Advances in
eISSN: 2572-8490


Research Article Volume 5 Issue 2
International Academy of Ecology and Medicine, Ukraine
Correspondence: Maya Vergolyas, Associate Professor, International Academy of Ecology and Medicine, Kiev, Ukraine
Received: June 24, 2019 | Published: July 22, 2019
Citation: Vergolyas M. Research of cytotoxic activity of water from different water supply sources. Adv Tissue Eng Regen Med Open Access. 2019;5(3):92-96. DOI: 10.15406/atroa.2019.05.00105
Objective: Evaluation of cytotoxic drinking water from various water sources in cell culture in vitro.
Materials and methods: We investigated cytotoxic effect of control water that obtained from laboratory as recommended State Standards of Ukraine (SSTU) 4174:2003, tap water, pump-room and bottled water "Znamenovskaya." Studies were conducted on cell lines HEK-293 (human embryonic kidney cells), lines L929 (mouse fibroblasts) and RTR (pigs cell testicles), from the Institute of Microbiology and Virology of the National Academy of Sciences of Ukraine.
Results: Research in MTT-test cytotoxic mentioned water samples showed the most toxic activity against human embryonic kidney cell (HEK-293) showed the pump room’s water and tap water (the number of viable cells – 64.41% and 77.00% respectively). In water "Znamenovskaya" number of viable cells – 81.75%, control water demonstrated less impact 91.73%. The test with sulforodaminom B number of living cells when exposed to control water was 103.00%, tap water-82.45%, water from the pump room-89.20%, "Znamenovskaya"-96.75%. The test with sulforodaminom B number of viable cells was highest after adding control water (81.45%), "Znamenovskaya"-69.45%, tap water-68.2%, and the least impact on water from the pump room-66 95%. Painting neutral red showed that the cytotoxic activity had all control water, as evidenced by reduction in the number of living cells 88.1% compared to the control sample.
Conclusion:The highest cytotoxic effect according to MTT-test and test with sulforodaminom B on human kidney cells (HEK-293) showed tap water, and the mouse fibroblast (L 929) and testicles of piglets RTR-water with the pump room. The least effect on cell viability caused water control. The negative impact of water on cell viability manifested in dysfunction and mitochondrial protein synthesis. More sensitive to the toxic effects of water appeared RTR cell lines that may indicate the possible negative impact of water on the reproductive system.
Contamination of the biosphere with harmful chemicals threatens the state of the environment, the health of the population and limits the possibility of further balanced development of society.1 One of the urgent and urgent problems of today is to provide the population of Ukraine with high quality and safe drinking water. The influence of drinking water on the consumer may vary depending on both the combination of the impurities themselves and their concentrations, even if they are in accordance with the established sanitary-and-hygienic standards. Biologically active impurities present in water may be present, which adversely affects human health and other living organisms. Modern physico-chemical methods of analyzing the composition of water do not allow comprehensively assessing the quality of water and predicting the effect of substances present in it on biological objects. Thus, there is a need to develop and use new methods for the integrated assessment of the safety and quality of drinking water with the possibility of predicting its effects on various living organisms.2 In modern toxicological practice, along with traditional laboratory tests, xenobiotic toxicity tests use alternative models of different biological organisms-invertebrates, hydra, microorganisms, plants, cultures of human cells and animals.3,4 Many studies have confirmed that in vitro methods are fairly accurate, fast in production and cost-effective. The prospect of research using in vitro methods is intensifying and increasing attention to the role of ethical aspects when choosing a research object, increasing the interest of scientists and the general public in a humane attitude towards warm-blooded animals, and reducing their number in scientific experiments.5
According to the recommendations of ISO (International Organization for Standardization), the ICCVAM (Interagency Coordinating Committee on the Validation of Alternative Methods) and other international organizations, the study of the cytotoxic effects of substances can be carried out on primary cultures of cells and tissues isolated from the body of animals, humans, as well as permanent cellular lines derived from certain types of tumors. The selection of target cells depends on the expected biological effects of the test substance. Research directly on the culture of human cells simplifies extrapolation of data and prediction of toxicity of a substance relative to the human body.4,6–8 The purpose of our study was to assess the quality of drinking water from different sources of water supply based on its cytotoxic effects on human and animal cell cultures in vitro.
