International Journal of
eISSN: 2573-2838


Mini Review Volume 3 Issue 1
1Chemistry Department, King Abdulaziz University, Saudi Arabia
2Chemistry Department, Polymer Chemistry Lab 122, Egypt
Correspondence: Mahmoud A Hussein, Chemistry Department, Faculty of Science, King Abdulaziz University, PO Box 80203, Jeddah 21589, Saudi Arabia
Received: July 28, 2017 | Published: September 7, 2017
Citation: Hajeeassa KS, Hussein MA, Al-amshany ZM. The influence of graphene nano-particles on the biological interest of polyvinyl alcohol nanocomposites. Int J Biosen Bioelectron. 2017;3(1):230–232. DOI: 10.15406/ijbsbe.2017.03.00054
Recently, the development of Nano-material has become the most interest issues for many researchers. For example, nanocarbons such as graphene (Gr) and graphene derivatives attracted more attention due to their better structural, high strength, low density, and good combability with polymer. Introduction these nanocarbon to the polymer matrix like polyvinyl alcohol (PVA) improved the properties of the polymer for instance: thermal, mechanical, and electrical properties. This led to its accessibility to many applications. This review, present recent studies of preparation PVA nanocomposites with Gr as filler and also illiterate the improvement of properties for further use.
Keywords: nano-material, polyvinyl alcohol, graphene, polymer nanocomposite
Nanocomposite is a multiphase material that made from a combination of two or more of constituent material (matrix and reinforcing phase) with different physical or chemical properties that, when they combined, the resulting material has characteristics differently from the individual compounds.1 Over the past decades, nanocomposites become a magnificent issue in our lives. Materials play the key role in every area of technology of the 21st century. It started to be used in medicals, industrials, computing, and sensor applications to make our lives more comfortable.2,3 One kind of nanocomposites according to the matrix is polymer nanocomposite. Polymer nanocomposites are a combination of polymer or copolymer and Nano-sized fillers (less than 5 wt%). Addition of nanofiller to the polymer matrix improve properties and can use in different application.4
Polyvinyl alcohol (PVA) is the most important polymer which is highly biocompatible, highly water-soluble, nontoxic, and synthetic polymer. PVA, in addition has been used widely in biological applications and also sensitive for water and humidity.5 PVA can be used as a matrix for fabricate polymer nanocomposites and use it in huge application.6 Nanocarbons are considered as carbon-based materials which can be bonded at the molecular level by different ways to improve unique properties.7 Carbon nanofillers like graphene and carbon nanotube plays an important role because of their great structure and efficacious properties e.g. thermal, electrical, and mechanical properties etc.8,9 Graphene (Gr) is a single layer two-dimensional (2D) carbon atoms arranged in a honeycomb pattern, has been invited particularly by technologists and chemists’ researchers due to unique properties like thermal, electrical, and mechanical properties.10,11. In this review, we focus and highlight the enhancement of properties and show some application of PVA with graphene as filler.
Graphene nanoparticles (GNP)
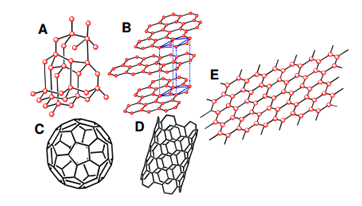
Carbon has been known to form many allotropes because of its ability to exist in different hybridizations. Different allotropes of carbon exist in nature such as graphite, diamond, fullerenes and amorphous carbon. Figure 1 shows the structures of some of the carbonaceous materials existing in nature.12 Among these, graphene is the building block of carbonaceous materials existing in nature with unique properties principally attributed to carbon atoms arranged into two-dimensional (2D) planer monolayer of sp2-hybridized orbitals with honeycomb (hexagonal) lattice structure.13
Properties of graphene nanoparticles
Graphene Nanoparticles exhibits a high level of electrical conductivity (7200Sm-1) due to the unique nanostructures; also the electron and hole in graphene sheets have extremely high carrier mobility behave like massless relativistic particles at room temperature (10,000–15,000cm2V-1S-1). Pristine graphene is highly thermally conductive (~5000W m-1K-1), with superior mechanical properties with a high Young’s modulus (~1.0TPa), these properties along with the large specific area (2630m2g-1).15,16 Two carbon atoms are bonded together with a σ C–C chemical bond which is an extremely strong bond in materials due to hybridized orbitals generated by the superposition of 2s with 2px and 2py orbitals. The planar orbitals form the energetically stable and localized σ-bonds with the three nearest-neighbor carbon atoms in the honeycomb lattice, and they are responsible for most of the binding energy and for the elastic properties of the graphene sheet. The remaining free 2pz orbitals perpendicularly oriented to the molecular plane hybridizes to form the conduction (π) and valence (π*) bands, which is responsible of the electron conduction of graphene15,17 pure graphene sheets are highly hydrophobic(non-dispersible in aqueous solution) in nature and easily agglomerates because of the strong π - π interactions along high surface area of graphene.12,18
Polyvinyl alcohol/graphene nanocomposites
Nanocomposite based of PVA and Gr have been synthesized and analyzed by many researchers to improve material’s properties. The strong interaction between PVA and Gr and because of this it led to use in different application.19 As an example of PVA with Gr as filler, Rade S et al. 2015, prepared PVA/Gr by simple dissolution technique. The introduction of Gr to PVA improves mechanical and thermal properties comparing with neat PVA. In this study, PVA/Gr nanocomposite was nontoxic for peripheral bloods nanomolecular cells (PBMC) and exhibited a strong antibacterial activity against E. coli (gram negative bacteria) and S. aureus (gram positive bacteria) that made it as an excellent candidate biomedical application (6) presented in Figure 2. Other study by Yuang-Cheng C et al.,20 also showed the different addition of Gr from 1%-10% improved the antibacterial activity against E. coli by 92%-97.1% and 92.3%-99.7% against S. aureus.20 In order to improve the biological application, PVA/Gr/AgNPs nanocomposites hydrogel synthesized by Abudabbus M et al.,21 silver nanoparticles (AgNPs) in this study used as agent for dressing antimicrobial material. The results showed the lowest cytotoxicity to PBMC culture and good inhibition for bacteria.21 Moreover, different types of nanofiller also cooperated with PVA. Recent study by Adel U et al.,22 PVA/graphene oxide/starch/silver in form (PVA/GO/starch/Ag) nanocomposites prepared by simple solvent method. The dimeter of zoon inhibition increased for the prepared nanocomposite against E. coli 17mm and S. aureus 20.5mm as illustrated in Figure 3, while pure PVA did not show any antibacterial response.22
One of the magnificent accomplishment in nanoscience and nanotechnology is polymer nanocomposites. The development of polymer nanocomposites has massive impact on biological research recently. This review considers brief summary the recent studies of PVA/Graphene nanocomposites. The study also demonstrates the improvement effect on some types of bacteria.
None.
The author declares no conflict of interest.

©2017 Hajeeassa, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.