performed in the Department of Biochemical Engineering, Changzhou University, Changzhou, China.
The flow pathway of the AFR consists of identical small heart-shaped cells in blue color as shown in Figure 1. Each module has variable cross sections, which cause the fluid to form a jet and repeat splitting and mixing actions along the pathway (divergent-convergent configuration).16 This mechanism helps to increase the contact area among the phases. Commercial AFR are designed in different sizes (LF, G1, G2, G3, and G4) depending on the state of the research and/or the scale of production throughput. The interconnected fluidic module design allows the use of several modules in cascades for multiple-step reactions. Each glass module is sandwiched between two fluid-heating plates, thus enabling temperature control.20 Figure 1 also shows the experimental set up with AFR-LF modules and their connections used in this study.
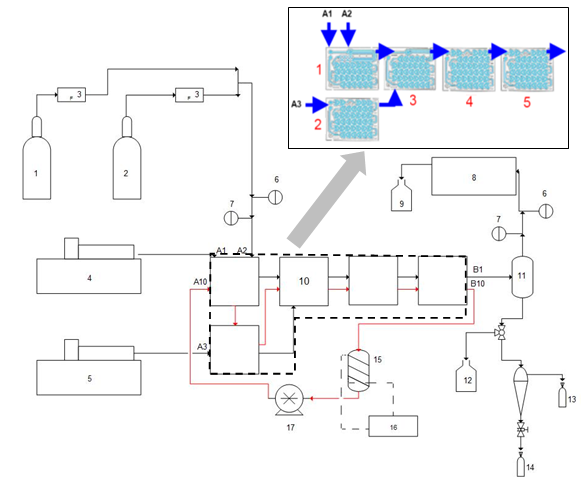
Figure 1 Schematic diagram of experimental setup for the Bunsen reaction using AFR-LF: (1) N2 tank; (2) SO2 tank; (3) Mass flow controller; (4) Organic syringe pump; (5) Water syringe pump; (6) Pressure indicator; (7) Temperature indicator; (8) Gas Chromatograph; (9) Scrubber; (10) Corning AFR-LF; (11) Gas-liquid separator; (12) Waste tank; (13) Organic phase sampling vial (14) Water phase sampling vial; (15) water (oil) bath (16) Temperature controller; (17) Hot fluid pump.
The internal volume of each fluidic module of the AFR-LF is about 0.4mL, which allows for liquid flow rates in the range of 0–10mL min-1. Fluidic modules may be used in parallel or series, depending on the research requirements. At the beginning, five modules were used in series: Module 1 had two fluid inlet ports (A1 and A2), allowing two fluids to mix and/or react. Module 2 had one inlet port (A3), allowing a third fluid to be introduced and conditioned to the same temperature. For experiments at room temperature, this module seemed useless. Modules 4 and 5 (when necessary) were used to extend the residence time. In later runs, one module was used for SO2 absorption to reduce residence time. For a typical run, a gas mixture of SO2 and N2, supplied from cylinders and controlled by two mass flow controllers (1179A Mass-Flo®, MKS), was fed to port A2. Water and toluene were also pumped into the AFR-LF via ports A1 and A3 or A3 and A1, using 100-Legato KD scientific syringe pumps (Figure 1).
The experiments with AFR-LF were carried out at room temperature (22–25°C), at different gas flow rates ranging from 53.6 to 85.7mL min-1 at standard temperature and pressure (STP) and at different liquid flow rates (water from 0.3 to 0.7mL min-1 and toluene from 0.65 to 1.3mL min-1). The outlet of the AFR-LF was open to the atmosphere. A manual control valve was designed to keep the level of the liquid constant at the lower-middle of the separator so that the outlet gas can be easily purged only from the top of the separator to an online GC for analysis. At the bottom of the separator, liquid was collected in a separatory funnel where the organic phase was sampled using a glass Pasteur pipet and the water phase was sampled from the bottom. The experiment setup with AFR-G1 was similar to what Figure 1 shows, except that the gas samples were collected by using Cole-Parmer Tedlar bags at specific time intervals and analyzed by using an off-line instrument. The internal volume of each fluidic module of the AFR-G1 is about 20times that of the AFR-LF, i.e., 8mL, with a maximum allowable liquid flow rate of 200mL min-1. The rest of the conditions, such as material, geometry, and design conditions, remained unchanged.
For this study two fluidic modules (modules 1 and 3) were usually used. Module 1 was used for mixing of two feeds, usually SO2-N2 and the organic phase, and module 3 was used for introducing water and mixing. As in the runs by using the AFR-LF, the gas mixture of SO2 and N2, from cylinders controlled by two mass flow controllers (5850S Smart Mass low, Brooks Instruments Inc.). Toluene and water were also pumped into the AFR-G1 by using a PTFE pump (Fuji Technic Tokyo) and a stainless steel pump (Jiangsu Hanbon Science & Technology Co.), respectively. The experiments were carried out at room temperature (22–25°C), at different gas flow rates ranging from 1600 to 2000mL min-1 at standard temperature and pressure (STP), and at different liquid flow rates (water from 30 to 60mL min-1 and toluene from 37 to 79mL min-1). The outlet of the AFR-G1 was also open to atmospheric pressure. Two 10L bottles of concentrated sodium hydroxide (5mol L-1) were used in series to capture non-reacted SO2 at the outlet of AFR-G1.
Chemicals used for the experiments with the AFR-LF were mostly ACS grade as received: sulfur dioxide (99.98%, Praxair), nitrogen (99.99%, Praxair), sodium hydroxide (97%, EMD), toluene (99.95%, BDH), and phenolphthalein and thyodene as the indicators. Chemicals were used as received without any modification or further steps of purification for the experiments with the AFR-G1 as well: sulfur dioxide (99.99%, Shanghai Weichuang Gas), nitrogen (99.999%, Changzhou Wujin Huayang Gas), sodium hydroxide (96%, Sinopharm Chemical Reagent), toluene (99.5%, Sinopharm Chemical Reagent), and phenolphthalein and thyodene were used as indicators. All the solutions used in both series of experiments and analyses were prepared by diluting the corresponding chemicals with deionized water.
For the experiments conducted using the AFR-LF, unreacted SO2 was analyzed using a Varian CP-3800 gas chromatograph equipped with a thermal conductivity detector (TCD) after reaching steady state, roughly when the equivalent of three times the internal volume of the AFR-LF (or AFR-G1 in later experiments) had been processed. The internal normalization method was used for the gas calibration because this technique corrects for the sample-size error.21 However, for the experiments conducted using the AFR-G1, the external calibration method was used. Due to unavailability of the online method, gaseous sulfur dioxide samples were collected in Cole-Parmer Tedlar bags and were then analyzed using a Taizhou Zhonghuan Analysis Instrument Co. RPP-2000S Fluorescence Sulfur instrument at Changzhou University. Because the N2 flow rate remains unchanged during absorption or reaction, the SO2 flow rate in gas after absorption can be determined once its concentration in gas stream is known. The SO2 absorption rate with respect to SO2 can then be determined from the difference between the inlet and outlet SO2 flow rates.
