eISSN: 2469-2778


Review Article Volume 4 Issue 1
1Department of hepato-pancreato-biliary surgery (HPB), Imperial College, UK
2John Radcliff Hospitals, UK
Correspondence: Jayant Kumar, Department of Surgery & Cancer, Imperial College, London, W120HS, UK
Received: January 05, 2017 | Published: January 31, 2017
Citation: Kumar J, reccia I, Kusano T, et al. A Systematic review on the erythropoietin receptors expression in various cancers and importance of recombinant erythropoietin in chemotherapy induced anaemia. Hematol Transfus Int J. 2017;4(1):13-17. DOI: 10.15406/htij.2017.04.00072
Erythropoietin (EPO) is the primary and influential mediator of red cell synthesis. Its action is mediated by various pathways as JAK2-STAT5, Ras-Raf-MAP kinase, PI-3K-Akt and protein kinase C and adaptor protein CrkL following interaction between EPO and erythropoietin receptors (EPOR) in the various cells. Apart from erythropoiesis it also regulates neuronal functioning and angiogenesis etc. Nevertheless, the pathological presence of these EPOR receptors is seen in various tumour cells that have raised concerns regarding enhanced tumour growth and impaired overall survival. Though the expression of these receptors and its function varies in tumour types and individuals. The recombinant EPO plays an important role in the treatment of cancer-related anemia. However, it must be used judiciously according to individual needs and may be in future EPOR status assessment should help in assessing response and predicting any unwanted adverse effects.
Keywords: erythropoietin, JAK2-STAT5, glycoprotein, paracrine
EPORs, erythropoietin receptors; RBC, red blood cell; HIF, hypoxia-inducible transcription factors; RHuEPO, recombinant human EPO
Erythropoietin (EPO) discovered in 1906 is a circulatory hemopoietic glycoprotein hormone involved in the red blood cell (RBC) production. Later it was found that it has multitude of actions apart from erythropoiesis as affecting endothelial cell growth, nerve cells and tumour cells expressing erythropoietin receptors (EPORs).1 The human EPO gene is located on chromosome 7q22 translates 193 amino acid proteins (Figure 1). Its expression in interstitial cells of kidney and liver is regulated by oxygen level through hypoxia via hypoxia-inducible transcription factors (HIF). There are certain other transcription factors also involved in this process as HNF-4 alpha (Hepatocyte nuclear factor 4-alpha), NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) and inhibitory GATA-2. The presence of normal oxygen tension hydroxylated the HIF alpha subunits, leading to its proteasomal. Degradation while in cases of hypoxia, as the HIF molecule cannot be hydroxylated and therefore stabilized. Post-translational modification as glycosylation and disulphide bond formation plays an important role in affecting synthesis, structural stability, secretion, plasma half-life and degradation.2 Renal cortex is the principal site of EPO production in an adult human, while the liver is main production site in the fetus. Following it’s secretion into blood EPO travels to bone marrow to bind with the EPOR present on the surface of the erythroid precursor cells, thus helps in their differentiation, proliferation and survival. Along with that it also influences other body tissues as neurons, endothelial cells and tumour cells through endocrine, autocrine and paracrine pathways.
EPOR in hemopoietic tissues
The Structure and function of EPOR were primarily studied in erythropoietic tissue. Initial identification of EPOR and its binding sites were done by incorporation of radio labeled thymidine into EPO. Development of recombinant human EPO (RHuEPO) has added new dimensions in the discovery of EPOR in various cells. The density of these receptors varies with the cell types as erythroid colony forming cells have highly enriched receptor number of approx. 1000 receptors per cell. Interestingly enough EPOR shows two different types of affinity influencing the inherent activity of these receptors.3 The EPOR gene is approximately 6kb in size and located on chromosome 19q12. Its expression in erythropoietic and neuronal tissues are modulated by the factors as anemia and hypoxia predominantly however, other factors such as stem cell factors, interleukin-1, interferon, ionomycin and phorbol also play important role. The EPOR, protein made of 508 amino acids is owned to a cytokine receptor super family. It has an extracellular region containing 225 amino acids, the Trans membrane region of 22 amino acids and cytoplasmic region of 236amino acids.4
Signal transduction
The interaction of EPO with EPOR induces many important signal transduction pathways with are worth mentioning here. The binding of EPO with EPOR induces a conformational change that caused activation of various kinases and other molecules of information waves towards nucleus. This takes place through phosphorylation of numerous kinases following activation of EPOR. The most common pathways are JAK2-STAT5 pathway, Ras-Raf-MAP kinase pathway, PI-3K-Akt pathway and protein kinase C pathway and adaptor protein CrkL pathway. Apart from directing signal to the nucleus and promoting erythropoiesis, they play convey other important signals. JAK2-STAT5 pathway has anti-apoptotic role in erythropoietic cells and other tissues (Figure 2).4–6
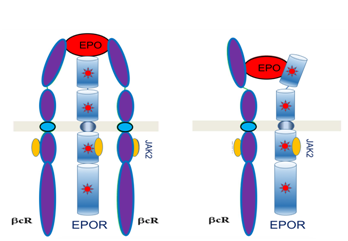
Figure 2 Showing EPOEPOR interaction and signal transduction pathways inside cell. The most common pathways are JAK2-STAT5 pathway, Ras-Raf-MAP kinase pathway, PI-3K-Akt pathway and protein kinase C pathway and adaptor protein CrkL pathway.
