International Journal of
eISSN: 2470-9980


Research Article Volume 4 Issue 1
1National Council of Research of Italy, Institute for the Science and Technology of Ceramics, Italy
2International Clean Water Institute, USA
3Nanodiagnostics srl, Italy
Correspondence: Dr. Antonietta Gatti, National Council of Research of Italy, c/o Nanodiagnostics Via E. Fermi, 1/L, 41057 San Vito (MO), Italy, Tel 059798778
Received: November 30, 2016 | Published: January 23, 2017
Citation: Gatti AM, Montanari S (2016) New Quality-Control Investigations on Vaccines: Micro- and Nanocontamination. Int J Vaccines Vaccin 4(1): 00072. DOI: 10.15406/ijvv.2017.04.00072
Vaccines are being under investigation for the possible side effects they can cause. In order to supply new information, an electron-microscopy investigation method was applied to the study of vaccines, aimed at verifying the presence of solid contaminants by means of an Environmental Scanning Electron Microscope equipped with an X-ray microprobe. The results of this new investigation show the presence of micro- and nanosized particulate matter composed of inorganic elements in vaccines’ samples which is not declared among the components and whose unduly presence is, for the time being, inexplicable. A considerable part of those particulate contaminants have already been verified in other matrices and reported in literature as non biodegradable and non biocompatible. The evidence collected is suggestive of some hypotheses correlated to diseases that are mentioned and briefly discussed.
Keywords:vaccine, disease, contamination, protein corona, biocompatibility, toxicity, nanoparticles, immunogenicity, foreign body, environment, industrial process, quality control
Vaccines are one of the most notable inventions meant to protect people from infectious diseases. The practice of variolation is century-old and is mentioned in Chinese and Indian documents dated around 1000 A.D. Over time, variolation has been replaced by vaccination, vaccines have been enhanced as to technology, and the vaccination practice is now standardized worldwide.
Side effects have always been reported but in the latest years it seems that they have increased in number and seriousness, particularly in children as the American Academy of pediatrics reports.1,2 For instance, the diphtheria-tetanus-pertussis (DTaP) vaccine was linked to cases of sudden infant death syndrome (SIDS);3 measles-mumps-rubella vaccine with autism;4,5 multiple immunizations with immune disorders;6 hepatitis B vaccines with multiple sclerosis, etc.
The notice of Tripedia DTaP by Sanofi Pasteur reports “Adverse events reported during post-approval use of Tripedia vaccine include idiopathic thrombocytopenic purpura, SIDS, anaphylactic reaction, cellulitis, autism, convulsion/grand mal convulsion, encephalopathia, hypotonia, neuropathy, somnolence and apnea”. The epidemiological studies carried out did not show a clear evidence of those associations, even if in 2011 the National Academy of Medicine (formerly, IOM) admitted: "Vaccines are not free from side effects, or adverse effects".7
Specific researches on components of the vaccines like adjuvants (in most instances, Aluminum salts) are already indicated as possible responsible of neurological symptoms8‒10 and in some cases, in-vivo tests and epidemiological studies demonstrated a possible correlation with neurological diseases.10,11 Neurological damages induced in patients under hemodialysis treated with water containing Aluminum are reported in literature.12
Recently, with the worldwide-adopted vaccines against Human Papillomavirus (HPV), the debate was reawaken due to some adverse effects reported by some young subjects.
Specific studies communicated the existence of symptoms related to never-described-before syndromes developed after the vaccine was administered. For instance, Complex Regional Pain Syndrome (CRPS), Postural Orthostatic Tachycardia Syndrome (POTS), and Chronic Fatigue Syndrome (CFS).13 The side-effects that can arise within a relatively short time can be local or systemic.
Pain at the site of injection, swelling and uncontrollable movement of the hands (though this last symptom can also be considered systemic) are described. Among the systemic effects, fever, headache, irritability, epileptic seizures, temporary speech loss, lower limbs dysaesthesia and paresis, hot flashes, sleep disorders, hypersensitivity reactions, muscle pain, recurrent syncope, constant hunger, significant gait impairment, incapacity to maintain the orthostatic posture are reported.14
It is a matter of fact that every day millions of vaccine doses are administered and nothing notable happens, but it is also irrefutable that, regardless of the amount of side effects that are not recorded and the percentage of which remains in fact unknown, in a limited number cases something wrong occurs. No satisfactory explanation or, in many cases, no explanation at all has been given and it seems that those adverse effects happen on a random and stochastic basis.
Those situations induced us to verify the safety of vaccines from a point of view which was never adopted before: not a biological, but a physical approach. So, we developed a new analysis method based on the use of a Field Emission Gun Environmental Scanning Electron Microscope investigations to detect possible physical contamination in those products.
44 types of vaccines coming from 2 countries (Italy and France) were analyzed. Table 1 groups them in terms of name, brand and purpose.
