International Journal of
eISSN: 2470-9980


Mini Review Volume 2 Issue 6
US Biologic, Inc., USA
Correspondence: Steve Zatechka, PhD, MBA, Chief Operating Officer, US Biologic, Inc, 20 Dudley, Suite 900, Memphis, TN 38103, USA, Tel 901-755-6868
Received: November 28, 2015 | Published: September 22, 2016
Citation: Zatechka S (2016) Reservoir-Targeted Vaccines as a One Health Path To Prevent Zoonotic Disease. Int J Vaccines Vaccin 2(6): 00049. DOI: 10.15406/ijvv.2016.02.00049
Reservoir-targeted vaccines (RTVs) are emerging as an effective means by which to control the spread of zoonotic and vector-borne diseases. Delivered as an orally-administered antigen, the biologistics involved in the distribution of RTVs are administered as part of a platform solution to address the growing risk from zoonotic and vector-borne diseases. Specific performance measures must therefore be considered in the stable presentation of antigen of a potency to elicit mucosal immunity and control disease transmission. Concomitantly, several criteria are also required in order to administer a commercially feasible and scalable safe and efficacious vaccine into field conditions, for oral consumption. Such a solution further aligns with the One Health Initiative. As an exemplary case study, an RTV addressing the epidemic of Lyme disease is presented.
Keywords: biologistics, encapsulation, enteric, gut associate lymphoid tissue, mucosal immunization, oral administration, reservoir targeted vaccine, stabilization
OspA, outer-surface protein A; RTV, reservoir-targeted vaccine
Since the distinctions between antibody-mediated and cell-mediated immunity were elucidated through the pioneering work conducted by both Doherty & Zinkernagel1 and recognized in 1996 with the awarding of the Nobel Prize in Physiology or Medicine, vaccine evolution has advanced with technologies for the specific presentation of antigen to enhance the immunity onset and duration. While a complex endeavor, early vaccine-mediated protective efficacy is conferred via the induction of antigen-specific antibodies; antibody avidity remains a principal determining factor of efficacy. Further, the duration of protection is dependent upon the persistence of vaccine antibodies. Hence the stable presentation of antigen in the context of an immunomodulator carrier is paramount to the generation of antibody titers at serological-protective levels. The generation of quality antibody titers in disease reservoirs, therefore, constitutes an effective, initiative event to be exploited in the control of zoonotic infectious diseases. Further, a means for the reduction in the prevalence of disease in harboring reservoirs in the field aligns with the strategy defined by the One Health Initiative.2,3 Introduced in this mini review is the concept of a biologistics platform employed as a means to deliver disease protection: the oral delivery of vaccine antigens to reservoir hosts, stably expressed to elicit a mucosal-administered quality antibody response to break the enzootic cycle. Herein, Lyme disease is presented in an exemplified model for the application of this platform.
Controlling zoonotic infectious diseases and antimicrobial resistance contributes to the reduction of disease transmissibility. Current strategies to control zoonotic infectious diseases include the deployment of pesticides as a means to eliminate the vector from the enzootic cycle. However, the use of pesticides presents with toxic off-target effects upon the subject and environment. The use of prophylactic and therapeutic antibiotics, concomitantly, has inadvertently led to the evolution of antimicrobial-resistant strains of infectious agents being maintained in the zoonotic cycle. Further, while susceptible host prophylactic or therapeutic agent campaigns often employ parenteral administration, such administrative methods pose cost and logistics challenges. Compared to parenteral methods, orally delivered prophylactic or therapeutic agents are manufactured more cost effectively, offer a significant ease of use, and cause fewer side effects. However, the development of oral vaccines to the animal reservoir targets responsible for zoonotic diseases presents several challenges for efficacy.
Reviewed here is a novel technology and platform, delivered as a reservoir-targeted vaccine (RTV) in the field, employed as a One Health solution to break the enzootic cycle and reduce the prevalence of zoonotic infectious diseases.
Given that 70% of all emerging and reemerging infections are vector-borne or zoonotic,4 the One Health Initiative is a global collaborative endeavor to strategically unite synergy among the human- and veterinary-medical efforts towards the collective enhancement of global health.2 The One Health collaboration further garners support from scientific-health and environmental related disciplines, which include the American Medical Association (AMA), the Centers for Disease Control and Prevention (CDC), the United States Department of Agriculture (USDA), and the U.S. National Environmental Health Association.
