eISSN: 2471-0016


Medical Relevance MR is the fundamental premise in Clinical Laboratories. Our mission is to provide reliable and timely information for decision making including diagnosis, prognosis, treatment and surveillance. In consequence, the necessity to establish adequate Analytical Goals AG for all the tests is evident, in order to guarantee that information provides maximum benefit with least risk and best cost, avoiding false positive and false negative results. Analytical Performance Specifications APS can be set using several strategies depending on three interdependent perspectives including Medical Relevance, Biological Variation and Technology. From technological perspective is important to acknowledge SIX SIGMA 6S which emerged in industry in 1979 to improve quality on manufacturing processes and achieve a level of only 3.4 Defects per Million Units Produced DPMUP. It comprises a whole system where importance is given to performance specifications in accordance to customer requirements, statistical measurements and technological status. To review and document SIX SIGMA 6S fundaments and tools and to assess applicability on Clinical Laboratories, comparing data with CLIA 2024 Performance Specifications.
Materials and methods: This is a revision of basic concepts and fundamental methods of SIX SIGMA 6S, including definitions, formulas and indicators of Analytical Variability. with the perspective of SIX SIGMA 6S on Analytical Goals AG according to Tonks and Aspen Analytical Coefficients of Variation ACV that have been used for decades in Clinical Laboratories as indicators of reliability, precision and accuracy. Calculation of Biological Variability BV in Laboratory Medicine depend primary on Reference Limits RL where Tonks criterion represents 25% of normal range and is equal to one Biological Standard Deviation. Aspen criterion is one half of Tonks level, thus representing 12.5% of normal range. To reach SIX SIGMA 6S level, it is necessary to improve Tonks level six times, which implies reducing Analytical Variability to 4.2% of Biological Variability. To evaluate relation between Biological Variability BV and Analytical Variability AV, it is recommended to calculate AV/BV ratio, which will result <1.0 according to Tonks, <0.50 according to Aspen and <0.17 when SIX SIGMA 6S level is achieved.
Discussion: Establishing attainable and challenging goals is first step in any AQCS Analytical Quality Control System. RCV Relative Coefficient of Variation calculation allows the integration of Medical Relevance, Biological Variation and Technology through a reliable and easy approach for any measurand with the only condition of having suitable reference limits for attended population and calculation of ACV Analytical Coefficient of Variation for each test. Continuous Quality Improvement CQI depends on understanding actual level in which laboratory is performing. In order to improve it is necessary to establish a formal WWWWH Strategic Plan, to enhance laboratory practices and to develop technology under Quality Management System QMS in order not to depend only on automation.
Keywords: clinical laboratories, biological variability, analytical performance specifications, quality health care
AG, analytical goals; AQC, analytical quality control; APS, analytical performance specifications; AV, analytical variability, BV, biological variability; CLIA, clinical Laboratory improvement amendments; CQI continuous quality improvement, CRV coefficient of relative variation; EBM, evidence based medicine; MR, medical relevance; PTR, proficiency testing requirements; QMS, quality management system; QHC, quality health care; RL, reference limits; 6S, six sigma
Medical Relevance is the fundamental premise in Clinical Laboratories. Our mission is to provide reliable and timely information for decision making including diagnosis, prognosis, treatment and surveillance. In order to guarantee that information provides maximum benefit with least risk and best cost, avoiding false positive and false negative results the necessity to establish adequate analytical goals for all the tests is evident, Analytical Performance Specifications APS also known as Analytical Goals AG can be set with several strategies depending on three interdependent perspectives including:
From technological perspective is important to acknowledge SIX SIGMA 6S which emerged in industry in 1979 to improve quality on manufacturing processes and achieve a level of only 3.4 Defects per Million Units Produced DPMUP. It comprises a whole system where importance is given to performance specifications in accordance to customer requirements, statistical measurements and technological status. SIX SIGMA 6S level is considered as an equivalent of "zero errors" it provides techniques and tools to measure and improve quality by reducing defects. It was developed by Motorola's Manufacturing Department in 1979, where millions of parts are made according to an identical process, it has been also been used on manufacturing process applying principles in design control.1 Recently, it has been proposed to implement SIX SIGMA 6S on many fields, including Clinical Laboratory and or Medical Services.2,3
Sigma is Greek letter used in statistics to represent Standard Deviation.4 Uncertainty is variable; increasing confidence interval reduces probability of error. Working on confidence interval of ±2 standard deviations means that level of certainty is >95.5% and level of error is <4.5%, while on a confidence interval of ±3 standard deviations quality is >99.999,7% and error is reduced to less than 0.000,3% (Table 1).