N |
Name |
Brand Name, Country of Distribution |
Description |
Production Batch, Expiry Date |
||
1 |
Vivotif Berna |
Berna Biotech SA, Italy |
Anti-Thyphoid Vaccine (Live), group Ty21a |
3000336 [2004] |
||
2 |
Typhim Vi |
Aventis Pasteur MSD, Italy |
Anti-Salmonella typhi Vaccine |
U1510-2 [2004] |
||
3 |
Typherix |
GlaxoSmithKline S.p.a., Italy |
Anti-Thypoid Vaccine (polysaccharide Vi) |
ATYPB061BB [2009] |
||
4 |
Anatetall |
Chiron (now Novartis) Italy |
Adsorbed anti-Tetanus Vaccine |
030106 [2004] |
||
5 |
Anatetall |
Novartis Vaccines and Diagnostics, Italy |
Adsorbed anti-Tetanus Vaccine |
060510 [2009] |
||
6 |
Tetabulin |
Baxter AG, Italy |
Adsorbed anti-Tetanus Vaccine |
VNG2G006A [2009] |
||
7 |
Dif-Tet-All |
Novartis Vaccines and Diagnostics, Italy |
Adsorbed anti-Tetanus and diphtheria Vaccine |
070501 [2009] |
||
8 |
Infanrix |
GlaxoSmithKline S.p.a., Italy |
Anti-Diphtheria, tetanus and pertussis vaccine |
AC14B071AJ [2009] |
||
9 |
Infanrix hexa |
GlaxoSmithKline Biologicals s, Italy |
Anti-diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis |
A21CC512A [ 2017] |
||
10 |
Infanrix hexa |
GlaxoSmithKline Biologicals s. a. France |
Anti-diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis |
A21CC421A [ 2017] |
||
11 |
M-M-R vaxPro |
Sanofi Pasteur MSD, Italy |
M-M-R vaxPro (measles, mumps, and rubella) |
L012437 [ 2017] |
||
12 |
Repevax |
Sanofi Pasteur MSD, France |
Anti-diphtheria-tetanus-pertussis-polio-vaccine |
L0362-1 [2017] |
||
13 |
Repevax |
Sanofi Pasteur MSD SNC France |
Anti-diphtheria-tetanus-pertussis-polio-vaccine |
L0033-1 [2016] |
||
14 |
Priorix |
GlaxoSmithKline S.p.a., Italy |
Anti--measles-mumps, and rubella (MMR) vaccine |
A69CB550A [2009] |
||
15 |
Morupar |
Chiron (now Novartis, ), Italy |
Anti-measles- mumps, and rubella (MMR) vaccine |
7601 [2004] |
||
16 |
Varilrix |
GlaxoSmithKline S.p.a., Italy |
Anti-Chicken pox vaccine (group OKA) |
A70CA567A [2009] |
||
17 |
Stamaril Pasteur |
Sanofi Pasteur MSD, Italy |
anti-yellow fever vaccine |
A5329-6 [2009] |
||
18 |
Allergoid-Adsorbat 6-Graser Starke B. |
Allergopharma, Germany |
Antiallergic vaccine |
Ch-B.:30005999-B [2006] |
||
19 |
Engerix-B |
GlaxoSmithKline S.p.a., Italy |
Adsorbed anti-hepatitis B vaccine |
AHBVB468BD [2009] |
||
20 |
Prenevar 13 |
Pfizer, Italy |
Antipneumococcal vaccine |
G79324 [2013] |
||
21 |
Prevenar 13 |
Pfizer, France |
Antipneumococcal vaccine |
N27430 [ 2018] |
||
22 |
Mencevax Acwy |
GlaxoSmithKline, Italy |
anti-Neisseria meningococcal group A, C, W135 and Y vaccine |
N402A47B 12 [2004] |
||
23 |
Meningitec |
Pfizer, Italy |
(group C 10) (adsorbed on Al-Phosphate) |
H92709 [2015] |
||
24 |
Meningitec |
Pfizer-Italy |
Anti-meningococcus (group C 10) vaccine (adsorbed on Al-Phosphate ) |
H20500 [2014] |
||
25 |
Meningitec |
Pfizer-Italy |
Anti-meningococcus vaccine sequestred by Procura della Repubblica |
G76673 [2014] |
||
26 |
Meningitec |
Pfizer-Italy |
Anti-meningococcus vaccine sequestred by Procura della Repubblica |
H99459 [2015] |
||
27 |
Meningitec |
Pfizer-Italy |
Anti-meningococcus vaccine sequestred by Procura della Repubblica |
H52269 [2015] |
||
28 |
Menjugate |
Novartis Vaccines and Diagnostics |
Anti-meningococcus group C |
YA0163AB [2010] |
||
29 |
Menveo |
Novartis Vaccines and Diagnostics |
Antimeningococcus groups A, C, W135, Y |
A15083 [2017] |
||
30 |
Meningitec |
Wyeth Pharmaceutical - France |
Anti-meningococcus group C vaccine |
E83920 [2011] |
||
31 |
Inflexal V |
Berna Biotech |
Anti-flu vaccine 2008/2009 |
3001463-01 [2009] |
||
32 |
Vaxigrip |
Sanofi Pasteur MSD |
Anti-flu vaccine 2008/2009 |
D9703-1 [2009] |
||
33 |
Vaxigrip |
Sanofi Pasteur |
Anti-flu vaccine 2012/2013 |
J8401-1 [2013] |
||
34 |
Vaxigrip |
Sanofi Pasteur, Italy |
Anti-flu vaccine, with inactivated and split virus |
M7319-1 [2016] |
||
35 |
Focetria |
Novartis Vaccines and Diagnostics |
Anti-pandemic flu H1N1 vaccine |
0902401 [2010] |
||
36 |
Agrippal |
Novartis |
Anti-flu vaccine 2012/2013 |
127002A [2013] |
||
37 |
Agrippal |
Novartis vaccines, Italy |
Anti-flu vaccine with inactivated and split virus 2015/2016 - |
152803 [2016] |
||
38 |
Agrippal S1 |
