International Journal of
eISSN: 2573-2889


Research Article Volume 2 Issue 5
1Department of Biochemistry, Ahmadu Bello University, Nigeria
2Department of Applied Biochemistry, Nnamdi Azikiwe University, Nigeria
3Department of Biochemistry, University of agriculture, Nigeria
Correspondence: Sanusi Babangida, Department of Biochemistry, Ahmadu Bello University, Zaria, Nigeria, Tel (+)2347065655363
Received: September 20, 2017 | Published: November 28, 2017
Citation: Babangida S, Idowu A, Muhammad A, et al. Genotypic variations may elucidate resistance to Plasmodium falciparum infection In vitro. Int J Mol Biol Open Access. 2017;2(5):149-152. DOI: 10.15406/ijmboa.2017.02.00035
Plasmodium falciparum malaria remains a significant cause of human suffering, and most malaria-related morbidity and mortality occurs in children living in sub-Sahara Africa. Evolutionary pressure has explained that various erythrocyte polymorphisms could protect against severe complications and death from Plasmodium falciparum malaria. Several mechanisms have been proposed to explain the protection of hemoglobin AS and SS from severe Plasmodium falciparum malaria. Sickle trait; the heterozygous and homozygous state of normal hemoglobin A (HbA) could confer protection against malaria in Africa. In the present study, we cultured Plasmodium falciparum infected red blood cells from AA, AS and SS for six days. During the six days period, the level of parasite load (Parasitemia) and the activity of arginase released by the parasite were monitored on daily basis. Result obtained shows a significant (P<0.05) increase in both the level of parasite load and the activity of arginase. This increase was found to be higher in AA genotype while lower in both SS and AS, but with AS been much lower. The mechanisms by which sickle trait confer such malaria protection might be as a result of change in structural conformation that alter with the parasite ability to invade into the cells through the membrane protein receptors and hence a decrease in its activity in both AS and SS respectively.
Keywords: genotype, infection, Plasmodium falciparum, arginase, parasitemia
Malaria is still among the most important public health problems with about 3billion at risk and 781,000 estimated deaths annually.1 Human malaria is caused majorly by two species of parasites: Plasmodium falciparum and Plasmodium vivax.1 Plasmodium falciparum malaria is more often with high morbidity and mortality rate among children living in sub-Saharan Africa.2 In highly endemic regions, the majority of children with malaria present with a mild form with only a small percentage often develop severe disease and subsequently death.3This is could be due to resistance developed over several years of exposure.3 While children susceptible to severe malaria die before reproductive age, therefore, genes that confer resistance to severe disease should be in the population living in sub-Saharan Africa.3,4 Red blood cell polymorphisms, such as hemoglobin S (HbS), α-thalassemia, β-thalassemia, glucose-6-phosphate dehydrogenase (G6PD) deficiency, hemoglobin E, and ovalocytosis shows some level of protections against severe P. Falciparum malaria because epidemiological evidence shows a relationship between areas highly endemic to malaria and distribution of these polymorphisms.1,5
Haemoglobinopathies are group of genetic deficiencies which result in synthesis of defective haemoglobin such as haemoglobin S.6 High prevalence (20 to 25%) of hemoglobin S (HBS) is known to be found in Africa and may reach up to 40% in some regions.7 Several studies suggested this polymorphism to confer some protection against severe form of malaria.7,8 The sickle cell trait (HBAS) comes as a result of a valine substitution with glutamic acid at position 6 of the hemoglobin chain.9 In mostly children, HBAS provides carriers with protection against severe Plasmodium falciparum malaria and this can explains the relatively high penetrance of this mutation in some areas reaching 30% in areas endemic to P. Falciparum infections.8,10,11
In this study, we attempted to reconcile some molecular mechanism proposed to explain this protections12,13 through comparative study on survival of P. Falciparum in some selected and most common red blood cell polymorphism. Since, the pathogenic course of Plasmodium falciparum infection is characterized with some molecular changes which include hypoglycemia, lactic acidosis, hemolytic anemia, hemoglobinuria, and hypoargininemia which all depend on the level of parasite load.14 In this study, we monitored the changes of parasite load and the level of arginase activities. The depletion of L-arginine is achieved by the help of arginase from the malarial parasite, which catalyzes the hydrolysis of the side chain guanidinium group to form L-ornithine and urea.15 Decrease in the level of L-arginine correlate with decreased level of immunity and nitric oxide (NO) production,14,16 thus favors parasite proliferations.17,18 In our work, we examine the activity of this enzyme in cultured blood cells from all the three most common genotypes alongside parasite proliferation.
Materials and reagents
RPMI 1640 media (Sigma Aldrich, Germany), Giemsa stain (Sigma Aldrich, Germany), Ninhydrin (sigma Aldrich, Germany), L-arginine (Sigma Aldrich, Germany).
Sample collection
Plasmodium falciparum infected and an uninfected blood sample (AA, AS, SS) from healthy and infected patient was obtained from Ahmadu Bello University Sickbay, Zaria. The sample was collected with the help of certified medical personnel.
Media preparation
O+ blood was collected, centrifuged at 10000 rpm for 20min at 4˚C. Serum was kept in aliquots and inactivated by keeping at 56˚C water bath (30min). 10ml inactivated O+ human serum was added to 90ml of incomplete media (96 ml of stock RPMI 1640 media containing 4ml of 5% sodium carbonate solution) making 100ml of working media.
Preparation of red blood cells and culture
O+ blood was collected, centrifuge (1500rpm for 10min) at room temperature. Plasma and buffy coat layer was removed and this washing process was repeated 3times in an incomplete media. Equal amount of complete media was added to the washed red blood cells and stored at 4˚C in refrigerator.
Infected red blood cell culture
50% suspension of infected cells(Plasmodium falciparum) in complete media (containing 15% serum) was added to equal amount of prepared uninfected O+ red blood cells to obtained an initial parasitemia ranged from 0.5 to 1.0%. The mixture was diluted with complete media to get 5% cell suspension (5% hematocrit). Culture was kept in CO2 incubator at 37˚C in Mary Hallaway Teaching Laboratories (MHTL).
Estimation of parasite load
After every 24 hours a portion of the cultured cells was collected (by removing the media without disturbing the cells) and a thin smear was prepared using Giemsa stain for examination of parasite load under light microscope. Fresh complete media (containing 10% serum) was added, mixed properly, and kept in the incubator. And % parasite load was counted using the formula below and erythrocyte containing ≥2 parasite is still counted as one infected erythrocyte.
<
Estimation of arginase activity
This is based on the facts that arginase act on it substrate (arginine) to produce ornithine and urea, under boiling temperature. A portion of the cultured cells was used to determine the activity of arginase spectrophotometrically by measuring the formation of L-Ornithine at 515nm with ninhydrin.
Statistical analysis
The data was analyzed using one way ANOVA followed with a multiple comparison test (Bonferroni) using statistical package for social science (SPSS) and the result was presented as mean ± standard deviation.
Progressive increased parasite load in P. Falciparum infected cells
There was a significant (P<0.05) increase in the level of parasite load in all the genotypes (AA, AS, SS) from day 1 to day 5 of the experiment period. This increase was observed to be higher in AA followed by SS and very slower in AS genotype (Figure 1-4). This could simply explain the ease at which the parasite proliferate in AA genotype and its ability to further invade into the uninfected cells present in the culture medium.

