International Journal of
eISSN: 2573-2838


Review Article Volume 2 Issue 5
1National Laboratory of Nanotechnology LANOTEC, Costa Rica
2National University of Costa Rica UNA, Costa Rica
3Invenio University, Costa Rica
Correspondence: Jose Roberto Vega Baudrit, National Laboratory of Nanotechnology LANOTEC, LANOTEC CENAT CONARE, San Jos, Costa Rica
Received: December 21, 2016 | Published: May 23, 2017
Citation: Gutierrez B, Bermúdez CV, Ureña YRC, et al. Nanobots: development and future. Int J Biosen Bioelectron. 2017;2(5):146–151. DOI: 10.15406/ijbsbe.2017.02.00037
Some decades ago, nanoscale machines called “nanorobots” (better known as “nanobots”) were only in science fiction. Today, they are expected to be the next generation of nanodevices and to change the technology related to medical diagnosis and drug delivery. Many challenges have to be faced to develop this technology; not only from the technical, biological and physicochemical point of view but also concerning the risks of using new materials and devices at nanoscale and their interaction with the environment and human beings. The aim of this review is to describe the nanobots, the technology and advances and in more detail the applications related to medicine.
Keywords: nanobots, nanomotors, nanomedicine, nanosensors
NEMS, nano electro mechanical system; AFM, atomic force microscopy; CPE, carbon paste electrodes; ATPase, adenosine triphosphate synthase; AFP, alpha-fetoprotein; CEA, carcinoembryonic antigen
The first scientist to mention the term “nanobots” was the physicist Richard Feyman in 1959; when he gave his popular talk named “There’s Plenty of Room at the Bottom. He commented about nanodips and nanobots for curing heart diseases.1,2 Later, the scientist Eric Drexler, inspired by the talk, published his book “Engines of Creation”, where genetically programmed molecular machines were mentioned as upcoming technologies in cellular biology.3 The first study related to nanobots was made by Robert Freitas.4 It was related to medical nanobots called respirocytes; resembling red blood cells.4 Nanobots could be defined as a controllable nanoscale machine composed of a sensor and a motor, capable by performing specific tasks.5 These are not comparable to a drone, instead are more similar to a complex piece of fabric.6 Robert Wood defined them as devices that detect friends or enemies; undergoing through a conformational change when they sense an enemy, catalyzing the release of a substance that can act against it.6
Nanobots properties
Nanobots can be produced using organic materials such as proteins and polynucleotides, or inorganic materials such as metals or diamond.1,7 In the case of diamond, this stands out for its high strength and high performance. Metals could have double purposes, for example silver. It can be the base of a nanobot and to have an antibacterial effect.8–10 In some cases, they can act as a virus causing irreversible cell damage.5 The surface properties of the nanobots are a key factor to define the solubility and interactions with other macromolecules or cell surfaces. Size or shape of a nanobot will affect directly their motion, permeabilization and reactivity.11 Different extracellular nanostructures could be used as model. Ga et al.12 used spiral water conduction vessels of plants coated with thin Ti and Ni layers, achieving efficient propulsion in biological media.12 Depending on the fuel used, the propulsion mechanism can be biocompatible or not.13,14 Further information about propulsion mechanisms will be presented in section 4.1.