EPOR in non-hemopoietic tissues
EPO and EPOR induced tissue protective effect has been noted in various non- hemopoietic tissues as neuronal tissues, blood vessels, heart, liver, kidney, gastrointestinal epithelium, pancreatic tissues, ovaries, uterine tissues, placenta.3,7–13 EPOR mRNA and EPOR protein present in central and peripheral nervous system induces both neurotrophic and neuroprotective effect by promoting proliferation and differentiation of neuronal stem and progenitor cells.14–18 Therefore, of major importance are the seminal studies are by Sakanaka et al.19 who reported prevention of neuronal ischemia and learning disability following introduction of EPO into the lateral brain ventricles of rodents.19,20 This might be because EPO up regulates Bcl-xl expression in neuronal and maintain mitochondrial function.21–24 The increased expression of EPOR activates PI-3 and the MAP kinase in endothelial cells of blood vessels and cardiac myocytes. Studies in animal model proved its role in revascularization, cardio-protective against ischemic injury whereas at high doses it showed anti-apoptotic effect and causes vasoconstriction.25–30
EPOR in tumour tissues
As seen in other tissue type EPOR are also expressed in various tumour tissue as prostate, colorectal, pancreas, breast, renal, hepatic cellular carcinoma, head and neck, melanoma, lung, endometrial, ovarian and uterine carcinomas are some important ones.31–37 The present essay is aimed to review the structural and functional aspects of expression of erythropoietin receptors (EPOR) in tumor cells and their possible influence on tumour growth. Along with, that further analysis has been to drawn regarding clinical significance of these receptors and effect of recombinant EPO on various tumors expressing these receptors.
In the landmark study by Sinclair et al.38 they demonstrated no evidence towards amplification of EPOR in various biopsy samples of tumour tissues as brain, colon, kidney, breast, lymphoma, prostate and lung. Although certain studies showed amplified EPOR expression based on the detection rate of polyclonal antibody C-20, M-20 but later studies refuted these results because of non-specific ability of these antibody to cross react with other proteins too.38–44
Clinical importance of EPO in EPOR expressing tumors
Various studies have shown the presence EPOR mRNA in culture lines of human cancer cells,44–46 used reverse transcriptase PCR techniques to demonstrate these receptors in different human cancer lines (HeLa, HEK293T, RCC4, SW 480, MCF-7, HepG2). However, they didn't contemplate any quantitative alterations in spite of hypoxia or EPO therapy.46 Certain scientists demonstrated hypoxia and EPO induced increase in expression of EPOR mRNA results in increased survival of MCF-7 breast cancer cell lines and SiHa cervical cancer lines in a paracrine manner and promoting angiogenesis.47,48 The effect of EPO in tumour cells are not straight forward as many tumour displayed no change in cancer cells while some tumour cell lines such as human neuroblastoma cells showed growth inhibition; however, administration of high doses EPO (≥10U/mL) has been found to be associated with significant protein phosphorylation and angiogenesis in breast cancer cell lines.49,50 Contrary to this cell lines of prostate and renal cancer cells showed increase growth with relatively lower concentration of EPO.35,41,51,52 Having said that, there is critical doubt over clinical relevance of these results in the real clinical world regarding assessing the tumour response. The discrepancy in results have been noted in between tumour cell lines and laboratories, a typical example could be study by Kokhaie et al.53 where they reported no response to EPO congeners in different cell lines of lymphoid and myeloma cells.53 How Bauer et al.54 studied the effect of EPO on 53 primary human renal and colorectal tumor cell lines. They demonstrated enhanced growth in 2 specimen and inhibition of growth in 5, while no effect in rest specimens.54 The conflicting behavior of this tumour cell could be due to the lack of growth synergy between tumour cells in culture lines, difference in culture conditions as absence or presence of cell lines, preparation of cell lines.55–57 Nevertheless, researchers tried to find out the role EPO in conjunction of other chemotherapeutic drugs though results are far from any fruitful surmise and consensus of clinician validates it as double edge sword. Carvalho et al.58 reported increased frequency of apoptosis on exposure of daunorubicin or vinblastine and EPO combination drugs in the renal carcinoma cell lines, whereas, Gewirtz et al. failed to exemplify any interference in MDA-MB231 and MCF-7 breast cancer cell lines.58 In antithesis to the above results few studies have reported resistance to chemotherapy in conjunction of very high threshold of EPO in HT 100 cervical cancer, U87 glioma, melanoma and HeLa cell line by inducing tyrosine phosphorylation of GM-CSF receptor and abatement of pro-apoptotic proteins Bcl-2 and Bcl-10.59,60