2 Sigma: ±2 SD ≥95.5% Defects ≤4.5 % DPMU ≤45,400 |
6 Sigma: ±3 SD ≥99.9997% Defects ≤ 0.000,34 % DPMU ≤3.4 |
SD, standard deviation DPMU, defect per million units |
Table 1 Sigma levels depend on defects per million units produced
Sigma Levels determine number of defects that occur. If a distribution is assumed to be Gaussian for variation of a process, area in tails of distribution can be used to estimate expected defects.
SIX SIGMA 6S aims perfection. It allows only 3.4 defects per million opportunities for each product or service transaction. It incorporates basic principles, statistics and reengineering and it relies primarily on experimental modification to reduce defects and increase quality while improves performance, reduces variation and maintains a consistent process, which in consequence improves productivity while delivers cost reduction.
SIX SIGMA 6S includes a variety of methods to detect, develop, and establish best practices on any process. Consultants and statisticians study several existing procedures and determine which method produce best results. Various combinations are tested on assumption that a given combination can improve process, and may be implemented until 99.9997% of all units produced are of excellent quality allowing 3.4 defects per million opportunities. If a given process cannot meet these criteria, it is reanalyzed, modified and tested to discover if there are any improvements. If none are found, process is reworked, system is modified, including structures and procedures, and tested again. This cycle is repeated until a significant improvement is found.
SIX SIGMA 6S implies visiting the best quality plant to find out why it performs better and to put into practice techniques learned. Key elements of SIX SIGMA 6S process improvement include clearly defining customer requirements, defining metrics and measures, improving design quality, with active employee participation, demonstrating continuous quality improvement. Company's Research & Development Department should use same techniques and learn from other departments. SIX SIGMA 6S is based on knowing how to share knowledge, through a procedure called Benchmarking, which is not really a SIX SIGMA 6S creation. It is based on principle that if a company's process unit produces a higher quantity and better quality, benchmarking team will be able to use it as a basis for improvement.
SIX SIGMA 6S applies five-phase approach developed by Motorola and referred to as DMAIM process:
Involving all employees is very important. The organization must provide opportunities and incentives to focus talent and capabilities on customer satisfaction. All team members must have a well-defined role, with documented and measurable objectives.
In United States of America, Clinical Laboratory represents less than 5% of cost of health services, with an impact of more than 70% in diagnosis. Recently, Institute of Medicine of same country published a study entitled "To Err is Human" focused on high rate of medical errors caused by health service providers which determine 50,000 to 100,000 preventable deaths each year. This study has led to considerable discussion in health care circles as to what to do about this problem.5
Several names have been used to identify Quality Programs. According to Westgard -an expert in field-, names have been changing in emphasis, but not in fundamentals. All programs have been developed in accordance to Theory for Quality Management, set forth by Deming, Juran, Shewart, Levey Jennings, Whitehead, and many others throughout the world.6–15
Achievement of SIX SIGMA 6S is a major challenge, so far there is no institution or Medical Laboratory that has been able to consistently achieve this level of quality, so it is important to understand that this is a unique opportunity to improve quality of services provided to patients and customers.
For Clinical Laboratory Professionals pursuit of quality is fundamental. The main challenge is reducing laboratory errors by striving for highest level of accuracy up to SIX SIGMA 6S, which is equivalent to a safety level of 99.9997% and represents a total of 3.4 defects per million units. What would it mean to reduce defects to less than 3.4 units per million and how can it be applied to clinical laboratory?
Taking as example a laboratory that works seven days a week where 100 patients are attended daily and 10 studies are reported per patient, reaching a SIX SIGMA 6S level would mean finding 0.1 defects per patient yearly and 1.1 defects per test yearly (Table 2).