The two-film theory of mass transfer was used to determine the mass transfer coefficients of SO2 across the gas/water and gas/toluene interfaces. According to this theory,22 the flux of the mass transfer of gas molecules A (SO2 in this case) into liquid through the interface (z=0) can be written as:
N=P−HC1k−Hk=K(P−P') or =PH−C1kH−1k=K(C'−C) (6)
Where NA is the molar flux of A molecules from gas to liquid in the direction perpendicular to the mass transfer interface (mol∙m-2s-1), PA is the partial pressure of A in gas phase (Pa), HA is Henry’s law constant (Pa m3mol-1), CA is the concentration of A in liquid (mol m-3), kG is the mass transfer coefficient in the gas phase (mol m-2s-1Pa-1) and kL is the mass transfer coefficient in the liquid phase (m s-1); P'A = HACA, and P'A = PA/HA, KG (mol Pa-1m-2s-1) is the liquid-side volumetric mass transfer coefficient based on partial pressure driving force and KL (ms-1) is the liquid-side volumetric mass transfer coefficient based on liquid driving force. As explained later, the ideal plug-flow is assumed for liquid and gas through the fluid channels of both the LF and G1, and the performance equation is developed by combing the rate equation with the material (mole) balance. Taking a slice of the reactor along the material flow direction as the volume element (dV) for mass balance, of which the interface area per volume is a (m2m-3, or m-1),
A lost in gas phase= −FGdyA=A gained in liquid= FLCtdCA= NA, z=0adV (7)
Where, FG and FL are the total molar flow rates of gas and liquid (mol∙s-1), respectively, and Ct is the total concentration of liquid (mol m‑3). Considering yA = PA/Pt (Pt is the total pressure of gas), and combining Eqs (6) and (7), integration on either side gives the reactor volume (Vr).
Vr=FGKGa.PtPA2∫PA1dPA(PA−P'A)=FLKLa.CtCA2∫CA1dCA(C'A−CA) (8)
Upon integration of the liquid-side equation from inlet (CA1) to outlet (CA2), the liquid-side volumetric mass transfer coefficient (KLa) becomes
KLa=1τlnC*A−CA1C*A−CA2=1τlnX (9)
Where,C*A is the equilibrium concentration of SO2 in liquid phases (water or toluene) corresponding to the partial pressure of SO2 in the gas phase at the inlet (PSO2 in), CA1 is the concentration of SO2 in the liquid phase at the inlet, which is always zero, τ is the residence time of the liquid flow in the AFR (VrCt/FL), and KLa is the liquid-side volumetric mass transfer coefficient. C*A1 is calculated based on the partial pressure of SO2 at the AFR inlet using Henry’s law;C*A2 is obtained at the AFR outlets based on the dissolved SO2 (the difference between SO2 inlet and outlet in the gas phase and the liquid flow). Considering the residence time of the liquid, the liquid-side volumetric mass transfer coefficients in the LF and G1 at different operating conditions are estimated by Eq. (9). In fact, Eq. (9) has been used to calculate KLa in microscale and milliscale AFR, not only for gas-liquid adsorption systems but also for liquid-liquid absorption systems.23 The volumetric mass transfer coefficient (KLa) is given because the liquid-side volumetric mass transfer coefficient based on liquid driving force, KL, and the specific interface area, a, cannot be separated from each other and the determination of a is not within the scope of this study.
The specific design of AFR is somewhat different from what is normally observed for straight channels. When the fluid enters the heart-shaped cell it hits the first curved post and splits into two streams until reaching a second cylindrical post where the two streams recombine.16 Therefore, for hydrodynamics study, the neck of the heart cell (H=400µm, W=300µm for the AFR-LF and H=1.12mm, W=1mm for the AFR-G1) is considered as the reference to define dimensionless parameters such as the Reynolds number.
Re=ρuDhμ (10)
Where ρ and µ are the average density and viscosity of the fluids, respectively, u is the total superficial velocity of the two phases, and Dh is the hydraulic diameter. The diameter of the fluid channel at the connection of the two heart-shaped cells in an AFR is used for Dh. The density and viscosity of the fluids used are given in (Table 1).
|
A1 |
A2 |
A3 |
SO2 outlet flow rate (mL min-1) |
SO2 outlet mole fraction |
SO2 absorption rate (mol m-3s-1) |
|
water |
SO2-N2 |
toluene |
16.5 |
0.5239 |
15.02 |
|
toluene |
SO2-N2 |
water |
16.55 |
0.5246 |
15 |
|
toluene |
water |
SO2-N2 |
16.43 |
0.5228 |
15.04 |
Table 1 SO2 absorption rate measured in LF-AFR at different inlet positions of gas and liquids (water flow rate=0.5mL min-1, toluene flow rate=0.5mL min-1, gas flow rate=85mL min-1, SO2 mole fraction=0.824, T=22-25oC).
Thus, for the AFR-LF, the gas and liquid flow rates range from 50 to 80 and 0.3 to 0.7mL min-1, respectively, and the range of the two-phase Re is from 2360 to 3790, which is just above the critical value implying the transition to turbulent flow. For the AFR-G1, Re ranges from 12820 to 16320 for SO2 and water flow rates within 800-1000 and 30-60mLmin-1, respectively, indicating fully developed turbulent flow. The visualization experiments, which were done for a CO2-water system by Nieves-Remacha et al.,16 using a AFR-G1, observed that two-phase flow in the AFR included a sequence of dynamic events: detachment, elongation, deformation, breakup, and coalescence of bubbles as they passed through the continuous phase. Confined geometry, small dimensions, and the presence of obstacles that disturb were mainly responsible for these events. Although these dynamic events were quite different from what had been observed in either stirred vessels or straight micro channels, tracer experiments for families of the AFR-LF and AFR-G1 showed the residence time distribution (RTD) to be narrow.24 This indicates that each heart-shaped cell can be considered as an ideal stirred vessel in which complete mixing occurs. However, there must be no back mixing among cells and therefore, the flow throughout the whole AFR can be treated as plug flow.
The AFR-LF was chosen to test the effect of mixing order of the gas mixture of N2 and SO2, water, and toluene, using the port combinations of A1, A2, and A3, which allow mixing of any two fluids in the first module and mixing of the third in the second module. Along with the six possible combinations of mixing as well as the other experimental variables, the absorption rate results are given in Tables 2 and 3. Absorption runs at a gas flow rate of 85mL min-1 at STP, SO2 mole fractions (ySO2=0.824), and two levels of water and toluene flow rates were conducted at room temperature. The results indicate that the SO2 absorption rate by the liquid was independent of the feeding positions of the fluids. However, the slightly higher absorption rate in the latter Table was due to the higher toluene-to-water ratio. More toluene, which has the higher SO2 solubility, would lead to a higher absorption rate. Although the same absorption rate can be achieved by either feeding sequence in the SO2-water-toluene system, to maintain consistency in the experiments, the first and second feeding scenarios, i.e., mixing SO2 with either liquid in the first module, were used for most of the runs in the subsequent experiments using both AFR-LF and AFR-G1 (Tables 2 and 3).