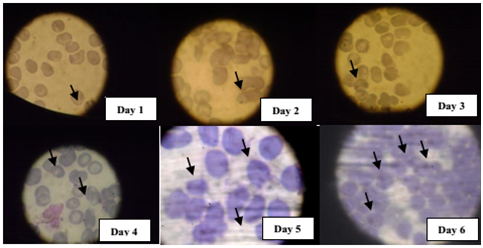

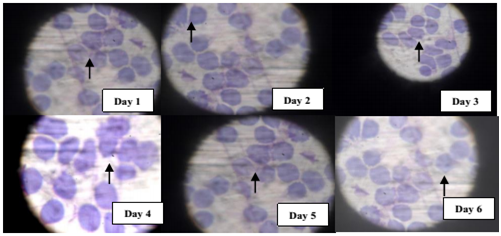
Progressive increased arginase activity in P. Falciparum infected cells
The activity of arginase was also found to increase with increasing parasite load and in accordance to the previous order with AA, SS and AS respectively. This could also explain in addition, the competitive advantage by the parasite to source more of its nutrient requirement for survival and further weakens the immune power by suppressing the activity of nitric oxide synthase (Figure 5).
The estimated parasitemia level shows a significant (P<0.05) increase as increase in number of days but these increment varies with as in most common blood genotype (AA, SS, AS). SS and AS genotype resistance to plasmodium infection was predicted to be as a result of distortion in the membrane of the cells, as a result the morphology of the binding receptors on the surface of red blood cells membrane, therefore cannot be recognize by Plasmodium falciparum binding ligand (pfbl).1 This mechanism help to reduce level of plasmodium infection in heterozygous and homozygous sickle cell allele.10
Arginase released by Plasmodium falciparum depend on level of parasitemia in the blood, thus causes an increased in haemoglobin degradation and release of various amino acid for parasite metabolism.14 Significant (P<0.05) increase in the activity of arginase in this study signify low levels of L-arginine and this correlate with decreased immunity and nitric oxide (NO) production.10 The depletion of L-arginine in culture is achieved by an arginase from the malaria parasite, which catalyzes the hydrolysis of the side chain guanidinium group to form L-ornithine and urea.19 Increased arginase activity characterizes the alternative immune response, which down-regulates inflammation and tissue damage while up-regulating angiogenesis and tissue repair mechanisms.14 Such conditions favor parasite growth through suppressed T cell and inflammatory responses to pathogens and increased concentrations of L-ornithine-derived polyamines, which facilitate cellular proliferation.1 Strong positive correlation between arginase activity and increase parasite proliferation demonstrate adaptive mechanism for parasite survival.
The present study demonstrate Plasmodium falciparum infection to confers a genetic based restrictive survival based on changes in parasite count and arginase release monitored over the duration of this experiments. Therefore, changes in genetic makeup can alter with morphological orientation of some vital structures that play a crucial role in the survival of the organism. And since malaria parasite (Plasmodium falciparum) releases arginase into the system in order to sustain it survival, development of specific arginase inhibitors can be a useful adjuvants in the treatment of malarial infections.
Ethical permission for the study was granted by Ahmadu Bello University teaching hospital (ABUZ). Individual written informed consent was provided by all study participants or their parents.
None.
Author declares that there is no conflict of interest.

©2017 Babangida, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.