During the study, the cytotoxic action of control water was determined, which was obtained in accordance with the recommendations of State Standards of Ukraine (SSTU) 4174:2003 in a laboratory based on the International Academy of Ecology and Medicine; water from the water supply; water from the pump room and packaged water. Selected samples of drinking water differed in their chemical composition. The studies were conducted on cells of the HEK-293 line (human embryonic kidney cells), lines L929 (mouse fibroblasts) and RTR (transplantation cells of testicular piglets) courtesy of the Institute of Microbiology and Virology National Academy of Sciences of Ukraine.
Cells were cultured in a RPMI 1640 ("SIGMA", USA) medium containing 4 mmol/l L-glutamine, 10% embryonic calf serum (SIGMA, USA), 40μg/mg gentamicin, in a thermostat at 37°C from 5 % СО2 on plastic tableware (SenteLab, Ukraine). Environmental changes were carried out every 2days. Peres cells were carried out with the Versen solution when the cells formed a solid monolayer (4-5days of growth). Studies were conducted on condition that at least 90% of live cells were present in the cell suspension. During the study, the cells were planted in 96-well plates at a concentration of 1×105/ml (100μl per well) in a complete growth medium. After 24hours, water samples were added to these media. For control, wells with cells that did not add water were taken. The cytotoxic activity of water in relation to cell cultures was determined in tests with methyltetrazolium (MTT) and sulfodoamine B (SR B) after 24h incubation. The experiment was repeated twice.
The principle of the MTT method is based on the ability of the succinate dehydrogenase-the enzyme of the mitochondrial membrane of the cell to restore the yellow salt of methyltetrazolium (3-[4,5-dimethylthiazol-2-yl] -2,5-diphenyltetrazolium bromide) to the crystals of the purple formazan accumulating as a result of this reaction in the cytoplasm of living cells. Thus, the intensity of the accumulation of crystals of formazan in the cytoplasm can be judged on the level of mitochondrial respiration of the cell, which is an indicator of their viability. The dyeing test by sulfurodamine B allows you to determine the content of total protein, which can be an indicator of cell growth and proliferation.9 The number of viable cells in both methods was calculated according to the formula: ODel / ODcl x 100%, where: ODcl-optical density of the solution in control wells; ODel is the optical density of the solution in the experimental wells. The optical density of control and experimental wells was determined using a multi-wave spectrophotometer Sunrise Tecan (Austria) at a wavelength of 540nm.
The results of the MTT test showed that the smallest cytotoxic effect on human embryonic kidney cells was made by control water, and the amount of viable cells was 91.73±0.06% under its influence. The largest cytotoxic activity in relation to the culture of the HEK-293 cells showed samples of water from the water supply and pump room (the number of viable cells for their effects was 64.41±6.98% and 77.00±6.14%, R<0.05 in comparison with the control water). For the influence of packaged water, the number of viable cells was 81.75±0.64%, (R<0.05). According to the data obtained in the sulfodoamine test, the amount of living cells after the 8addition of control water was 103.00±1.10%, while water from the water supply was 82.45±0.50%, and the water from the pump room was 89.20±3.40% and packaged water-96.75±1.05% (R<0.05) (Table 1). These data may indicate that control and packaged water did not significantly affect protein synthesis.
Study |
% of viable cells for the effects of various water |
|||
Water control |
Water from the water supply |
Water from the pump room |
Packaged Water |
|
MTT- test |
91,73±0,06 |
64,41±6,98* |
77,00±6,14* |
81,75±0,64* |
Sulfo-rhodamine B test |
103,00±1,10 |
82,45±0,50* |
89,20±3,40* |
96,75±1,05* |
Table 1 Cytotoxic activity of drinking water from different sources of water supply to cells of the HEK-293 line
Note: * marked probability difference (R<0,05) with respect to indicators for control water
In the study of cytotoxic activity of drinking water in relation to cells of the line L929 it was found that when adding control water, the number of living cells was 88.55±8.55%. Water from the pump room showed the highest cytotoxic activity in relation to the mouse fibroblasts (the number of viable cells was 58.56±1.45%) and water was packed (the number of viable cells was 61.90±0.80%). When added to the culture medium of L929 water from the water supply, an increase in the vitality index is set to 134.30±0.01%, this increase may be a sign of stimulation of the respiratory function of these cells (Table 2). According to the test for sulfodoamine "B", all the water samples studied had approximately the same effect on the vitality of the L929 cells. Thus, the number of viable cells after adding to the control water culture was 70.40±1.50%, water from the water supply-66.85±3.05%, water from the pump room-65.50±1.60% and water packed-68.90±4.50% (Table 2).