Work load |
Level |
Tonks |
ASPEN |
Six sigma |
||||
Indicators |
Standard deviation |
1.0 Std D |
1/2 Std D |
1/6 Std D |
||||
100 patients per day |
Conformity % |
95.50% |
97.75% |
100.00% |
||||
10 tests per patient |
Defects % |
4.50% |
2.25% |
0.00% |
||||
Number |
Day |
Month |
Year |
Defects x day |
x month x year |
|||
Patients |
100 |
3000 |
36,500 |
4.5 |
2.3 |
0.0003 |
0.009 |
0.1 |
Tests |
1000 |
30,000 |
3,65,000 |
45 |
22.5 |
0.003 |
0.09 |
1.1 |
Table 2 Theoretical impact of reaching the six-sigma level in the clinical laboratory
Six sigma 6S in clinical laboratory
In Health Sector Including Clinical Laboratories SIX SIGMA 6S is a new standard. Although SIX SIGMA 6S didn't start in Clinical Labs it is a powerful tool that can be a useful approach that concentrates on prevention of problems through a focus on process analysis and application of statistical methods.4,9,10 Procedures used in laboratories can be renewed through different approaches. Method works on basis of a fundamental question: What is critical level of quality that we must deliver to customers? From there, a rigorous analysis is developed in each and every process to determine if it is delivering what our doctors and patients require.
AG: analytical goals
Selected coefficients of variation have evolved over more than fifty years depending on technological developments in Clinical Laboratory. Several Performance Specifications based on Biological Variation BV have been defined through years (Figure 1). Including Tonks, Aspen and SIX SIGMA 6S levels depending on reference limits of most commonly used tests in Clinical Laboratories.10,14
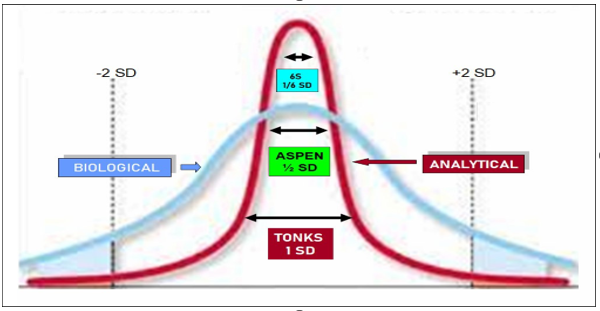
Figure 1 Analytical goals with medical relevance for clinical laboratory depending on biological limits.
Tonks criteria is the most widely accepted procedure, equivalent to one Biological Standard Deviation covers 1/4 =0.25 of normal range. In 1977, nineteen years later, E.Cotlove at CAP Conference in Aspen, Colorado, USA, recommended to modify this criteria reducing Tonks criteria by half =0.125.15,16 More recently on XXI century, when automation, informatics, and robotics experimented a considerable evolution and development, and new technologies have been well established in Clinical Laboratories, it has been proposed that Analytical Performance Specifications APS level may be improved in order to reach SIX SIGMA 6S level where challenge is to reduce variability in 1/6 of Biological Standard Deviation =0.042.17
AQC: analytical quality control
A state of control is achieved when a procedure in laboratory makes it possible to confirm that Analytic Variability is narrower than Biological Variability, [AV < BV] determining in consequence that reliability of results of studies in patients can be guaranteed.18
AQC =AV < BV RCV = AV / BV AQC= RCV <1.0
In essence, precision represents the reproducibility of results that is achieved on a continuous basis. In Clinical Laboratory it is quantified through Coefficient of Variation [CV% =Std.Dev/X]. To improve confidence of a laboratory test, the strategy should start assessing Analytical Variability [AV] according to Reference Limits [RL]. Precision observed in IQCP Internal Quality Control Program is generally closer to Aspen’s ½ SD criteria, while EQAS External Quality Evaluation Schemes are closer to Tonk’s 1 SD level due to statistical reasons, confidence intervals vary inversely to level of uncertainty, given number of variables involved in process. In consequence Tonks criterion may be a more suitable coefficient of variation for External Quality Assessment EQAS, while Aspen criterion is the level that should be achieved in Internal Quality Control Program IQPC. SIX SIGMA 6S criteria are actually considered as a level of excellence to be achieved by laboratories with a cutting-edge status of automation (Table 3).