Among the pioneer programs of RTV implementation and deployment is the National Rabies Management Program.5 Initially established in 1995, the Program has been regulated by the USDA Animal and Plant Health Inspection Service (APHIS) and managed by the USDA APHIS Wildlife Services (WS) division.6 Rabies is caused by a virus that infects the central nervous system in mammals. The virus is transmitted through the bite of a rabid animal, most commonly raccoons, skunks, foxes and bats. Considering that the infection is invariably fatal and rabies has expanded through the U.S., another approach such as a vaccination program offers a potential effective method for protection.5
In recognition of rabies as a zoonotic infectious disease with dynamic risk upon human populations in specific geographies, the National Rabies Management Program was established to prevent further spread of wildlife rabies, with the ultimate goal of eliminating terrestrial rabies in the U.S. through the use of the Oral Rabies Vaccination Program (ORV) (Figure 1).

Figure 1 Rabid Animals Reported in the U.S. during 2009.7
ORV employs an RTV as part of a cooperative program to administer vaccine baits to targeted areas. Consumption of the bait by targeted animals vaccinates the animal rendering it immune to the rabies virus. As the number of vaccinated animals in an area increases, herd immunity creates a buffer zone impeding the potential spread of infection to other wildlife, domesticated animals, and humans. As part of the National Rabies Management Program, the distribution of the baits in the ORV program are monitored in real-time and can be accessed through https://usdaaphis.maps.arcgis.com/apps/Viewer/index.html?appid=887bab2f96444b7d9ffa 6127f634bca4.
Commercially, the ORV RTV baits are manufactured by Merial, Inc. under the trade name Raboral V-RG®.8 Post-baiting serology results from the deployment of the ORV RTV have demonstrated a steady increase in sero conversion to levels that maintain a reduction in the number of rabid animals in the wild.9
Considering the precedent established by the utility of the ORV, RTV technologies have emerged as an effective means by which to address other notifiable zoonotic infectious diseases. According to the CDC, Lyme disease remains in the top 10 of the notifiable diseases in the U.S. as reported in 2012,10 and in 2013 the CDC reported that the prevalence of Lyme in the U.S. may have been previously under-reported by a factor of 10X.11 Lyme disease is now the most common vector-borne disease in North America, and the areas where the disease is endemic is expanding, emerging as an epidemic12 (Figure 2).
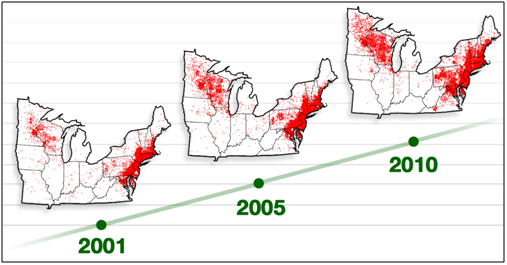
Figure 2 Northeast Progression of Lyme disease in the U.S.11
Globally, the occurrence of Lyme disease follows the same geographical distribution parallel as that which is demonstrated for the prevalence in the U.S. Such foci include the forested areas of Asia and north-western, central, and eastern Europe. As global prevalence potentially increases, given the projected trajectories for endemic areas, Lyme, then, presents as a possible international travel risk (Figure 3).
Borrelia burgdorferi, the etiological agent of Lyme disease, is vector-transmitted among vertebrate wildlife hosts, domesticated animals, and humans by the blacklegged tick, Ixodes scapularis.15 I. scapularis has an approximate two-year life-cycle comprising tick life-stages of egg, larva, nymph and adult; the nymph is responsible for infecting the majority of domesticated animals and humans16,17 (Figure 4).
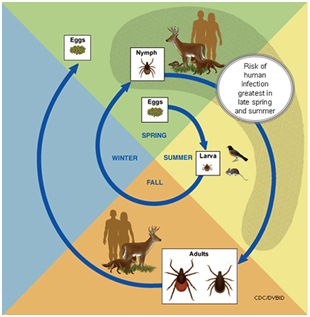
Figure 4 Life Cycle of Ticks that Spread Lyme disease.17
Measures for controlling the spread of infection by targeted disruption of the triad vector-etiological agent-vertebrate reservoir will reduce the B. burgdorferi prevalence in the wild, and should reduce the incidence of Lyme disease18,19 (Figure 5).
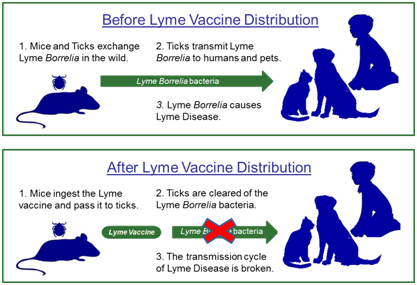
Figure 5 Disruption of the Lyme Disease Transmission Cycle Via a Reservoir-targeted Vaccine (US BIOLOGIC).