Novartis Vaccines and Diagnostics |
Anti-flu inactivated/superficial antigene v - 2014/2015 |
147302A [2015] |
||
39 |
Fluarix |
GlaxoSmithKline - GSK |
Anti-flu vaccine 2013 |
AFLUA789AA [2014] |
||
40 |
Fluad |
Novartis Vaccines and Diagnostics |
Anti-flu inactivated/superficial antigene vaccine - 2014/2015 |
142502 [2015] |
||
41 |
Gardasil |
Sanofi Pasteur MSD, Italy |
Anti-HPV types 6,11,16,18 vaccine |
NP01250 [2012] |
||
42 |
Gardasil |
Sanofi Pasteur MSD, Italy |
Anti-HPV (types 6,11,16,18) vaccine |
K023804 [2016] |
||
43 |
Cervarix |
GlaxoSmithKline Biological, Italy |
Anti-HPV (type 16,18) |
AHPVA238AX [2017] |
||
44 |
Feligen CRP |
Virbac S.A. - Carros - Italy |
anti-panleucopenia, infectious rhinotracheitis and infections |
3R4R [2013] |
||
Table 1 List of vaccines analyzed, according to their purpose
Some vaccines, in fact a minority, are meant to deal with a single bacterium or virus, while others are multi-valent. The list of vaccines we analyzed may contain repeated names, because we considered different batches and years of production of the same vaccine: the ones against influenza in particular.
The study was aimed at verifying a possible physical contamination. To do that, we performed a new kind of investigation based on observations under a Field Emission Gun Environmental Electron Scanning Microscope (FEG-ESEM, Quanta 200, FEI, The Netherlands) equipped with the X-ray microprobe of an Energy Dispersive Spectroscope (EDS, EDAX, Mahwah, NJ, USA) to detect the possible presence of inorganic, particulate contaminants and identify their chemical composition.
A drop of about 20microliter of vaccine is released from the syringe on a 25-mm-diameter cellulose filter (Millipore, USA), inside a flow cabinet. The filter is then deposited on an Aluminum stub covered with an adhesive carbon disc. The sample is immediately put inside a clean box in order to avoid any contamination and the box is re-opened only for the sample to be inserted inside the FEG-ESEM chamber. We selected that particular type of microscope as it allows to analyse watery and oily samples in low vacuum (from 10 to 130 Pa) at a high sensitivity.
When the water and saline the vaccine contains are evaporated, the biological/physical components emerge on the filter and it is then possible to observe them. This type of microscope (low-vacuum observations) prevents the possible sample contamination and the creation of artefacts. The observations are made with different sensors (SE: secondary-electron sensor and BSE: backscattered-electron sensor), and are performed at a pressure of 8.9 e-1 mbar, at energies ranging from 10 to 30kV to detect the particulate matter’s size, morphology and its elemental composition. The method identifies clearly inorganic bodies with a higher atomic density (looking whiter) than the biological substrate. So, organic entities are visible and easy to distinguish from inorganic ones. The method cannot distinguish between proteins and organic adjuvants (e.g. squalene, glutamate, proteins, etc.) or viruses, bacteria, bacteria’s DNA, endo-toxins and bacteria’s waste, but their comparatively low atomic density allows us to identify these entities as organic matter. In some vaccines, the organic matter contains white-looking debris named aggregates, while a high concentration or inorganic debris is called a cluster.
Single inorganic particles or organic-inorganic aggregates are identified, evaluated and counted. The counting procedure is repeated three times by three different operators, with an error lower than 10%. When a layer of salts (Sodium chloride or Aluminum) is detected, we record the situation but we do not do body count.
The investigations verified the physical-chemical composition of the vaccines considered according to the inorganic component as declared by the Producer. In detail, we verified the presence of saline and Aluminum salts, but further presence of micro-, sub-micro- and nanosized, inorganic, foreign bodies (ranging from 100nm to about ten microns) was identified in all cases, whose presence was not declared in the leaflets delivered in the package of the product (Table 2).