External Quality Assessment Schemes |
EQAS |
4 SIGMA |
Manual |
4 SIGMA |
|
Internal Quality Control Programs IQCP |
Semiautomated |
5 SIGMA |
Automated |
6 SIGMA |
Table 3 Sigma analytical goal depend on technological level
RCV: relative coefficient of variation
Calculation of realistic and challenging Goals is first step on any Quality Control System. Relative Coefficient of Variation RCV allows performance specifications for any analyte, provided only that there are adequate reference limits for population attended. RCV Relative Coefficient of Variation calculation allows the integration of Medical Relevance, Biological Variation and Technology through a reliable and easy approach for any measurand with the only condition of having suitable reference limits for attended population and calculation of ACV Analytical Coefficient of Variation for each test.10,18,20,21
Measurement of Biological Coefficient of Variation BCV and Analytical Coefficient of Variation ACV allows calculation of Relative Coefficient of Variation RCV and estimation of Sigma Level.19
RCV = ACV%/ BCV%
RCV < 0.250 = 4 SIGMA = TONKS RCV < 0.125 = 5 SIGMA = ASPEN RCV < 0.04 = 6 SIGMA
Biological Variation BV and Analytical Variation AV are inherent to any measurand in nature including laboratory tests. Coefficients are variable depending on when and where are used including Internal Quality Control Programs IQCP or External Quality Assessment Schemes EQAS. When calculating Relative Coefficient of Variation RCV in well-controlled methods, it is found that relation between Biological Variability BV and Analytical Variability AV expressed as RCV is always less than 1.0, since otherwise there would be a risk of reporting false positive and false negative results. As laboratory precision improves RCV will give a result <0.25 according to Tonks, <0.12 according to Aspen and <0.04 when 6 SIGMA level is reached.
Analyzing data from Tables IV, V, VI, VII, VIII, IX, X it is evident that in most of the tests CLIA´s Target Values 2024 are still closer to Tonks 1SD than to Aspen ½ SD Criteria, a level proposed by E. Cotlove at CAP Conference in Aspen, Colorado, USA, 1977 almost 50 years ago when actual state of the art technologies were not available (Table 4).
TEST |
Units |
Min |
Max |
Tonks 1SD |
Aspen 1/2 SD |
6 Sigma 1/6SD |
CLIA 2024 Target VALUE ± |
CLIA 2024 RCV=CLIA/Tonks |
Hemoglobinn |
g/dL |
13.5 |
18 |
7.10% |
3.60% |
1.2% |
7.0% |
1.0 |
Glucose |
mg/dL |
60.0 |
100 |
12.50% |
5.60% |
1.9% |
20.0% |
1.6 |
Creatinine |
mg!dL |
0.5 |
1.2 |
20.60% |
10.30% |
3.4% |
15.0% |
0.7 |
Cholesterol |
mgldL |
100.0 |
200 |
16.70% |
8.30% |
2.8% |
10.0% |
0.6 |
Total Protein |
g/dL |
6.4 |
8.8 |
7.90% |
3.90% |
1.3% |
10.0% |
1.3 |
Sodium |
mEq/L |
130.0 |
145 |
2.70% |
1.40% |
0.5% |
2.9% |
1.1 |
Calcium |
mg/dL |
8.2 |
10.2 |
5.40% |
2.70% |
0.9% |
11.0% |
2.0 |
AVERAGEE |
10.40% |
5.10% |
1.70% |
10.8% |
1.0 |
Table 4 Comparision of CLIA proficiency testing requirements vs criteria based on Tonk´s biological variation21
Achieving Analytical Goals AG depend on level at which are applied. Not all laboratories operate in same conditions, there are laboratories that still use manual methods, while others are semi-automated and some have a high level of technological development. Quality Improvement of Performance depends to a large extent on establishing level at which each laboratory is at present time. From there, continuous improvement of good practices should be sought by raising level of automation under an intelligent strategic plan.