Earlier studies have indicated that immunization of mice with a vaccine expressing the Borrelia outer-surface protein A antigen (OspA) can protect mice from infection and eliminate B. burgdorferi from feeding ticks.20,21 Such studies laid the translational ground-work for the employment of the OspA antigen in a human vaccine,22 and complemented supporting studies towards controlling Lyme disease at the level of the reservoir host in an effort to disrupt the enzootic cycle and, respectively, the incidence of infection in humans.23,24
An RTV based upon the work of Maria Gomes-Solecki utilizes an OspA-vectored E. coli carrier as the core of an orally-administered vaccine. Initial research into the development of this technology demonstrated that the vaccine, when orally-administered to mice, could disrupt the Borrelia transmission cycle25 and elicit a yearlong neutralizing antibody response.26 Follow up studies, to include a 5-year CDC-sponsored field trial, demonstrated vaccine efficacy as assayed by the normalized tick infection prevalence.27 In forested field plots, a vaccine amalgam of oat-based baiting substrate was distributed across test fields using Sherman traps stations. Results from the test fields yielded a greater than 75% reduction in the tick infection prevalence to B. burgdorferi by the conclusion of the 5-year study. This, in comparison to a greater than 60% increase in the infection prevalence in ecologically-comparable control fields where vaccine was not distributed. Such results, at the research and development stage, are indeed suggestive of good measure for translating the technology to the subsequent commercial application.
To accomplish the goal of administering an efficacious RTV on a commercial scale, several key technical considerations must be implemented. These factors will be briefly explored.
A baiting formulation, manufactured at efficient scale, must be employed as a means to target the administration of the vaccine specifically to the species reservoir harboring the disease. Further, the bait must be palatable in terms of affording an opportunity for manipulation and oral consumption by the targeted species. While biodegradation is of importance for environmental distribution, the baiting substrate must persist long enough to accommodate targeted consumption. The use of biomarkers (Rhodamine B (RB), tetracycline) present as effective measures to track consumption metrics on targeted reservoir hosts, and have been historically used in the bait consumption studies of Raboral®.28 RB has been widely used as a wildlife marker29 and was employed in the initial commercial development of the RTV for Lyme disease to evaluate baiting consumption metrics, and administration concentrations followed that which are described elsewhere.30,31 In collaboration with the Connecticut Agricultural Experiment Station, distribution of RB-amended baiting resulted in an observed 91% consumption rate by visual inspection (UV lamp) and whisker analysis (RB-induced whisker banding) for the baiting substrate in the target species, P. leucopus, over multiple trapping rounds.32
Given a baiting formulation that targets the disease reservoir host species, there must be engineered into the vaccine bait an inherent biological stability measure to ensure shelf-life and stable administration. Once consumed, oral vaccines are subject to the harsh conditions of the digestive system and an enteric stabilization matrix enhances delivery of the antigen, for conformational presentation, to the mucosa-associated lymphoid tissue (MALT) of the gut, the gut-associated lymphoid tissue (GALT).
Animal mucosal surfaces present as principle entry sites for many infectious agents. Consequently, mucosal immunity offers an initial line of defense against infectious agents. The mucosal immune response interferes with the infectious process by hindering pathogen attachment to the mucosal epithelium, neutralizing viral and bacterial agents, and providing the means by which to remove pathogens through phagocytosis. The mucosal-associated immune system functions to prevent microbial penetration and infection through the internal regions of the animal. Because of the extensive immunological nature associated with the mucosal immune system, the region is also effectively targeted for vaccine administration to elicit prophylactic immune responsiveness against several pathogens.33‒37
The local immune response is most effectively induced in response to direct application of antigens to the mucosal surface. The site-specific presentation of antigens in native conformation, however, presents a challenge in terms of the available administrative carriers and the toxicity associated with the antigens. Therefore, local immunity may be induced via the stimulation of the MALT, an integrated network of immunologically active mucosal tissue. Exposure by antigen at the mucosal surfaces of the gut or lung triggers the complex migration of lymphocytes to all mucosal regions where production of antigenic-responsive antibodies are produced. Antigenic exposure further induces a population of memory lymphocytes, which serve to generate antibodies in response to subsequent exposure by the same antigen. Hence, the MALT presents as a target for effective vaccination, by triggering the local immune response (Ibid.).