N |
Company |
Name |
Alluminum |
Elements Identified |
1 |
Allergopharma - Germany |
Allergoid |
yes |
Al |
2 |
Aventis Pasteur MSD Lyon - Francie |
Typhim Vi |
no |
BrKP, PbSi, FeCr, PbClSiTi |
3 |
Baxter AG |
Tetabulin |
no |
SiMg, Fe, SiTiAl, SBa, Zn |
4 |
Berna Biotech |
Vivotif Berna |
no |
FeAl, ZrAlHf, SrAl, BiAlCl |
5 |
Berna Biotech |
Inflexal V |
no |
CuSnPbZn, Fe, CaSiAl, SiAl, NaPZn, ZnP, AlSiTi |
6 |
Chiron |
Anatetall |
Al(OH)3 |
FeAl, SZnBaAl |
7 |
Chiron |
Morupar |
no |
/ |
8 |
GlaxoSmithKline- Belgium |
Mencevax ACWY |
no |
FeCrNi, ZrAl, FeCrNiZrAlSi |
9 |
GlaxoSmithKline |
Infanrix |
Al(OH)3 |
Al, AlTi, AlSi |
10 |
GlaxoSmithKline Biologicals |
Infanrix hexa |
Al(OH)3 |
SBa, FeCu, SiAl, FeSi, CaMgSi, AlCaSi, Ti, Au, SCa, SiAlFeSnCuCrZn, CaAlSi |
11 |
GlaxoSmithKline Biologicals |
Infanrix hexa |
Al(OH)3 ,AlPO4. 2H2O |
W, FeCrNi, Ti |
12 |
GlaxoSmithKline |
Typherix |
no |
Ti, TiW, AlSiTiWCr, SBa, W, SiAl, AlSiTi |
13 |
GlaxoSmithKline |
Priorix |
no |
WCa, WFeCu, SiAl, SiMg, PbFe, Ti, WNiFe |
14 |
GlaxoSmithKline |
Engerix-B |
no |
Al (precipitates) |
15 |
GlaxoSmithKline |
Varilrix |
no |
FeZn, FeSi, AlSiFe, SiAlTiFe, MgSi, Ti, Zr, Bi |
16 |
GlaxoSmithKline |
Fluarix |
no |
AlCu, Fe, AlBi, Si, SiZn, AlCuFe, SiMg, SBa, AlCuBi, FeCrNi, SPZn |
17 |
GlaxoSmithKline Biologicals |
Cervarix |
Al(OH)3 |
AlSi,FeAl, SiMg,CaSiAl, CaZn, FeAlSi, FeCr, CuSnPb |
18 |
Novartis Vaccines and Diagnostics |
Anatetall |
Al(OH)3 |
Al, FeCrNi, AlCr, AlFe, BaS, ZnAl |
19 |
Novartis Vaccines and Diagnostics |
Dif-Tet-All |
Al(OH)3 |
Fe,SBa, SiSBa, AlZnCu, AlZnFeCr |
20 |
Novartis Vaccines and Diagnostics |
Menjugate kit |
Al(OH)3 |
SiAl,Ti,FeZn, Fe, Sb, SiAlFeTi, W, Zr |
21 |
Novartis Vaccines and Diagnostics |
Focetria |
no |
Fe, FeCrNiCu, FeCrNi, SiFeCrNi, Cr, SiAlFe, AlSiTiFe, AlSi, SiMgFe, Si, FeZn |
22 |
Novartis |
Agrippal S1 |
no |
Ca, Fe, SBa, SBaZn, Cr, Si, Pb, Bi, e FeSiAlCr, SiAlSBaFe, CaAlSi, Zn, CeFeTiNi, FeCrNi |
23 |
Novartis Vaccines and Diagnostics |
Agrippal S1 |
no |
SiAlK, Si, SiMgFe, CaSiAl, SBaZn |
24 |
Novartis vaccines |
Agrippal |
no |
Cr, Ca, SiCaAl, ZrSi, SBa, CuZn, SCa |
25 |
Novartis Vaccines and Diagnostics S |
Fluad |
no |
CaSiAl, FeSiTi,SiMgAlFe, SBa |
26 |
Novartis Vaccines and Diagnostics |
Menveo |
no |
CaSiAl, SiAlFe, FeCrNi, Fe, Al, SBa |
27 |
Pfizer |
Prenevar 13 |
no |
FeCr |
28 |
Pfizer |
Prevenar 13 |
no |
W, CaAlSi, Al, CaSiAlFe, FeS, FeCr, FeCrNi, Fe, , CaP, FeTiMn, Ba, SiMgAlFe |
29 |
Pfizer |
Meningitec - ctrl |
no |
Cr, Si |
30 |
Pfizer |
Meningitec - ctrl |
no |
FeCrNi, W |
31 |
Pfizer |
Meningitec |
no |
CaSiAl, CaSi, SiAlFeTi, FeCrNi, W, Fe, Pb |
32 |
Pfizer |
Meningitec |
no |
Cr (precipitates), Ca, AlSi |
33 |
Pfizer |
Meningitec |
no |
W, SiCa, CaSi, Pb, FeCrNi, Cr |
34 |
Wyeth Pharmaceutical - UK |
Meningitec |
no |
SiAlFe, SiAlTi, SiMgFe, W, Fe, Zr, Pb, Ca, Zn, FeCrNi |
35 |
Sanofi Pasteur MSD-France |
Vaxigrip |
no |
Fe, FeCrNi, SiAlFe, AlSi, SiAlFeCr |
36 |
Sanofi Pasteur MSD |
Stamaril Pasteur |
no |