CLIA: clinical laboratory improvement amendments
PTR: proficiency testing requirements
Clinical Laboratory Improvement Amendments of 1988 (CLIA) are Federal Regulatory Standards applicable only within United States of America. The objective this program is to ensure quality laboratory testing for health assessment or to diagnose, prevent, or treat disease. Laboratories must be properly certified to receive Medicare or Medicaid payments.
In 2019 CLIA proposed a new set of quality requirements for proficiency testing. In July 2022 new goals slated for implementation in 2024. Recent revision of PT Proficiency Testing Requirements and update to the criteria for acceptable performance and administrative processes for PT programs will be effective on July 11, 2024, two years after the publication date of the final rule in the Federal Register.20,21 CLIA Requirements have been evolving continuously while new tests are developed faster representing challenges on traceability, verification, validation which may depend on understanding the relation among BV Biological Variation and AV Analytical Variation to establish Reference Values QC Standards including Analytical Goals & Performance Specifications that can be established through 6S methodology described in this document. Quality improvement depends on establishing actual level in which laboratory is performing. In order to improve quality it is necessary to establish a formal WWWWH Strategic Plan, to enhance laboratory practices and to develop technology under human responsibility in order to not only elevate status of automation level (Table 4).
2024 CLIA Proposed Acceptance Limits for Proficiency Testing
Technology and Quality level are never static. Each time a process delivers an outcome that is under the established level it shall be considered a defect and must be catalogued as a non-conformity. Emphasis should be encouraged on analysis of data to uncover cause of defects to eliminate variability from process. Ultimate goal is to deliver results to customers, exceeding critical level on every occasion to generate “virtual perfection" from customer’s perspective. Common sense and will power are not enough to achieve dramatic improvements. Only way is to ask consistent questions and use rigorous statistical and financial analysis. When problems are reduced, costs decline, while customer satisfaction and confidence increase e(Table 5–8).
CQI: continuous quality improvement
Solving problem through teamwork is an important element. Improving organizational performance overall commitment to quality encompassing not only to work processes, but also Top Management as one. Main responsibility at Top Level is to establish an intelligent strategic plan to evaluate, understand and determine how various components of the organization are resourced in order to work together to deliver quality to customers (Figures 2 & 3).
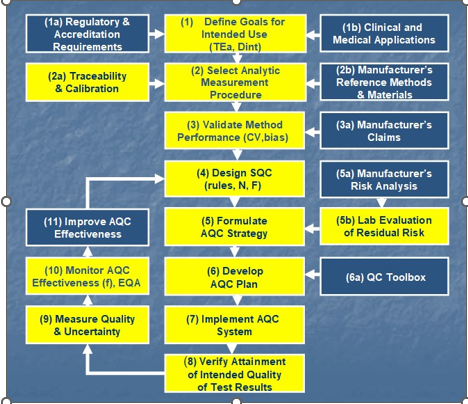
Figure 2 Analytical quality control AQC plan according to Je Westgard.28
A Strategic Plan such as WWWWH is required to comply with to ISO 15189 and CLIA 2024 requirements, emphasizing medical relevance, the establishment of Analytical Goals AG on biological bases and the revision of the metrological traceability of the method that will be used to calculate uncertainty and the risks of the strategy that will be used. Before beginning it is advisable to carefully plan and organize the program in addition to develop a budget in which the cost/benefit is calculated to eliminate all non-essential elements, leaving only those that are considered crucial (Table 9).