Absorption in AFR-LF
Absorption runs with a constant gas flow rate (75mL min-1 at STP) at various SO2 mole fractions were conducted at room temperature and three water flow rates in the AFR-LF. (Table 4) details the experimental variables and calculated parameters from which the values of the overall mass transfer coefficients are obtained. Figure 2 shows the effects of SO2 mole fraction and water flow rate on the SO2 absorption rate and the liquid-side volumetric mass transfer coefficient, respectively.
|
A1 |
A2 |
A3 |
SO2 outlet flow rate (mL min-1) |
SO2 outlet mole fraction |
SO2 absorption rate (mol m-3s-1) |
|
SO2-N2 |
toluene |
water |
13.56 |
0.4749 |
15.84 |
|
SO2-N2 |
water |
toluene |
13.35 |
0.471 |
15.9 |
|
toluene |
water |
SO2-N2 |
13.39 |
0.4717 |
15.89 |
Table 2 SO2 absorption rate measured in LF-AFR at different inlet positions of gas and liquids (water flow rate = 0.1mL min-1, toluene flow rate=0.9mL min-1, gas flow rate=85mL min-1, SO2 mole fraction=0.824, T=22-25oC).
|
QL(mL min-1) |
yA,in |
Pt in (kPa) |
C*A1 (mol L-1) |
Pt out (kPa) |
C*A2 (mol L-1) |
CA2 (mol L-1) |
1/τ (s-1) |
ln(X)# |
KLa×100 (s-1) |
|
0.3 |
0.636 |
231 |
2.02 |
148 |
1.21 |
1.1 |
0.00189 |
0.786 |
0.148±0.01 |
|
0.737 |
231 |
2.33 |
148 |
1.42 |
1.32 |
0.00189 |
0.836 |
0.158±0.01 |
|
|
0.824 |
258 |
2.91 |
148 |
1.6 |
1.73 |
0.00189 |
0.903 |
0.170±0.02 |
|
|
0.933 |
231 |
2.96 |
145 |
1.82 |
1.86 |
0.00189 |
0.99 |
0.187±0.01 |
|
|
0.5 |
0.636 |
251 |
2.19 |
145 |
1.12 |
1.11 |
0.00314 |
0.707 |
0.22±0.01 |
|
0.737 |
245 |
2.47 |
144 |
1.33 |
1.29 |
0.00314 |
0.736 |
0.231±0.01 |
|
|
0.824 |
238 |
2.68 |
141 |
1.48 |
1.58 |
0.00314 |
0.891 |
0.280±0.01 |
|
|
0.933 |
238 |
3.04 |
141 |
1.75 |
2 |
0.00314 |
1.07 |
0.337±0.01 |
|
|
0.7 |
0.636 |
258 |
2.24 |
144 |
1.06 |
1.04 |
0.0044 |
0.624 |
0.275±0.01 |
|
0.737 |
258 |
2.6 |
141 |
1.24 |
1.27 |
0.0044 |
0.669 |
0.295±0.01 |
|
|
0.824 |
231 |
2.61 |
137 |
1.4 |
1.46 |
0.0044 |
0.822 |
0.362±0.01 |
|
|
0.933 |
231 |
2.95 |
134 |
1.63 |
1.88 |
0.0044 |
1.01 |
0.446±0.01 |
Table 3 Details of overall mass transfer calculations for SO2-water in LF-AFR (SO2 mole fraction=0.933, T=22-25oC, A1: water and A2: SO2-N2)
#Second term of Eq. (9)
A

B
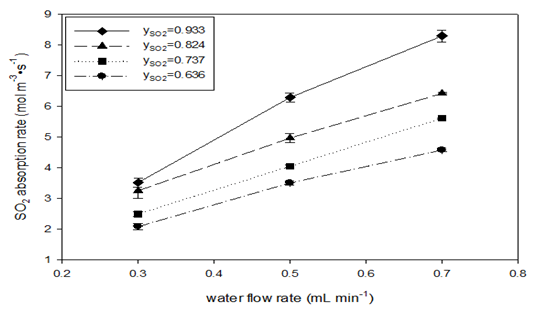
Figure 2 SO2 absorption rate (A) and liquid-side volumetric mass transfer coefficient (B) vs. water flow rate in AFR-LF (QG = 75mL/min, T = 22-25oC, ports: A1, water and A2, SO2-N2).
At a constant mole fraction of sulfur dioxide, a sharp increase could be seen in sulfur dioxide absorption rate when the flow rate of water was increased. Increase in the mole fraction of SO2 at a constant water flow rate also increased the SO2 absorption rate. The higher the water flow rate, the stronger the increasing effect of the mole fraction of SO2 on its absorption rate. The same trend was observed as well when plotting the volumetric mass transfer coefficients vs. water flow rate for four levels of SO2 mole fraction. At lower water flow rate, the effect of mole fraction is insignificant because the resistance to mass transfer is on the liquid side where the KLa value is low. At higher water flow rate, the resistance is shifted to the gas side to some extent and the gradient of SO2 in gas phase begins to play a role. Nieves-Remacha et al. studied the effects of operating conditions on the hydrodynamics of a water-CO2 system using a AFR-G1.16 They observed that the average bubble size and bubble size distribution depend on the gas and liquid flow rates. At the lower flow rate of water, most of the heart-shaped cells were occupied by large bubbles, which resulted in inadequate contact between the gas and liquid, and poor mass transfer across the interface. On the other hand, the bubble size decreased with increasing liquid flow (QL), which increased shear rates at the inlet of the heart-shaped cells. Greater turbulence in the water flow and increased specific area per volume of water resulted in larger KLa values.
Table 3 also shows that increasing the mole fraction of sulfur dioxide stabilized the equilibrium concentration of sulfur dioxide in the water at inlet C*A1 and C*A2 outlet, respectively, leading to an increase in volumetric mass transfer coefficients. Comparing the values of C*A2 andCA2 , two different behaviors were observed. At low mole fraction values of sulfur dioxide, stayed below the equilibrium values, while at high mole fractions, the latter was higher. This was mainly due to the higher partial pressure of sulfur dioxide at the inlet that made SO2 dissolve quickly in water. However, when the pressure at the outlet dropped significantly, a new equilibrium was hard to reach and the outlet concentration CA2 became oversaturated (Table 4 & Figure 2).