CaSiAl, AlSi, Fe, SiMgFe, SiMgAlFe, CrSiFeCr, CrSiCuFe |
37 |
Sanofi Pasteur MSD |
Gardasil |
AlPO4. 2H2O |
AlCuFe, PbBi, Pb, Bi, Fe |
38 |
Sanifi Pasteur MSD |
Gardasil |
AlPO4. 2H2O |
CaAlSi, AlSi, SiMgFe, Al,Fe, AlCuFe, FeSiAl, BiBaS, Ti, TiAlSi |
39 |
Sanofi Pasteur |
Vaxigrip |
no |
Ca, CrFe, FeCrNi, CaSZn, CaSiAlTiFe, Ag, Fe |
40 |
Sanofi Pasteur |
Vaxigrip |
no |
SiMgFe, CaSiAl, AlSiFe, AlSi,FeCr, FeZn, Fe |
41 |
Sanofi Pasteur MSD |
Repevax |
AlPO4 .2H2O |
Bi, Fe, AlSiFe, SiMg, SBa, Ca |
42 |
Sanofi Pasteur MSD S |
Repevax |
AlPO4.2H2O |
Ti, Br, AuCuZn, Ca, SiZn, SiAuAgCu, SiMgFe,FeCrNi.AlSiMgTiMnCrFe, SiFeCrNi, FeAl |
43 |
Sanofi Pasteur MSD |
M-M-R vaxPro |
no |
Si, SiFeCrNi, FeCrNi,FeNi, Fe, SCa, AlSiCa, CaAlSiFeV, SBa, Pt, PtAgBiFeCr |
44 |
Virbac S.A. - Carros - France |
Feligen CRP |
no |
Ca,SiAl |
Table 2 List of the vaccines according to their manufacturers with the chemical composition of the debris identified in each sample. The elements most represented are reported
Play Video
Figure 1a shows a layer of crystals of Sodium chloride (NaCl) embedding salts of Aluminum phosphate (AlPO4) in a drop of Gardasil (anti-HPV vaccine by Merck) as the EDS spectrum (Figure 1b) shows. Saline is the fluid base to any vaccine preparation and Aluminum salts or Aluminum hydroxide Al(OH)3 are the adjuvants which are usually added.
Looking at the area outside these precipitates but inside the liquid drop, we identified other things: single particles, clusters of particles and aggregates (organic-inorganic composites) that are due to an interaction of the inorganic particulate matter with the organic part of the vaccine.
Figure 2a-2f shows the different typology of entities identified in the vaccines (Repevax, Prevenar and Gardasil); single particles, cluster of micro- and nanoparticles (<100nm) and aggregates with their EDS spectra (Figure 2d-2f). The images (Figure 2a & 2d) show debris of Aluminum, Silicon, Magnesium and Titanium; of Iron, Chromium, Silicon and Calcium particles (Figure 2b & 2e) arranged in a cluster, and Aluminum -Copper debris (Figure 2c & 2f) in an aggregate.
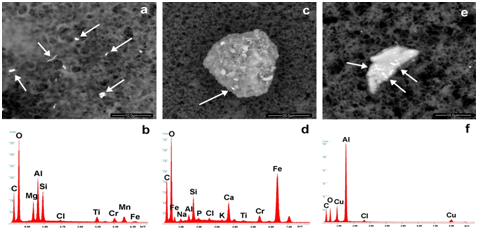
Figure 2 Images of single particles, cluster of micro- and nanoparticles (<100nm) andaggregates with their EDS spectra. They are respectively composed of (a,b) Aluminum, Silicon, Magnesium, Titanium, Chromium, Manganese, Iron, (c,d) Iron, Silicon, Calcium Titanium, Chromium, (e,f) Aluminum, Copper. The arrows show the points where EDS spectra were taken.
As can be seen, the particles are surrounded and embedded in a biological substrate. In all the samples analyzed, we identified particles containing: Lead (Typhym, Cervarix, Agrippal S1, Meningitec, Gardasil) or stainless steel (Mencevax, Infarix Hexa, Cervarix. Anatetall, Focetria, Agrippal S1, Menveo, Prevenar 13, Meningitec, Vaxigrip, Stamaril Pasteur, Repevax and MMRvaxPro).
Figure 3a-3d show particles of Tungsten identified in drops of Prevenar and Infarix (Aluminum, Tungsten, Calcium chloride).