Internal quality control
Traceability is manufacturer’s responsibility; laboratory should request the documentation. Validation is a laboratory responsibility. Proper personnel should review and comply with manufacturer recommendations, but not only limit to them. The number, frequency and level controls depend on the number of tests and the magnitude of the analytical round; type test: manual or automated; the type of round (continuous flow or batch); the number of shifts per day and days of the week that performs test plus additional events such as batch change, calibrations, blackouts, etc. Each Head of the Laboratory must design and document a control plan. In general, it may be recommended to locate a normal control and an abnormal at the beginning and end of the round (n = 4 controls) compare and assess the results. If you notice differences, it is appropriate to add a couple at the midpoint of the round. Evaluate the outcomes of patients by calculating the average and median of all the results that are within of reference limits. Note and also monitor the percentage of abnormal results. Perform a detailed statistical control of all data (controls and patients).22–25
External quality assessment
The Proficiency Test Provider must be Accredited ISO 17043:2010. Uniformity and accuracy evaluation is the responsibility of the Proficiency provider. The program must use unbiased high-quality controls. The frequency must be at least monthly. The laboratory must retain an aliquot to make verifications. Report results must be within the first 72 hours so that appropriate measures can be applied. The most important for evaluating accuracy parameter is Bias% vs. Assigned Value. The laboratory must apply preventive and corrective measures utilizing independent «Third Opinion Controls» systematically.
Laboratory Medicine is a Medical Specialty, in which multiple disciplines converge, including Clinical Chemistry, Clinical Pathology, Molecular Biology, etc. so that nowadays the concept of Clinical Laboratory Professionals has been increasingly accepted. Given that the Medical Relevance MR is currently the fundamental premise, the understanding and application of BV Biological Variability and AG Analytical Goals is better understood and accepted in order to meet the needs of the EBM Evidence Based Medicine to grant the maximum benefit at the lowest risk and at the best cost for patient and other customers.26
The key point of EBM depends mostly on Clinical Laboratory, which generates the basis on which more than 70% of medical decisions are taken. Medical Relevance MR of Clinical Laboratory is the fundamental premise. Quality Health Care QHC from the perspective of efficiency lies precisely in the Clinical Laboratory where the need for technical competence and Quality Management Systems QMS is clear; including traceable, well controlled and validated methods. The first step to achieve Quality is the elaboration of a strategic plan that shall include specific, measurable, achievable and challenging Analytical Goals AG that actually are being developed worldwide on Biological Variation BV basis in order to achieve Medical Relevance MR. Technology has been evolving on today’s society including automation, cybernetics, informatics, robotics, and telecommunications through the last two decades of the 20th Century and the first two decades of the 21st Century. From the perspective of Laboratory Medicine, it is feasible to predict that its importance, significance, growth and development will continue on this trend that has been improving depending greatly on cybernetics.
Quality Management Systems QMS are living entities that evolve, increasing availability, reliability, importance, significance and dependence of laboratory information in all medical fields and specialties, since the sum of three elements including Internal Quality Control Programs IQCP, External Quality Assessment Schemes EQAS and Accreditation Programs has resulted on a highly significant impact on the importance of Laboratory Medicine, resulting in an increase in the diversity and number of tests and procedures available to detect diseases, confirm the diagnosis, establish the prognosis, indicate and evaluate treatment.27
Establishing attainable and challenging goals is first step in any AQCS Analytical Quality Control System. RCV Relative Coefficient of Variation calculation allows the integration of Medical Relevance, Biological Variation and Technology through a reliable and easy approach for any measurand with the only condition of having suitable reference limits for attended population and calculation of ACV Analytical Coefficient of Variation for each test.
Continuous Quality Improvement CQI depends on understanding actual level in which laboratory is performing. From there it is necessary to systematically improve technology under Clinical Laboratory Professionals supervision in order to elevate Quality. Technological Dimension must always be submitted under the authority of QMS Quality Management System which depends on Human Dimension. In order to improve it is necessary to establish a formal WWWWH Strategic Plan, to enhance laboratory practices and to develop technology under Quality Management System QMS in order not to depend only on automation.
Every laboratory has unique patient population, clinical and interpretative needs, etc. It’s not possible to create a single, top-down set of performance specifications that is appropriate for all laboratories and all clinical care situations. So, each laboratory should make their own choice even if in today’s practice labs don’t generate their own specific goals for targets for quality. Generally, they simply adopt recommendations from the scientific literature and regulatory/accreditation requirements, such as the CLIA PT goals. The fact that they are judged by the CLIA PT goals makes those goals the most relevant performance specifications to use for laboratory testing in America and elsewhere as consequence.
None.
The author declares that there are no conflicts of interest.
None.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.