|
QL (mL min-1) |
QG (mL min-1) |
Pt in (kPa) |
C*A1 (mol L-1) |
Pt out (kPa) |
C*A2 (mol L-1) |
CA2 (mol L-1) |
1/τ (s-1) |
ln(X)# |
KLa×100 (s-1) |
|
0.3 |
53.57 |
191 |
2.45 |
125 |
1.56 |
1.62 |
0.00189 |
1.09 |
0.205±0.01 |
|
64.29 |
219 |
2.79 |
142 |
1.79 |
1.67 |
0.00189 |
0.91 |
0.172±0.02 |
|
|
75 |
232 |
2.97 |
152 |
1.92 |
1.65 |
0.00189 |
0.814 |
0.154±0.02 |
|
|
85.72 |
252 |
3.22 |
163 |
2.06 |
1.66 |
0.00189 |
0.725 |
0.137±0.01 |
|
|
0.5 |
53.57 |
205 |
2.62 |
125 |
1.53 |
1.69 |
0.00314 |
1.04 |
0.325±0.02 |
|
64.29 |
219 |
2.8 |
131 |
1.63 |
1.77 |
0.00314 |
1 |
0.316±0.01 |
|
|
75 |
239 |
3.06 |
142 |
1.76 |
1.86 |
0.00314 |
0.938 |
0.295±0.01 |
|
|
85.72 |
259 |
3.31 |
152 |
1.9 |
1.87 |
0.00314 |
0.83 |
0.261±0.01 |
|
|
0.7 |
53.57 |
198 |
2.53 |
118 |
1.41 |
1.6 |
0.0044 |
0.998 |
0.439±0.01 |
|
64.29 |
218 |
2.79 |
124 |
1.51 |
1.65 |
0.0044 |
0.897 |
0.395±0.01 |
|
|
75 |
232 |
2.97 |
131 |
1.61 |
1.77 |
0.0044 |
0.911 |
0.401±0.02 |
|
|
85.72 |
252 |
3.22 |
142 |
1.74 |
1.87 |
0.0044 |
0.867 |
0.382±0.01 |
Table 4 Details of overall mass transfer calculations for SO2-water in LF-AFR (SO2 mole fraction=0.933, T=22-25oC, A1: water and A2: SO2-N2).
#Second term of equation (9)
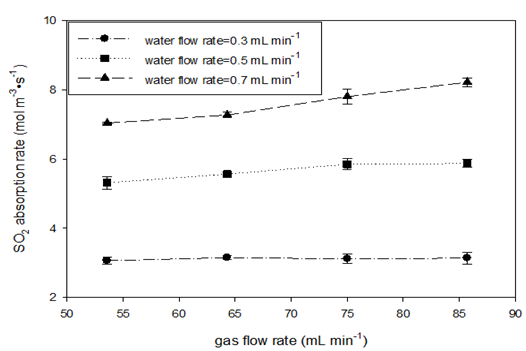
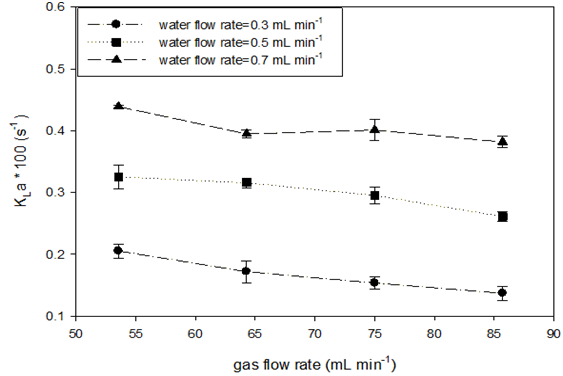
Absorption rates for a high mole fraction of gas (ySO2=0.933) at various gas flow rates (at STP) were also measured at room temperature and three water flow rate levels. A high mole fraction was used to try to minimize the effect on the gas side. Table 5 shows the experimental variables and calculated parameters from which the values of mass transfer coefficients are obtained. (Figure 3) depicts the effects of gas flow rate and water flow rate on the SO2 absorption rate and the volumetric liquid-side mass transfer coefficient. At two lower water flow rates, no significant changes in SO2 absorption rate were observed when the gas flow rate was increased, as shown in Figure 3 (A). At the highest water flow rate, a small increase in absorption rate by gas flow was observed. The absorption capacity is determined by liquid flow once sufficient SO2 is provided. However, the liquid-side volumetric mass transfer coefficients tended to decrease with increasing gas flow rate as shown in Figure 3 (B).
|
QL (mL min-1) |
QG (mL min-1) |
Pt in (kPa) |
C*A1 (mol L-1) |
Pt out (kPa) |
C*A2 (mol L-1) |
CA2 (mol L-1) |
1/τ (s-1) |
ln(X)# |
KLa×100 (s-1) |
|
30 |
1600 |
191 |
1.31 |
102 |
0.45 |
0.63 |
0.0625 |
0.657 |
4.10±0.02 |
|
1800 |
184 |
1.26 |
102 |
0.47 |
0.66 |
0.0625 |
0.745 |
4.66±0.04 |
|
|
2000 |
205 |
1.4 |
102 |
0.46 |
0.75 |
0.0625 |
0.761 |
4.75±0.03 |
|
|
45 |
1600 |
205 |
1.4 |
102 |
0.38 |
0.49 |
0.0937 |
0.43 |
4.03±0.02 |
|
1800 |
205 |
1.4 |
102 |
0.4 |
0.54 |
0.0937 |
0.482 |
4.51±0.01 |
|
|
2000 |
226 |
1.55 |
102 |
0.4 |
0.6 |
0.0937 |
0.487 |
4.56±0.02 |
|
|
50 |
1600 |
219 |
1.5 |
102 |
0.35 |
0.48 |
0.104 |
0.383 |
3.98±0.02 |
|
1800 |
226 |
1.55 |
102 |
0.36 |
0.52 |
0.104 |
0.411 |
4.27±0.01 |
|
|
2000 |
240 |
1.64 |
102 |
0.38 |
0.56 |
0.104 |
0.416 |
4.33±0.01 |
|
|
60 |
1600 |
226 |
1.55 |
102 |
0.33 |
0.41 |
0.125 |
0.307 |
3.83±0.01 |
|
1800 |
240 |
1.64 |
102 |
0.33 |
0.46 |
0.125 |
0.329 |
4.11±0.02 |
|
|
2000 |
260 |
1.78 |
102 |
0.34 |
0.5 |
0.125 |
0.332 |
4.14±0.02 |
Table 5 Details of overall mass transfer calculations for SO2-water in G1-AFR (SO2 mole fraction=0.50, T=22-25oC, A1: water and A2: SO2-N2).
#Second term of equation (9)
A
B
Figure 3 SO2 absorption rate (A) and liquid-side volumetric mass transfer coefficient (B) vs. gas flow rate in AFR-LF (ySO2 = 0.933, T = 22-25oC, ports: A1, water and A2, SO2-N2).
This confirms the argument that increasing the gas flow rate increases the average bubble size, which in turn reduces the specific interface area.16 On the other hand, the absorption capacity was reached, resulting in quite similar outlet SO2 concentrations in water CA2 for each level of water flow rate. Because of the resistance to flow, the increase in gas flow led to total pressure increase and thus the increase in C*A1 . As a result, the logarithmic term of Eq. (9) began to decrease and then the coefficients became smaller. Comparing the values of C*A2 to CA2 , oversaturation was also observed for most of the runs for SO2 absorption in water. The third reason for a decrease in KLa with gas flow rate increase could be the effect on real residence time. At higher gas flow rate, the residence time determined by water flow rate was obviously longer than the real residence time. Then, the calculated KLa became smaller than it should be (Table 5 & Figure 3).