Of the immunologically active mucosal surfaces, the MALT of the GALT in the intestines presents with the greatest accumulation of lymphoid tissue. As the GALT contains populations of functional T and B lymphocytes in conjunction with antigen-presenting accessory cells, the region is one of significant immunological surveillance and respectively reactive to antigen. Specifically, the B lymphocyte population of the GALT comprises a significant subset of cells committed to the generation of the immunoglobulin A (IgA) class of antibodies. As a neutralizing antibody, the localized availability of IgA of the GALT provides an effective means of preventing invading pathogens from attaching to, and penetrating the epithelial layer of the mucosal surfaces. This form of immune responsiveness differs from that of the systemic lymphoid tissues where the IgA antibody class is not effectively induced through the conventional intramuscular (IM) or subcutaneous (SubQ) methods of immunization administration (Ibid.).
An epithelial cell layer of the GALT separates the underlying lymphoid layer of the mucosa from the lumen of the gut. Interspersed within the epithelial layer are accessory cells with a committed function for antigen presentation. Such accessory cells actively sample luminal antigenic samples, internalizing the samples for processing and presentation to the adjacent lymphoid cells. Presentation and exposure of antigen to the GALT initiates the clonal expansion of antigen-specific B and T lymphocytes. The IgA-committed B lymphoblasts consequently migrate through the roesenteric lymph nodes, by means to trigger an enhanced specific immune response in all mucosal sites, including the intestinal tract, nasopharyngeal and respiratory tracts, lung, oral cavity, ocular regions, mammary gland, and genitourinary tract. Therefore, immuno-stimulation of the GALT through effective oral vaccination may consequently result in the prevention of infectious diseases at a variety of mucosal surfaces (Ibid.).
Successful and efficacious orally administered vaccines require stability of the antigen as it passes through the digestive tract. As functional proteins dependent upon conformational structure, the configuration of antigens can be denatured under the digestive process of the gut. Enteric drug-delivery systems have been developed to protect the pharmaceutically active compounds for passage to the intestine. Recent enteric formulations have been developed that offer stabilized presentation of biologics and antigens for passing through to the gut.38‒40 Moreover, such methods can be translated for whole-cell stabilization and encapsulation and serve to maintain cellular enzymatic reaction potential for stabilizing the whole-cell antigen for effective and specific presentation to the GALT, and often with application to other regions of MALT.
Bacterial-based whole-cell antigenic carriers offer advantages of expressing proteins that enhance the immunogenicity of the core expressed vaccine antigen. The natural adjuvantization affords the use of the whole-cell as the modulator of the immune system without the employment of chemical adjuvants in complex with the antigen. Considering that the GALT elicits a natural response to whole-cell organisms as potential pathogenic threats, their use as immunogenic enhancers of expressed antigen can further exploit this phenomenon.
Concomitantly, vaccine stability is extended through optimized drying procedures to include logistics stability considerations for enhanced shelf life.39 Stability is further a factor for stable distribution logistics under environmental conditions where temperature can impart constraints upon vaccine administration scheduling, post-production.
Ultimately, when translating a technology from the research and development stage to the commercial application, production and cost considerations must be fully explored. For an RTV such as that presented here, the flexibility of the administration platform facilitates a multi-species application process; given the implementation of a feasible stabilization technology platform, various antigens may then be considered for enrollment in RTV programs as control for other diseases of zoonotic origin. To this end, a production process that is both scalable and cost efficient allows for rapid deployment of an effective RTV program, which can be governed by seasonal distribution metrics.
Finally, presented here is a mass-scale, orally-administered vaccine platform technology for potential application in the control of zoonotic infectious diseases. In addition to Lyme borreliosis, the platform is currently under consideration for translation and optimization for the presentation of vaccine antigens for other tick-borne diseases, such as anaplasmosis , babesiosis, and ehrlichiosis. In addition, it is further conceivable that the platform can be applied to control the prevalence of vector-borne disease in livestock or other companion animals. Clinical studies are presently underway testing the efficacy and safety of the platform for a host of livestock disease applications, and the molecular engineering of the platform continues to optimize the technology to accommodate the stable presentation of a host of protein antigens that elicit immunoresponsiveness in targeted host animals.
The successful implementation of an RTV distribution program clearly presents a biologistics solution to address a concerning and growing market for the control of zoonotic infectious diseases. This concept represents another tool in the toolbox for use in an integrated approach to control tick-borne and other vector-borne diseases.41 The RTV-based platform presented here further offers the flexibility for application to address multiple diseases of zoonoses and those communicable at the human-animal ecosystems interface.42
The author wishes to extend his sincere appreciation to Dr. Kirby Stafford of the Connecticut Agricultural Experiment Station (CAES) for his insightful review of this article.
S. Z. has relevant pending patents (assignee, US Biologic, Inc.) that might pose a conflict of interest. S. Z. is a shareholder in US Biologic, Inc.
None.

©2016 Zatechka. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.