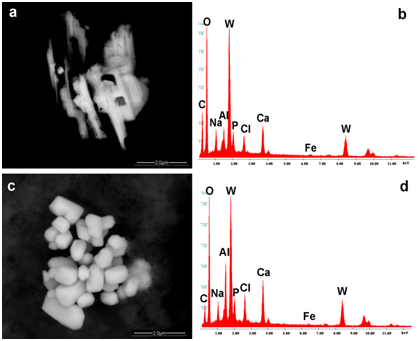
Figure 3 Images of Tungsten particles identified in drops of Prevenar and Infarix. They are composed respectively of Tungsten, Aluminum, Iron but in different concentrations. The arrows show the points where EDS spectra were taken.
Figure 4a-4d present singular debris found in Repevax (Silicon, Gold, Silver) and Gardasil (Zirconium).
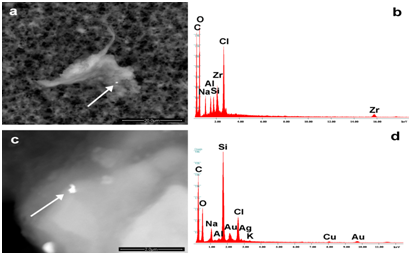
Figure 4 Images show examples of nano biointeraction. The aggregate (a,b) identified in Gardasil contains nanoparticles of Chlorine, Silicon, Aluminum, Zirconium, while the debris found in Repevax contains Silicon, Gold, Silver (c,d). The arrows show the points where EDS spectra were taken.
Some metallic particles made of Tungsten or stainless steel were also identified. Other particles containing Zirconium, Hafnium, Strontium and Aluminum (Vivotif, Meningetec); Tungsten, Nickel, Iron (Priorix, Meningetec); Antimony (Menjugate kit); Chromium (Meningetec); Gold or Gold, Zinc (Infarix Hexa, Repevax), or Platinum, Silver, Bismuth, Iron, Chromium (MMRvaxPro) or Lead,Bismuth (Gardasil) or Cerium (Agrippal S1) were also found. The only Tungsten appears in 8/44 vaccines, while Chromium (alone or in alloy with Iron and Nickel) in 25/44. The investigations revealed that some particles are embedded in a biological substrate, probably proteins, endo-toxins and residues of bacteria. As soon as a particle comes in contact with proteic fluids, a nano-bio-interaction6 occurs and a "protein corona" is formed.7‒10 The nano-bio-interaction generates a bigger-sized compound that is not biodegradable and can induce adverse effects, since it is not recognized as self by the body.
Figure 5a-5f show examples of these nano-bio-interactions. Aggregates can be seen (stable composite entities) containing particles of Lead in Meningitec, (Figure 5a & 5b) of stainless steel (Iron, Chromium and Nickel, Figure 5c & 5d) and of Copper, Zinc and Lead in Cervarix (Figure 5e & 5f). Similar aggregates, though in different situations (patients suffering from leukemia or cryoglobulinemia), have already been described in literature.
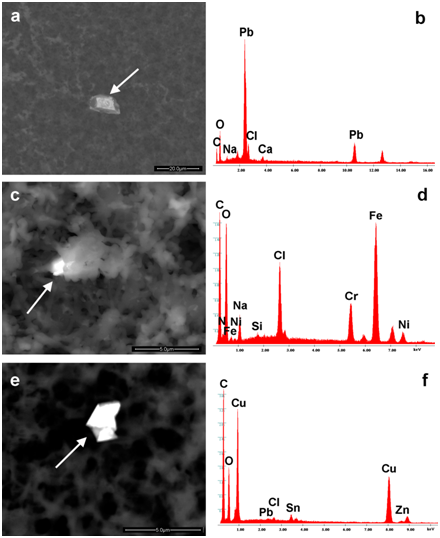
Figure 5 Show particles surrounded by an organic compound. They are composed of Lead (a,b), Iron, Chromium, Nickel (stainless steel; c,d), Copper, Tin, Lead (e,f). The arrows show the points where EDS spectra were taken.
The link between these two entities generates an unfolding of the proteins that can induce an autoimmune effect once those proteins are injected into humans.
Figure 6a & 6b show one of the foreign bodies identified in Agrippal. The particle is composed of Cerium, Iron, Titanium and Nickel. (Figure 7a & 7b) present an area of Repevax where the morphology of red cells - we cannot tell whether they are human or animal- is clearly visible.
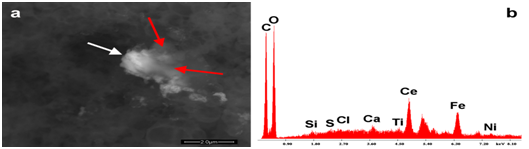
Figure 6 Show an organic aggregate containing a debris made of Cerium, Iron, Nickel, Titanium. The red arrow indicates the organic layer (less atomically dense) that covers the Cerium particle.

Figure 7 Image of an area in a Repevax drop where the morphology of red cells (red arrows) were identified. It is impossible to know whether they are human or animal origin. Among the debris of saline and Aluminum phosphate, there is the presence of debris (white arrows) composed of Aluminum, Bromine, Silicon, Potassium, Titanium.