Absorption in AFR-G1
Absorption runs with a constant mole fraction of gas (ySO2=0.50) at various gas flow rates at STP were conducted at room temperature and four water flow rate levels in the AFR-G1. (Table 6) provides the detailed experimental variables and calculated results. Figure 4 shows the effects of SO2 flow rate and water flow rate on the SO2 absorption rate and the volumetric liquid-side mass transfer coefficient, respectively.
|
QL (mL min-1) |
yA in |
Pt in (kPa) |
C*A1 (mol L-1) |
Pt out (kPa) |
C*A2 (mol L-1) |
CA2 (mol L-1) |
1/τ (s-1) |
ln(X)# |
KLa×100 (s-1) |
|
0.65 |
0.636 |
202 |
5.78 |
133 |
2.67 |
1.8 |
0.004 |
0.373 |
0.149±0.01 |
|
0.737 |
202 |
6.69 |
126 |
3.08 |
2.22 |
0.004 |
0.404 |
0.162±0.01 |
|
|
0.824 |
202 |
7.48 |
123 |
3.59 |
2.59 |
0.004 |
0.425 |
0.170±0.01 |
|
|
0.933 |
169 |
7.08 |
116 |
4.22 |
3.41 |
0.004 |
0.658 |
0.263±0.01 |
|
|
0.9 |
0.636 |
217 |
6.2 |
130 |
2.31 |
1.47 |
0.00563 |
0.271 |
0.152±0.01 |
|
0.737 |
210 |
6.96 |
124 |
2.65 |
1.84 |
0.00563 |
0.307 |
0.173±0.01 |
|
|
0.824 |
204 |
7.52 |
120 |
3.14 |
2.16 |
0.00563 |
0.339 |
0.191±0.01 |
|
|
0.933 |
176 |
7.36 |
109 |
3.56 |
2.83 |
0.00563 |
0.484 |
0.273±0.01 |
|
|
1.27 |
0.636 |
238 |
6.78 |
127 |
1.83 |
1.22 |
0.00797 |
0.199 |
0.158±0.01 |
|
0.737 |
231 |
7.62 |
120 |
1.93 |
1.56 |
0.00797 |
0.228 |
0.182±0.01 |
|
|
0.824 |
203 |
7.52 |
116 |
2.55 |
1.73 |
0.00797 |
0.261 |
0.208±0.01 |
|
|
0.933 |
177 |
7.41 |
107 |
2.86 |
2.2 |
0.00797 |
0.353 |
0.281±0.01 |
Table 6 Details of overall mass transfer calculations for SO2-toluene in LF-AFR (gas flow rate=75mL min-1, T=22-25oC, A1: toluene and A2: SO2-N2).
#Second term of equation (9)
A

B
Figure 4 SO2 absorption rate (A) and liquid-side volumetric mass transfer coefficient (B) vs. water flow rate in AFR-G1 (ySO2 = 0.50, T = 22-25oC, ports: A1, water and A2, SO2-N2).
At a constant gas flow rate, a gradual increase in sulfur dioxide absorption could be seen when the water flow rate was increased. At a constant water flow rate, the absorption rate of sulfur dioxide was also increased steadily by increasing the gas flow rate. On the other hand, the volumetric mass transfer coefficient values were relatively stable with the change of the water flow rate at 1600mL min-1 of the gas flow rate. While the data of KLa vs. water flow at 1800 and 2000mL min-1 gas flow rates overlapped at larger values, they tended to decline marginally with the increase in water flow rates. This observation differs from the findings in the AFR-LF as well as the result from the study of the CO2-water system by Nieves-Remacha et al.16 As can be seen from (Table 6), at a constant gas flow rate, the value of CA2 was reduced by increasing the flow rates of water, while was increased, the larger gap between the two concentrations resulted in smaller values of the second term of Eq. (11). On the other hand, the residence time was reduced, which in turn resulted in an increase of 1/τ values. At these critical conditions, the effect of shortening the residence time and the change of SO2 concentrations by gas and water flow rates balanced, leading to relatively stable volumetric mass transfer coefficients. While the shortening of residence time by increasing the water flow tipped the scales, a decrease of the coefficients was observed. At constant values of water flow, the increase of gas flow from 1600 to 1800mL min-1 led to a relatively small increase in KLa; however further increase in gas flow rate resulted in no further change in KLa. This may indicate that the mass transfer in absorption was not controlled by the SO2 transportation on the gas side (Table 6 & Figure 4).
Absorption in AFR-LF
In runs similar to those using water as solvent for SO2 absorption, the solvent was switched toluene and the absorption was first run with a given gas flow rate (75mL/min at STP) at four levels of SO2 mole fraction (0.636–0.933), three toluene flow rate levels (0.65–12.7mL min-1), and at room temperature. (Table 7) details the experimental variables and the calculated results.
|
QL (mL min-1) |
QG (mL min-1) |
Pt in (kPa) |
C*A1 (mol L-1) |
Pt out (kPa) |
C*A2 (mol L-1) |
CA2 (mol L-1) |
1/τ (s-1) |
ln(X)# |
KLa×100 (s-1) |
|
0.65 |
53.57 |
148 |
6.21 |
95 |
3.14 |
2.81 |
0.004 |
0.603 |
0.241±0.01 |
|
64.29 |
162 |
6.79 |
109 |
3.82 |
3.14 |
0.004 |
0.622 |
0.249±0.01 |
|
|
75 |
169 |
7.08 |
116 |
4.22 |
3.41 |
0.004 |
0.658 |
0.263±0.01 |
|
|
85.72 |
176 |
7.31 |
119 |
4.41 |
3.76 |
0.004 |
0.723 |
0.289±0.01 |
|
|
0.9 |
53.57 |
148 |
6.21 |
95 |
2.65 |
2.2 |
0.00563 |
0.438 |
0.247±0.01 |
|
64.29 |
162 |
6.79 |
95 |
2.9 |
2.54 |
0.00563 |
0.469 |
0.264±0.01 |
|
|
75 |
176 |
7.36 |
109 |
3.56 |
2.83 |
0.00563 |
0.484 |
0.273±0.01 |
|
|
85.72 |
183 |
7.65 |
112 |
3.81 |
3.11 |
0.00563 |
0.522 |
0.294±0.01 |
|
|
1.27 |
53.57 |
142 |
5.96 |
96 |
2.02 |
1.65 |
0.00797 |
0.324 |
0.258±0.01 |
|
64.29 |
158 |
6.63 |
96 |
2.33 |
1.94 |
0.00797 |
0.346 |
0.276±0.01 |
|
|
75 |
177 |
7.41 |
107 |
2.86 |
2.2 |
0.00797 |
0.353 |
0.281±0.01 |
|
|
85.72 |
184 |
7.69 |
110 |
3.16 |
2.46 |
0.00797 |
0.385 |
0.307±0.01 |
Table 7 Details of overall mass transfer calculations for SO2-toluene in LF-AFR (SO2 mole fraction=0.933, T=22-25oC, A1: toluene and A2: SO2-N2).