Parts
One of the main interests for the research of nanobots in medicine has been to develop treatments targeting the exact point where it is needed, with the idea of minimizing the impact on healthy parts of the body that any regular treatment has.15 This idea suggests having nanobots designed to detect and mobilize to a determined part of the body where the problem is located and, in the best scenario, send feedback. Because of these determinant tasks (detect and mobilize), two devices can be identified as essential: sensors and propulsion equipment; and from this information it can be deduced some other devices will be needed as well, like power supplies and molecular computers, without excluding devices to develop a specific task like storage compartments or manipulators.2
Sensors
Sensors are one of the most important parts in nanobots. Mechanical, thermal, optical, magnetic, chemical and biological sensors have been tested in nanobots applications.14 Any sensor that uses a nanoscale phenomenon for its operation is classified as a nanosensor.14 On the organic part, biosensors utilize biological reactions for detecting target analytes,16 and considering the need to accomplish the target treatments goals of nanobots in medicine, this type of sensors are the most evident devices to explore in the field of nanorobotics. A clear example of this type of sensors is the utilization of nano cantilevers as a Nano Electro Mechanical System (NEMS). This system utilizes biological material that will be attached by itself to a coated cantilever, causing fundamental changes in mass or its surface tension.17
However, in general terms, sensors provide two functions to the surface, detecting the presence of the target molecules and indirectly know the amount of damage that exists from the change in the functional properties of nanobot. Therefore, many types of sensors were developed in function of the target molecules that are wanted to be detected, in addition, it has been demonstrated that they possess a high and fast analysis of time.18 The cantilevers are characterized by their high sensitivity in providing a good atomic resolution in the image of a certain surface, so they are commonly used in atomic force microscopy (AFM). Currently, utilizing the AFM as an effector, nano-cantilevers are being developed as sensors to be used in nanobots. The main advantage is real-time detection, directly and quickly. In addition, they are able to measure cell mass, biomolecules, nucleic acids and others, as well as detecting specific molecules or even manipulate and place nanoobjects in a predefined arrangement.19–21 In general, it can be used to manipulate objects at a single-molecule level.22
Cantilevers can operate in two different methods: dynamic mode and static mode. In the first case, the cantilevers respond to the biochemical interactions of the surface through a change in the resonance frequency, which is caused by the mass or rigidity of the target molecule. This way, the frequency is used to analyze the topography of the surface, and detect the different molecular bonds. On the other hand, working in static mode relies on the absorption of determined analytes from a nonmoving cantilever, which causes differential surface stress (bending) and consequently, deflection regarding the reference point.20,23 This differential surface stress defines the relationship between the sensor and the analyte.23 These cantilever’s attributes can be utilized to detect the presence of target biomolecules in a small volume of sample, which in medicine can imply an early detection of diseases, such as cancer.24
Another type of nanosensors are the carbon paste electrodes (CPE). They are made from graphite and a pasting liquid, and are used for voltammetric measurements and even in coulometry, as a renewable surface for electron transfer reactions. There are some advantages that make CPE attractive for nanomedicine: they can be miniaturized, easy to fabricate, good electrical and mechanical resistance, low cost, among others. Therefore, these nanosensors are being used to detect nucleic acids since adenine and cytosine have been shown to provide redox signals, while guanine gives an oxidative signal.26 Also, the thymine together with the other nitrogenous bases already mentioned, has given good signals in carbon electrodes. This is supported by a calibration curve obtained with very good linearity detecting concentrations from 1ng/ml to 100ng/ml.27
Propulsion equipment
Propulsion is in charge of the movement of nanobots, and this is the reason why many different motors and propulsion equipment in general have been designed. Nanomotors can be defined as nanoscale devices with their own propulsion, obtaining the energy by chemical reactions of the medium, electricity, magnetic or acoustic fields.28,29 Even so, nanotechnology presents great challenges to control movement on this scale; the main ones are due to the viscosity and Brownian motion. It should be noted that Brownian motion corresponds to the random motion of particles caused by the thermal collisions between the molecules of the solvent and the colloidal particles.30 In other words, the main problem is having a nanobot with sufficient energy to overcome the properties of a fluid in nanoscale and achieve movement.31
There are two approaches being utilized to facilitate the movement of the nanobots: external and internal. In the external approach for propulsion, MRI devices became the leading option after some predecessor studies in the eighties using magnets and super conducing-magnets.15 There are several advantages of this system: the nanobots speed and direction are controlled from an external computer, decreasing risk considerably,32 the MRI can be used to get real-time feedback of the behavior of the nanobots, having all the power depending on the external machine, there is no need to develop complicated and power consuming nanomotors, among others. However, with respect to the internal approach, the best models, as is to be expected, are the biomotors that already exist in nature; Therefore, it is a field in continuous development to find and to understand new mechanisms of movement. Major examples include cell division, protein synthesis, DNA transcription, among others; all of the above are cases in which chemical energy becomes movement.31