Table 3 summarizes the number and morphology of the debris identified, in term of single particles, clusters of particles or aggregates (organic-inorganic compounds), while Figure 8 shows the graph obtained calculating the total number of particles (particles plus clusters plus aggregates) identified for 20microl of every vaccine.
Name |
Total Debris n. |
Size Range in mm |
Cluster n. |
Size Range in mm |
Aggregate n.(Range of Particles) |
Size Range mm |
|
Allergoid |
NaCl precipitates |
/ |
/ |
/ |
/ |
/ |
|
Typhim Vi |
394 |
0,1-2,5 |
3[9-350] |
Feb-35 |
|||
Tetabulin |
519 |
0,1-15 |
3[100-180] |
25-60 |
|||
Vivotif Berna |
4 |
1,5-15 |
|||||
Inflexal V |
103 |
0,1-17 |
1 |
20 |
3[35-45] |
25-Oct |
|
Anatetall |
2 |
3-Jan |
|||||
Morupar |
/ |
/ |
/ |
||||
Mencevax ACWY |
13 |
0,2-5 |
|||||
Infanrix |
3 |
5-Jan |
1 |
25 |
|||
Infanrix hexa |
1821 |
0,1-15 |
15[1820] |
20-140 |
|||
Infanrix hexa |
162 |
0,3-7 |
12 |
60 |
2[7 debris] |
3.5-44 |
|
Typherix |
8 |
0,2-8 |
1 |
15 |
|||
Priorix |
641 |
0,05-30 |
1 |
10 |
3[600] |
20-70 |
|
Engerix-B |
precipitates |
/ |
|||||
Varilrix |
2723 |
0,1-4 |
36 [120-2000] |
15-40 |
|||
Fluarix |
1317 |
0,1-40 |
3[83] |
30-Jul |
|||
Cervarix |
1569 |
0,2-3 |
2 |
10-May |
4[1400] |
30-Aug |
|
Anatetall |
47 |
0,05-40 |
|||||
Dif-Tet-All |
111 |
0,2-3 |
|||||
Menjugate |
73 |
0,1-5 |
|||||
Focetria |
35 |
0,7-10 |
|||||
Agrippal S1 |
430 |
0,2-6 |
13 |
0.2-80 |
5[410] |
20 |
|
Agrippal S1 |
1029 |
0,1-12 |
9[1025] |
35-80 |
|||
Agrippal |
480 |
0,1-6 |
11[ 460] |
Feb-80 |
|||
Fluad |
605 |
0,2-15 |
4 |
25-Dec |
6[ 600] |
70 |
|
Menveo |
452 |
0,1-13 |
4[430 ] |
30-110 |
|||
Prenevar 13 |
precipitates + 5 debris |
7-Jan |
|||||
Prevenar 13 |
precipitates + 81 debris |
0,2-50 |
3 |
May-40 |
1 [60] |
25 |
|
Meningitec |
3 |
20-Oct |
|||||
Meningitec |
24 |
Aug-60 |
|||||
Meningitec |
673 |
0,1-20 |
1 |
7 |
9[624] |
5-110 |
|
Meningitec |
precipitates + 40 |
0,1-3,5 |
2[40] |
25-70 |
|||
Meningitec |
177 |
0,2-10 |
3[165] |
15-100 |
|||
Meningitec |
241 |
0,1-15 |
1 |
50 |
2[230] |
50 |
|
Vaxigrip |
86 |
0,1-7 |
2[50] |
2-2,5 |
|||
Stamaril Pasteur |
152 |
0,1-7 |
2 |
7-May |
3[145] |
20-Apr |
|
Gardasil |
304 |
0,05-3 |
1[300] |
15 |
|||
Gardasil |
454 |
0.1-30 |
2 |
20-Jul |
9[445] |
May-60 |
|
Vaxigrip |
304 |
0,1-10 |
1 |
13 |
2[300] |
35 |
|
Vaxigrip |
674 |
0,3-25 |
2 |
12-Feb |
10[660] |
9-150 |
|
Repevax |
137 |
0,1-20 |
2[130] |
40-50 |
|||
Repevax |
214 |
0,1-10 |
6[150] |
30-May |
|||
M-M-R vaxPro |
93 |
0,1-15 |
2[50] |
15-Oct |
|||
Feligen CRP |
92 |
0,1-12 |
1 |
12 |
1 (40 debris) |
25 |
|
Table 3 List of the debris’ number identified in each vaccine as single particle, clusters and aggregates.
Characterization is made by shape, size range and variability of the number of particles counted in each aggregate [in brackets]
Similar aggregates were already described by other scientists who identified them in the blood e.g. in leukemic patients15 and in subjects affected by cryoglobulinemia.16
Not all the vaccines analyzed contain the same contamination, though the same vaccine belonging to different batches and, in some cases, coming from different countries can contain a similar contamination (e.g. the vaccines by Glaxo Infarix, Typherix and Priorix contain Tungsten. Tungsten was also identified in Menjugate kit by Novartis, and Prevenar, Meningitec by Pfizer and Meningitec by Wyeth).