In presence of great uncertainty with in vitro studies scientist tried to withdraw conclusion of EPO-EPOR response in preclinical animal models. In a landmark study of ovarian and uterine transplanted nude mice by Yashuda et al.32 exhibited tumour downgrading following the application of soluble EPOR or anti-EPO antibodies. Likewise, they also demonstrated anti-angiogenesis and anti-apoptotic effect following imposition of EPOR blocking peptides in human melanoma and choriocarcinoma xenograft models. The possible explanation of these could be EPO mediated anti-apoptotic effect causing tumor growth.1,32 The RHuEPO therapy in cancer patients have been given to treat or prevent chemotherapy or radiotherapy induced anemia and to curtail the need of blood transfusion, nevertheless, they also increase the efficacy of chemotherapeutic drugs and radiotherapy through improved oxygenation. Reported increased efficacy of cyclophosphamide than carboplatin in rat models in presence of RHuEPO, as maintained hematocrit and improved oxygenation aids in sensitization of cytotoxic effect of chemotherapy in animal tumor models.61,62 At the same time these effect alter with tumour type as noted reduction in tumor growth with cisplatin and RHuEPO though couldn't produce similar effect with mitomycin C or cyclophosphamide.63 McKinney et al.64 and Debeljak et al.65 proposed three possible theory mechanisms regarding tumor progression and reduces survival in some cancer types following RHuEPO therapy for cancer chemotherapy induced anemia. First due to local effects in tumour microenvironment affecting tumor cells or other as blood vessel endothelium and tumor-associated macrophages; second is direct or indirect systemic effect that impairs survival; third could be direct effect of EPO on stem cells leading to tumour progression and poor survival in some cancer patients. Recombinant EPO has been in successfully used in clinical practice for treatment of anemia in the cancer patients along with increase effectiveness of chemo-radio therapy though by contrast theoretical possibility of tumour growth is not denied. Mutiple randomized trials and subsequent analysis gave apt evidence towards its beneficence in treatment of cancer related anemia.66–68 In a meta-analysis demonstrated only 0.64 relative risk for blood transfusion following recombinant EPO therapy.59,69 The most perilous adverse events causing mortality risk were increased risk for cardiovascular and thromboembolic events, tumour growths and pure red cell aplasia. The detailed analysis various studies according to tumour response and patient survival have been under taken. Antonadou et al.70 reported statistically significant disease free survival in patients with pelvic malignancies undertaking radiotherapy with recombinant EPO. Similarly, Blohmer et al.71 demonstrated significantly better disease free survival in high risk cervical carcinoma patients receiving combined recombinant EPO with chemo-radiotherapy. In the same line Leyland et al.72 in Breast cancer and Henke et al.73 in Head and Neck cancer and Wright et al.74 in lung cancer have found the negative influence of erythropoietin Several other studies, showed increased tumour growth and reduced tumour survival, where increased EPOR expression in these tumors contributes towards the poor survival, tumor growth or disease recurrence.12,75 The expression of EPOR acts as an important prognostic factor i.e. intensified expression is inversely related with prognosis and reduced response to chemo-radio therapy.76–78 Further studies based on EPOR subtypes are needed in order to better understand the tumour biology and substantiate the treatment justification in cancer chemotherapy induced anemia.
In the midst of the present evidences there is no doubt that EPO has detrimental influence in cancer producing EPOR, leading to increase tumour growth and poor survival. The main concern for its use is chemotherapy induced anemia in cancer patients which needs attention, having said that there is no clear cut guideline regarding initiation of EPO in anemic cancer patients. The consensus is towards assessing the EPOR receptors status in tumors like estrogen-progesterone receptors in breast cancers. So that EPO simulating agents could be prescribed in patients with EPOR negative tumors. In 2008, American Society of Clinical Oncology laid its guidelines regarding treatment of anemia in cancer patients and fixed the upper limit of treatment at hemoglobin level of 12g/dl.79
None.
The author declares no conflict of interest.

©2017 Kumar, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.