# Second term of equation (9)
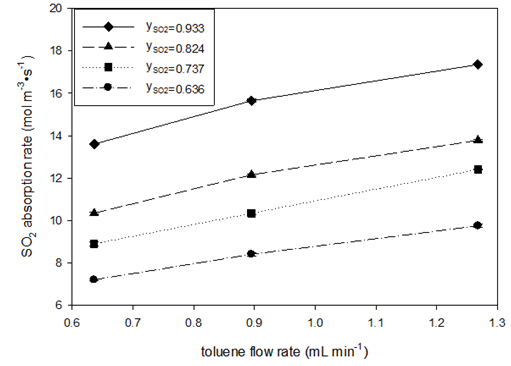
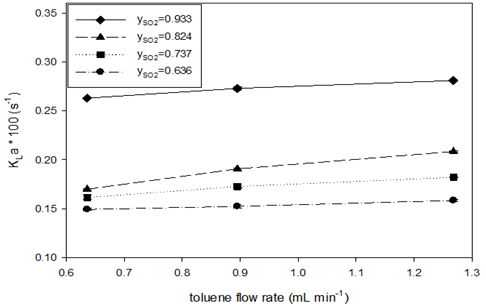
Compared to (Table 4), the flow rate range of toluene was two times that of water. This is roughly determined by the stoichiometry of the Bunsen reaction, the solubility of iodine in toluene and water, and the excessive use of water. (When multiphase absorption and the Bunsen reaction (part two) are carried out with iodine-toluene solution, the flow rate of toluene used can be 2 to 4 times that of water.) This also means that, relative to the same levels of SO2 flow rate, higher flow rate of toluene was used than that of water. Table 7 also shows that the values of the outlet equilibrium concentrations C*A2 are always larger than the outlet concentration CA2 of SO2 in toluene, indicating that absorption equilibrium has not been achieved in toluene absorption. Unlike in water absorption, no oversaturated absorption was observed for toluene. (Figure 5) depicts the effects of SO2 mole fraction and toluene flow rate on the SO2 absorption rate and the volumetric liquid-side mass transfer coefficient.
A
B
Figure 5 SO2 absorption rate (A) and liquid-side volumetric mass transfer coefficient (B) vs. toluene flow rate in AFR-LF (QG = 75mL/min, T = 22-25oC, ports: A1, toluene and A2, SO2-N2).
The absorption rate was reduced by increasing SO2 mole fractions and toluene flow rates, which can be explained by the increase in absorption driving force. The higher values of KLa at higher mole fraction of SO2 may imply resistance to mass transfer on the gas side, and the increasing KLa values at higher toluene flow rate means greater interface areas between gas and liquid for a unit volume of liquid. Comparing the effects of water and toluene flow rate on SO2 absorption, it was found that the increase of absorption rate and the volumetric mass transfer coefficient with water flow rate is sharper than that with toluene flow rate. At a similar liquid flow rate (water at 0.7mL min-1 and toluene at 0.65mL min-1) with the range of SO2 mole fraction in gas phase (0.646–0.933), toluene showed higher absorption rates (7–13.5mol m-3s-1) than water (4.2–8.4mol m-3s-1); however, water had higher values of the volumetric mass transfer coefficient (0.0028–0.0047s-1) than toluene (0.0015–0.0026s-1). The higher absorption rate in toluene can also be attributed to differences in the driving force of mass transfer. The solubility of SO2 in toluene is more than 10 times its solubility in water. With the same partial pressure of SO2 in gas, the mass transfer driving force, , would be 10 times that in toluene initially when CA is zero. The lower KLa for toluene may be because the specific interface area in toluene is smaller than in water and the diffusivity of SO2 is smaller in toluene than in water.
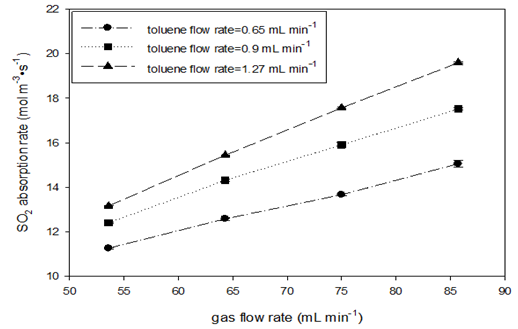
(Table 7 & Figure 5) Absorption rates at various gas flow rates (at STP) with a high mole fraction of SO2 (ySO2=0.933) were also measured at room temperature and three toluene flow rate levels. High SO2 mole fraction was used to minimize gas-side mass transfer resistance. (Table 8) gives the detailed experimental variables and calculated results. Figure 6 shows the effects of gas flow rate on the SO2 absorption rate and the volumetric liquid-side mass transfer coefficient. At a constant toluene flow rate, SO2 absorption rate and mass transfer coefficients were sharply increased with gas flow rates. This behavior is different from what has been observed in SO2-water systems where the absorption rate seemed to be independent of the gas flow rate and the KLa value decreased with increasing gas flow. However, this observation is in agreement with Nieves-Remacha et al.,16 where increase in flow rate of each gas and liquid phase has a positive effect on the volumetric mass transfer coefficient of CO2 into water. It is believed that the increase in KLa with increasing gas flow is due to the increase in specific interfacial area (Table 8 & Figure 6).
|
QL (mL min-1) |
QG (mL min-1) |
Pt in (kPa) |
C*A1 (mol L-2) |
Pt out (kPa) |
C*A2 (mol L-1) |
CA2 (mol L-1) |
1/τ (s-1) |
ln(X)# |
KLa×100 (s-1) |
|
39.63 |
1600 |
171 |
3.83 |
102 |
0.37 |
0.58 |
0.0826 |
0.164 |
1.35±0.01 |
|
1800 |
185 |
4.14 |
102 |
0.38 |
0.64 |
0.0826 |
0.168 |
1.38±0.01 |
|
|
2000 |
192 |
4.3 |
102 |
0.41 |
0.66 |
0.0826 |
0.168 |
1.39±0.01 |
|
|
59.45 |
1600 |
219 |
4.92 |
102 |
0.23 |
0.48 |
0.124 |
0.103 |
1.28± 0.01 |
|
1800 |
233 |
5.23 |
102 |
0.25 |
0.53 |
0.124 |
0.107 |
1.33±0.01 |
|
|
2000 |
247 |
5.54 |
102 |
0.25 |
0.59 |
0.124 |
0.113 |
1.40±0.01 |
|
|
66.06 |
1600 |
219 |
4.92 |
102 |
0.23 |
0.43 |
0.138 |
0.092 |
1.27± 0.01 |
|
1800 |
233 |
5.23 |
102 |
0.24 |
0.48 |
0.138 |
0.096 |
1.32±0.01 |
|
|
2000 |
247 |
5.54 |
102 |
0.25 |
0.53 |
0.138 |
0.1 |
1.38±0.01 |
|
|
79.26 |
1600 |
267 |
6 |
102 |
0.18 |
0.38 |
0.165 |
0.066 |
1.10±0.01 |
|
1800 |
274 |
6.15 |
102 |
0.18 |
0.43 |
0.165 |
0.073 |
1.20±0.01 |
|
|
2000 |
281 |
6.31 |
102 |
0.21 |
0.46 |
0.165 |
0.076 |
1.26±0.01 |
Table 8 Details of overall mass transfer calculations for SO2-toluene in G1-AFR (SO2 mole fraction=0.50, T=22-25oC, A1: toluene and A2: SO2-N2).