A hybrid engine was obtained in the year 2000 by Bachand et al.33 This invention marked the beginning of bionanomotors and had interesting results. They integrated nano-electro-mechanical devices with adenosine triphosphate synthase (ATPase) like nanomotor. For this purpose, individual F1-ATPase molecules, corresponding to a structural domain of the molecule, were used and placed in nickel matrices by means of electron beam lithography. After conducting a test, the performance of the motors was evaluated by these molecules, obtaining three stages of rotation by part of the γ subunit during the hydrolysis of ATP. Further, during this hydrolysis, the γ subunit of the ATPase was rotated at a rotation speed of about 3-4 rps. Experiments have shown that the nanohybrid structure can be fed with a biological nanomotor.33 Several authors describe the fabrication of nanospherical motors driven by catalytic reaction of H2O2.31,34,35 One of them is the Janus motor, formed by Janus particles, that is, nanoparticles that on their surface have two or more sides with different properties.36 The mechanism of this motor consists of a chemical reaction on only one side of the Janus particles, which produces the force for the movement of these motors. There are different types, one of these motors is characterized by having a catalytic Pt layer that catalyzes the H2O2 decomposition reaction.34 For example, there is a solid silica based Janus spherical motor, which has a catalytic layer of Pt for H2O2 decomposition. Another example of this propulsion is an Au/Pt bimetallic nanomotor.35 However, other propulsion mechanisms were suggested, for example: a nano sphere with a magnetic hemisphere or propulsion by osmosis.31,37 For the sphere based propulsion, nano spheres are created with two different hemispheres. This allows one-half to be coated with a catalyst that causes an asymmetrical distribution of a reaction product, which moves the spheres along.31 Meanwhile, osmotic propulsion is based on the artificial version of the reaction generated from the release of a semi permeable membrane after it separates a fluid containing colloidal particles, because of a flow that was induced from the low to the high particle concentration side. The pressure stopping the flow is the osmotic pressure. The osmotic pressure difference between the two sides will cause it to move until equilibrium is established. In this way the microscopic kinetic energy of the particles is transformed into macroscopic mechanical motion and work.37
In addition, tubular nanomotors made by Pt and two other layers of In GaAs and GaAs, can also use H2O2 as fuel. Initially the fuel contact with the catalytic material produces O2 bubbles and when the bubbles come out of inside they produce the propulsion.31 Nanowires motor has also been developed in recent years, a flexible fuel-free nanowire motor is being manufactured with three segments: Ni-Ag-Au. They are even able to achieve a speed similar to microorganism and this make them promising for future biomedical application.38 Gold-nano wired ultrasound-driven motors are being developed for their utilization as drug delivery devices in cancer cells. These motors are based on the nanoporous gold segment for increasing superficial area and hence the loading capacity of the drug. Improved versions of these devices combine ultrasound propulsion with magnetic guidance and an infrared light as a trigger to release the load.39 Although the sensor and motor are principal parts, nanobots may have other important components. Nanocomputers can be electronic, biochemical, organic or quantum and have the function of controlling or directing nanobots inside the body. For example, the development of a computer at molecular level made up of DNA, which can regulate gene expression. It has a soft ware coded with the four letters of the DNA nitrogenous bases. In addition, it was programmed to detect the type of mRNA associated with specific genes that in case of being over expressed or its opposite induce the cancer. This allows diagnosing different types of cancer and counter acting the disease with the indicated drug.40 On the other hand, nanotweezers and nanochips also have important roles in nanobots, such as nanomachines movement within the body or its control.41
Uses in medicine
In near future, nanobots will play an important role in medicine, like a “nanodoctor” inside the body. The cancer treatment has a special interest because is one of the leading causes of death in the world.42 The nanobot will move independently around the body and detect a cancer cell, Figure 1. Therefore, when are inside or interacting with the cancer cells they will release the drug. In this way, the side effects of anti-cancer drugs on other parts of the body will be reduced.43 In addition to the cancer drug attack, some nanoscale robots can destroy cancer cells through thermal necrosis when a non-invasive external source of heating or stimulus will be applied. As example, super magnetic beads that can penetrate and destroy cancerous tissues with a magnetic field.44
For atherosclerosis and other cardiovascular diseases such as ischemic or arrhythmias, nanobots will be injected into the circulatory system by intravenous infusion techniques, for acting against fatty deposits. In addition, nanobots are also a proposal for diagnosis and treatment of demyelination, since they can reach the layer of myelin in the nerves. Also, specific nanobots that detect Alzheimer's disease based on the amyloid protein β deposits.45 Dentifrobots are especial nanobots suggested for use in odontology. They could be a complement of the mouthwash or toothpaste and basically, their functions will consist in the metabolization of the trapped material and in the identification and destruction of pathogenic bacteria inside of the mouth.46 Nanobots can also be used to travel through the patient's gingiva, and thus they will deliver the analgesic to the target site.20 It is expected surgeries at nano level in the retina and surrounding membranes and pregnant women.47,48 Also, in gene therapy comparing two DNA chains; sensing proteins inside the cell and replacing chromosomes.47 The applications of nanobots in medicine are many and diverse.