Feligen, the only veterinary vaccine tested, proved to be the only sample free from inorganic contamination, while Allergoid generates a layer of inorganic salts so thick that it does not allow to detect other particulate contaminants.
The quantity of foreign bodies detected and, in some cases, their unusual chemical compositions baffled us. The inorganic particles identified are neither biocompatible nor biodegradable, that means that they are biopersistent and can induce effects that can become evident either immediately close to injection time or after a certain time from administration. It is important to remember that particles (crystals and not molecules) are bodies foreign to the organism and they behave as such. More in particular, their toxicity is in some respects different from that of the chemical elements composing them, adding to that toxicity which, in any case, is still there, that typical of foreign bodies. For that reason, they induce an inflammatory reaction.
After being injected, those microparticles, nanoparticles and aggregates can stay around the injection site forming swellings and granulomas.17 But they can also be carried by the blood circulation, escaping any attempt to guess what will be their final destination. We believe that in many cases they get distributed throughout the body without causing any visible reaction, but it is also likely that, in some circumstances, they reach some organ, none excluded and including the microbiota, in a fair quantity. As happens with all foreign bodies, particularly that small, they induce an inflammatory reaction that is chronic because most of those particles cannot be degraded. Furthermore, the protein-corona effect (due to a nano-bio-interaction18 can produce organic/inorganic composite particles capable of stimulating the immune system in an undesirable way.19‒22 It is impossible not to add that particles the size often observed in vaccines can enter cell nuclei and interact with the DNA.23
In some cases, e.g. as occurs with Iron and some Iron alloys, they can corrode and the corrosion products exert a toxicity affecting the tissues.24‒26
The detection of presence of Aluminum and NaCl salts is obvious as they are substances used by the Producers and declared as components, but other materials are not supposed to be in the vaccine or in any other injectable drug, at that, and, in any case, Aluminum has already been linked with neurological diseases.27‒29
Given the contaminations we observed in all samples of human-use vaccines, adverse effects after the injection of those vaccines are possible and credible and have the character of randomness, since they depend on where the contaminants are carried by the blood circulation. It is only obvious that similar quantities of these foreign bodies can have a more serious impact on very small organisms like those of children. Their presence in the muscles, due an extravasation from the blood, could heavily impair the muscle functionality.30,31
We come across particles with chemical compositions, similar to those found in the vaccines we analyzed, when we study cases of environmental contamination caused by different pollution sources. In most circumstances, the combinations detected are very odd as they have no technical use, cannot be found in any material handbook and look like the result of the random formation occurring, for example, when waste is burnt. In any case, whatever their origin, they should not be present in any injectable medicament, let alone in vaccines, more in particular those meant for infants.
Other forms of so-far unknown contaminations have recently been observed and, in any case, vaccines contain components that could themselves be the cause of adverse effects. It is a well-known fact in toxicology that contaminants exert a mutual, synergic effect, and as the number of contaminants increases, the effects grow less and less predictable. The more so when some substances are unknown.
As a matter of fact, no exhaustive and reliable official data exist on the side-effects induced by vaccines. The episodic evidence reported by people allegedly damaged by vaccines is twofold: some say the damage occurred and became visible within a few hours from administration, and some maintain that it was a matter of some weeks. Though we have no indisputable evidence as to the reliability of those attestations, we can put forward a hypothesis to explain the different phenomena. In the former case, the pollutants contained in the drug have reached the brain and, depending on the anatomical site interested, have induced a reaction. If that is the case, the whole phenomenon is very rapid. In the latter circumstance, the pollutants reached the microbiota, thus interfering with the production of enzymes necessary to carry out neurological functions.32‒35 That possibility takes time, as it involves the production of chemical compounds in a sufficient quantity, and an elapse of some weeks between injection and clinical evidence is reasonable. Of course, ours is no more than a hypothesis open to discussion and in need of proof, hoping that a chance of further investigation is allowed.
The analyses carried out show that in all samples checked vaccines contain non biocompatible and bio-persistent foreign bodies which are not declared by the Producers, against which the body reacts in any case. This new investigation represents a new quality control that can be adopted to assess the safety of a vaccine. Our hypothesis is that this contamination is unintentional, since it is probably due to polluted components or procedures of industrial processes (e.g. filtrations) used to produce vaccines, not investigated and not detected by the Producers. If our hypothesis is actually the case, a close inspection of the working places and the full knowledge of the whole procedure of vaccine preparation would probably allow to eliminate the problem.
A further purification of the vaccines could improve their quality and could probably decrease the number and seriousness of the adverse incidental effects.
The Authors are indebted to Dr. Federico Capitani, Dr. Laura Valentini and Ms. Lavinia Nitu for their technical assistance. The opinions and conclusions are not necessarily those of the Institution.
Author declares there are no conflicts of interest.
None.

©2017 Gatti, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.