# Second term of equation (9)
A

B
Figure 6 SO2 absorption rate (A) and liquid-side volumetric mass transfer coefficient (B) vs. gas flow rate in AFR-LF (ySO2 = 0.933, T = 22-25oC, ports: A1, toluene and A2, SO2-N2).
Absorption in AFR-G1
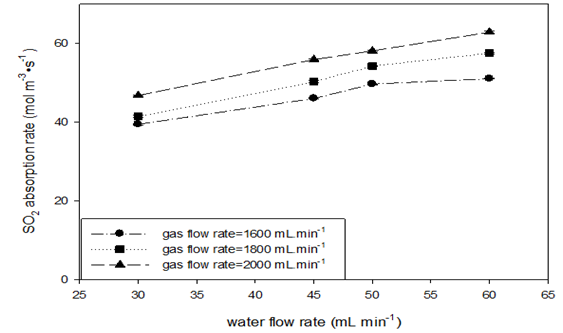
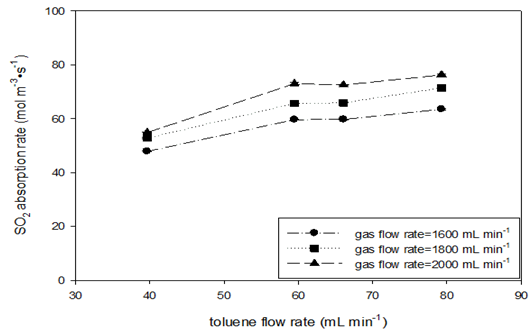
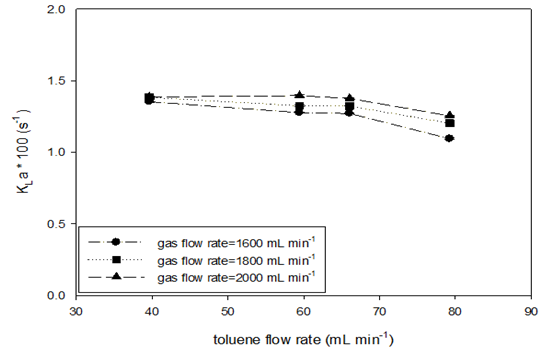
Absorption runs with a constant gas mole fraction (ySO2=0.50) at various SO2 flow rates at STP were conducted at room temperature and four toluene flow rate levels. Table 9 shows the experiment variables and calculated results. Figure 7 shows the effects of increasing gas and toluene flow rates on the SO2 absorption rate and the liquid-side volumetric mass transfer coefficient. Increasing the toluene flow rate led to an increase in the absorption rate values in the beginning (Figure 7 (A)); however, the data became constant at the higher flow rate of toluene. At each level of toluene flow rate, the effect of increasing gas flow on the absorption rate appeared more obvious, similar to what was observed using the AFR-LF. In (Figure 7 (B)), the KLa values seem to be insensitive to both toluene and gas flow rates, which is similar to the observation for the AFR-LF. Increasing the toluene flow rate by a factor of two maintained the KLa values at the same margin (0.011–0.014s-1). At lower toluene flow, the KLa value change by gas flow increasing from 1600 to 2000mL min-1 (40% increase) is negligible (<3 %) Even at higher toluene flow, this change is still small (<15 %). However, the KLa value obtained in the AFR-G1 is 4-6 times that obtained in the AFR-LF (Table 9 & Figure 7).
|
One-fluidic module LF-AFR |
Five-fluidic module LF-AFR |
|||
|---|---|---|---|---|
|
QL (mL min-1) |
SO2 absorption×106 (mol s-1) |
KLa×100 (s-1) |
SO2 absorption×106 (mol s-1) |
KLa×100 (s-1) |
|
0.3 |
7.55 |
1.18 |
8.11 |
0.205 |
|
0.5 |
13.1 |
1.9 |
14.1 |
0.325 |
|
0.7 |
18.5 |
2.55 |
18.7 |
0.439 |
|
1 |
24.5 |
3.19 |
25.1 |
0.545 |
Table 9 Comparison of SO2-water absorption runs in LF-AFR using different fluidic modules.
|
One-fluidic module LF-AFR |
Five-fluidic module LF-AFR |
|||
|---|---|---|---|---|
|
QL (mL min-1) |
SO2 absorption×106 (mol s-1) |
KLa×100 (s-1) |
SO2 absorption×106 (mol s-1) |
KLa×100 (s-1) |
|
0.65 |
29.7 |
1.66 |
29.8 |
0.241 |
|
0.9 |
32.6 |
1.69 |
32.9 |
0.247 |
|
1.27 |
34.8 |
1.7 |
34.9 |
0.258 |
|
1.5 |
35 |
1.71 |
35.1 |
0.266 |
Table 10 Comparison of SO2-toluene absorption runs in LF-AFR using different fluidic modules.
A
B
Figure 7 SO2 absorption rate (A) and liquid-side volumetric mass transfer coefficient (B) vs. toluene flow rate in AFR-G1 (ySO2 = 0.50, T = 22-25oC, ports: A1, toluene and A2, SO2-N2).
Absorption in AFR-LF
Absorption runs with a constant mole fraction of SO2 (ySO2 =0.933) and liquid flow rate (1.5mL min-1) at various gas flow rates at STP were conducted at room temperature (22–25oC) for SO2 in water-toluene mixture where the gas phase resistance was negligible. The purpose of this experiment was to understand the effects of increasing water volumetric fraction in water-toluene and SO2 flow rate in SO2-N2 mixtures in the described system as shown in Figure 8 (A). By definition water volumetric fraction equals water flow rate divided by total liquid flow rate. As can be seen, at a constant water fraction, a sharp increase could be seen in sulfur dioxide absorption rate when the flow rate of SO2 was increased from 53.57 to 85.72mL min-1. The lower the water fraction, the stronger the increasing effect of the SO2 flow rate on its absorption rate. On the other hand, an increase in water fraction at a constant gas flow rate decreased the SO2 absorption rate. This decreasing trend was less tangible at lower water fraction rate when mixture was mainly consisted of toluene. The absorption runs were repeated at room temperature with constant mole fraction of gas (ySO2=0.933) and constant water volumetric fraction at various gas flow rates at STP and five liquid flow rate levels as shown in Figure 8 (B).