An example of biomimetic derivated nanobots are the microbivores; similar to white blood cells in the bloodstream. They are thought to bind to a specific target surface site, and next to destroy the pathogen. As a result, amino acid residues, fatty acids, simple sugars are released, among others.1 A second example of biomimetic nanobots are the respirocytes. They are a kind of artificial red blood cell. They will have the capacity of pumping 3 million molecules of O2 and CO2, which represents a delivery of oxygen to tissues per unit volume 200 times higher than natural red blood cell. The respirocytes are planned to treat anemia and lung problems.1 Other example are the biobots, they are genetically modified bacteria. Its genetic material will be removed and replaced by an artificially synthesized DNA. The function of these biological robots is to supply or collaborate in the patient's metabolic needs, i.e. in the production of hormones, vitamins, enzymes, and also in the absorption of toxins. It is important to mention that for this case, in 2003 the first patent related to chemical synthesis of gene and gene networks was published.1,25
Patents and other applications in medicine
A patent related to nanowire motors fuel free was available since 2013.49 There are two designs of fuel-free nanomotors: magnetically and electrically-propelled.50 Another patent corresponds to a spherical nanomotor of 50 to 90 nm in size formed by mesoporous silica and a layer of platinum on the partial surface. The preparation method consists of five steps: synthesizing mesoporous silica nanoparticles, performing hydrophilic treatment to silicon chips, dripping silicon dioxide nanoparticle dispersion liquid onto the hydrophilic silicon chips, drying and sputtering platinum onto the silicon chips, and finally drying after ultrasonic vibration for shaking off.51
Actually, the application of nanobots in medicine is on the first steps. In 2012 a patent “Diagnostic-nanosensor and its use in medicine” was published”.52 Basically, the objective of the invention was to obtain a new nanosensor for diagnosis in medicine and others applications, based on metallic semiconductors and magnetic nanoislands. The surface of these nanoparticles can be modified to achieve interaction with the molecule of interest or recognize specific substrate surface as a cell membrane. In that way, the invention can be used for the detection of pathogens or toxins inside the body.46 There are available patents about nanosensors that could be used for nanobots based on AFM cantilevers and magnetic relaxation nanosensors.47,48
Specifically in nanobots, several recent patents were found. Fritsch et al.53 proposed the robo-lens. They will have the task of moving across the corneal surface lens and remove microorganisms and impurities, improving the vision of the person, hygiene and durability of the lens.53 The second patent is “Sensor molecular machine and controller added to programmable nano-robot” published in 2016. This invention has three parts: the sensor, the rotor and the controller. According to the patent “this is the first time sensing in a single nano-robot created by organic synthesis”. These nanobots are not only able to sense the environment; they can move, be effective even under high noise and establish communication with external devices. With a controller installed they can also make logical decisions, Figure 2.54
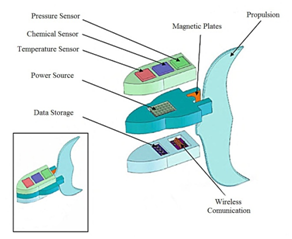
Figure 2 Components of nanobots, figure modified from Mavroids et al. 15.