A

B
Figure 8 (A) SO2 absorption rate vs. water volumetric fraction in AFR-LF and (B) SO2 absorption rate versus liquid flow rate in the AFR-LF (liquid flow rate=1.5 mL/min for Fig.8 (A), water volumetric fraction=0.25 for Fig.8 (B), mole fraction of SO2=0.933, T=22-25oC, ports A1, toluene, A2, SO2-N2 and A3, Water).
Clearly, at all gas levels, enhancing liquid flow rates increased the absorption rates. However, larger liquid flow rates gave a smoother rise to absorption values mainly due to the fact that complete absorption of SO2 in toluene was more likely achieved at larger liquid flow rates. Similar to (Figure 8 (B)), absorption rates of SO2 were increased at a constant liquid flow rate by increasing gas flow rates, leading to an increase in mass transfer at high flow rates. The observation in section 3.6 confirms the argument in previous sections where the higher absorption rate in toluene was attributed to differences in the driving force of mass transfer due to higher solubility of SO2 in toluene than in water. In the following section, similar experiments were conducted for the ternary SO2-water-toluene system in AFR-G1 at a constant water volumetric fraction.
Absorption in AFR-G1
The absorption runs with a constant mole fraction of gas (ySO2=0.50) and water volumetric fraction (0.15) at various gas flow rates at STP were conducted at room temperature (22–25oC) and four liquid flow rate levels. Figure 9 shows the effects of the liquid flow rate and gas flow rate on the SO2 absorption rate. Increasing the liquid flow rates smoothly increased the absorption rates. An increase in gas flow rates also increased the absorption rates of SO2 at a constant liquid flow rate which is fairly similar to what was observed in AFR-LF (Figure 9).

Figure 9 SO2 absorption rate versus liquid flow rate in the AFR-G1 (mole fraction of SO2=0.50, water volumetric fraction =0.15, T=22-25oC, ports A1, SO2-N2, A2, toluene and A3, Water).
Scalability study of SO2 absorption in LF and G1
The foregoing discussion is not definitive concerning the scalability of the two levels of AFR’s. Specifically, the results showing the effect of water flow rate on KLa from the AFR-LF is not in agreement with that from the AFR-G1. The KLa values of the SO2-water and SO2-toluene absorption for both AFR are not in the same order of magnitude either. Experiments using different numbers of modules of AFR-LF (one vs. five) for two systems (SO2-water and SO2-toluene) resulted in the same magnitude of overall absorption rate and a different order of magnitude for KLa (Tables 10).
The values of KLa from one module are about 6–7 times those from five modules. This indicates that the absorption has been completed in one module and that the remaining modules contribute negligibly to the absorption, which merely extends the residence time and results in smaller KLa from the AFR-LF as in previous sections. Therefore, absorption runs were conducted using one single-module AFR-LF, as described in this section. Then, the new SO2 absorption results of the one single module AFR-LF are compared with the results of the SO2 absorption runs using the AFR-G1, where a single module was used. K.F. Jensen’s group at MIT studied the scalability of these two scales of AFR for liquid-liquid mass transfer.23 The method that was used was to plot the KLa values vs. the residence time for both AFR. By analyzing the magnitude of the KLa and the curves of KLa vs. residence time, the scalability across these two sizes of the AFR is evaluated. (Figure 10) plots the KLa of SO2 absorption in water and toluene using one-module LF and G1 vs. residence time.
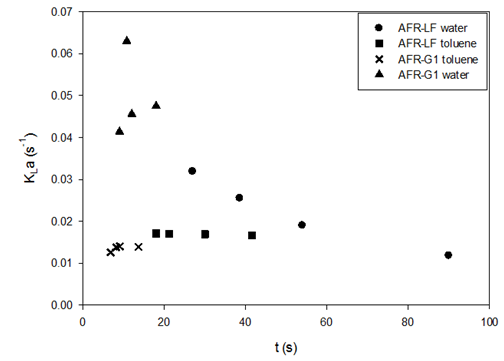
Figure 10 Liquid-side volumetric mass transfer coefficients vs. liquid residence time (t) in AFRs for SO2 absorption in water and toluene at room temperature.
Due to the significant differences in channel sizes and heart-shape cell layout, the smooth change of the KLa vs. residence time across the two levels of ARFs and the relatively similar magnitudes of volumetric mass transfer coefficients indicate seamless scalability of the AFR. The shape of KLa vs. residence time for SO2-water is the same as that for the liquid-liquid system in;23 however, the shape for the SO2-toluene system is different. The changes in polarity, viscosity, and surface tension of the solvent may change the mass transfer behavior for a given gas in the same AFR (Figure 10).
To study low-temperature Bunsen reaction using iodine-toluene solution in the H2S splitting cycle for hydrogen production, the absorption behavior of SO2 in water and toluene was studied in the absence of iodine and the scalability of mass transfer across both micro-and milli-scales of the AFR modules was evaluated. The following conclusions can be drawn from this study:
|
a |
Interfacial area per unit volume of the reactor, m2m-3; |
|
CA |
Concentration at any point in the reactor, mol m-3 |
|
Equilibrium concentration of A at the interface of gas and liquid, mol m-3 |
|
|
Ct |
Total concentration in the liquid phase, mol m-3 |
|
Equilibrium concentration of A (i=1 inlet, i=2 outlet of the reactor), mol m-3 |
|
|
DA |
Diffusivity of A in the gas phase, m2s-1 |
|
FG |
Molar flow rate of inert in the gas phase, mol s-1 |
|
FL |
Molar flow rate of inert gas in the liquid phase, mol s-1 |
|
HA |
Henry’s coefficient, Pa m3mol-1 |
|
kG |
Gas-side mass transfer coefficient, mol m-2Pa-1s-1 |
|
kL |
Liquid-side mass transfer coefficient, m s-1 |
|
KGa |
Gas-side volumetric mass transfer coefficient, mol m-3Pa-1s-1 |
|
KLa |
Liquid-side volumetric mass transfer coefficient, s-1 |
|
Keq |
Equilibrium constant |
|
NA |
Flux of straight mass transfer at any point in the reactor, mol m-2s-1 |
|
n |
Number of moles, kmol |
|
PA |
Partial pressure of A in the bulk of gas, Pa |
|
Partial pressure of A at the interface of gas and liquid, Pa |
|
|
Pt |
Total pressure in the gas phase, Pa |
|
QG |
Volumetric gas flow rate, mL min-1 |
|
QL |
Volumetric liquid flow rate, mL min-1 |
|
T |
Temperature, K (or oC) |
|
Vr |
Reactor volume, mL (or m3) |
|
ySO2 |
mole fraction of SO2 in gas phase |
|
z |
Film thickness, m |
|
Z |
Coordinates, m |
Greek letters
τ Residence time, s
None.
The author declares that there are no conflicts of interest.

©2019 Moniri, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.