Drugs used in cancer treatments like a bleomycin or mitomycin cause seriously side effects, for example: anemia, vomiting and others. So, in order to create more efficient cancer markers and achieve a better process, a new nanobot was developed and patented in 2016 by Zhang et al. This invention is composed of three different parts: a loading component, a power component and a connecting component. The objective of this invention is using the nanobot as a car to release a labeling reagent at the specific point where there is a tumor. The motor of the invention is propelled by ATP, the payload (labeling reagent) could be alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA) or other tumor-specific markers. It has an inorganic nanomaterial connecting component. When the nanobots arrive at the target point the connecting component will be broken, and then the tagging reagent flows out and mark the sick cells. In that way, the doctor can determine the distribution of cancer cells and recognize the disease status. In addition, the most effective treatment of the disease.54
Nanobots will have sensors for detecting levels of glucose and, if it is necessary, secrete insulin into the blood.50 This may be done through hSGLT3 for detecting glucose concentration on diabetic patients, this protein has into its functions regulating glucose levels.48 On the other hand, nanobots that destroy brain cancer cells were recently simulated by Loscrí et al.55 who called them Nano Bee.55 Nano Bees are able to detect cancer cells, destroy them, emit an acoustic signal when detected the tumor and detect the acoustic waves.37,38 Also, the nanodevices can be used in nerve system, specifically myelinated disorders. Many diseases can be generated as a result of the loss of myelin sheath; multiple sclerosis, transverse myelitis and chronic demyelinating polyneuropathy are some of them. So nanobots were considered being able to detect this demyelinating and eventually to improve treatment procedure.51
Risks
As expected, the nanoparticles involved in certain medical devices interact with both the environment and the human body. Currently, the number of innovations in the field of nanomedicine increases and it is necessary to develop protocols for evaluating the risks.11 The main risks to humans are in the adsorption of biomolecules or materials and oxidative stress, causing DNA damage. Furthermore, the inability to control the movement of these nanoparticles throughout the body creates a risk that can reach undesirable locations and thus lead to side effects. Moreover, as already mentioned, one of the main characteristics of nanoparticles is that they can cross biological barriers.56 This can be a disadvantage, the unnecessary cross of barriers may be a trigger for inflammatory reactions, At the same time, it is necessary to consider the whole manufacturing process, because for some nanodevices toxic reagents are used during this stage, which limit the applications that they can have in the diagnosis or the administration of drugs. The most common fuel H2O2 produces oxygen bubbles that activate a propulsion mechanism. However, the body tolerates only very low concentrations. In other words, even in concentrations less than 1% is highly toxic for cells, while at high concentrations can lead to protein denaturation. For this reason, the use of non-toxic fuels such as water or glucose is also suggested. Likewise, the ethical aspects involved in the diagnosis of diseases should be evaluated. For example, in a case where patients are known to be prone to disease, through gene analysis, one should consider: who will possess such information, how fundamental rights can be protected, how can responsible use of nanomedicine be promoted, etc.56 In addition, the environmental impact of nanoparticles must also be considered, since they remain dispersed in the environment from the moment they are discarded from the body, even from the laboratory stages. Unlike the risk to human level, the risk they represent for the environment is much more complex by the large number of interactions that are involved.11 On the other hand, the nanotechnology has risks and benefits; a balance should be done before using a technology.56
The nanobots are not an idea written on paper anymore, they are currently under development. The components are sensors, propulsion and navigation systems. Nowadays, the research is mainly focus on nanomotors; a key part of the propulsion component. Chemically, magnetic and acoustic driven nanomotors have been produced and applied; mostly in the field of nanomedicine. However, for medicine, a major effort should be done in technologies based on fuel free and biocompatible approaches. There is a lot of research, but it is going to be needed a lot more in order to create a functional nanobot, able to accomplish tasks beneficial for the human being; surface modifications, structures, components and body response has to be well understood. However, the quick development in nanotechnology, biotechnology and computing science will influence the prompt creation of nanobot. In this moment, we are still in the design and experimentation stage, even some patents are in the market.
None.
The author declares no conflict of interest.

©2017 Gutierrez, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.