Study method |
% of viable cells for the effects of various water |
|||
Water control |
Water from the water supply |
Water from the pump room |
Packaged Water |
|
MTT- test |
88,55±8,55 |
134,30±0,01* |
58,56±1,45* |
61,90±0,80* |
Sulfo-rhodamine B test |
70,40±1,50 |
66,85±3,05 |
65,50±1,60 |
68,90±4,50 |
Table 2 Cytotoxic activity of drinking water from different sources of water supply to cells of line L 929
Note: * marked probability difference (R <0,05) with respect to indicators for control water
Studies that were performed on RTR cells in the MTT test allowed to establish that, when cell culture with control water was cultured, the number of viable cells was 72.80±0.60%. The largest cytotoxic activity was water from the pump room (the number of viable cells was 61.50±8.00%). Water from the water supply and "Znamenovskaya" had almost the same effect on the survival of the testicular cells of the piglets (65,50±4,40% and 66,75±5,45% of living cells) (Table 3).
Study method |
% of viable cells for the effects of various water |
|||
Water control |
Water from the water supply |
Water from the pump room |
Packaged Water |
|
MTT- test |
72,80±0,60 |
65,50±4,40* |
61,50±8,00* |
66,75±5,45 |
Sulfo-rhodamine B test |
81,45±6,90 |
68,20±1,80* |
66,95±1,95* |
69,45±4,65 |
Table 3 Cytotoxic activity of drinking water from different sources of water supply to RTR cell
Note: * marked probability difference (R <0,05) with respect to indicators for control water
After adding L929 to the water from the water supply, it was observed that they were rounded, increased vacuolization, increased nuclei, reduced their adhesion ability, and changed shape. All of this may be signs of cell apoptosis (Figure 1C). The incubation of cells with water from the pump room contributed to the disturbance of cellular contacts, rounding and lysis of cells (Figure 1D). Under the influence of water "Znamenovskaya" significant changes in the morphology of cells L929 was not established. By appearance, the cells were similar to cells in control wells (Figure 1E).
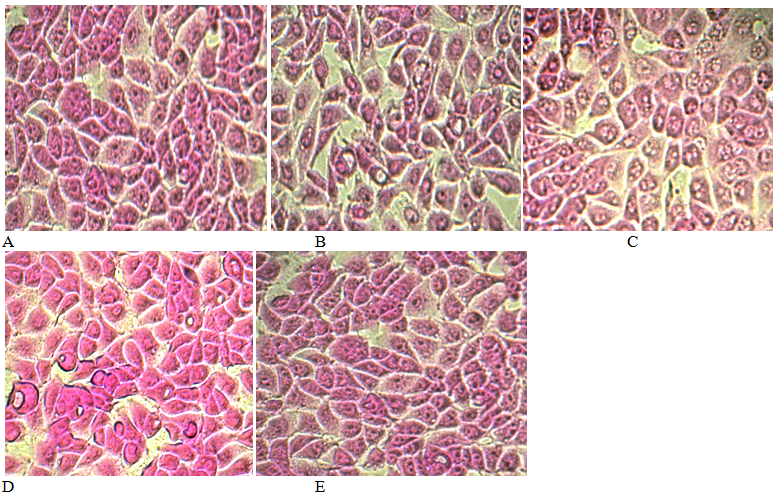
Figure 1 L929 cells after 24 hours of incubation with samples of drinking water (coloring with ulfodoamine B, s.o. 100). A) Control (intact) cells; B) Water control; C) Water from the water supply; D) Water from the pump room; E) Water packed.
Figure 2 shows the intensity of formation of formazan crystals in L929 cells after 24 h incubation with the samples of drinking water (MTT test data). The most intense formation of formazan crystals was found in L929 cells exposed to water from the water supply (Figure 2A-2C), but less than the action of water from the cuvette (Figure 2D). Weak formation of crystals of formazan is a sign of low cell viability, in particular respiratory function of mitochondria, indicating cytotoxic activity of water. In the study of morphology of RTR cells after incubation with drinking water from different sources, the following changes were detected (Figure 3A). Thus, after incubation of cells with control water, the rounding of individual cells and the presence of large vacuoles similar to the air bubbles in them were observed, while the monoshock violation was not observed (Figure 3B). When added to the cells of the RTR water from the water supply was found violation of the monolayer due to rounding and separation of cells, an increase in the number of dead cells. In separate cells there were enlarged vacuoles (Figure 3C).
Water from the pump room when added to the cells of the RTR caused the greatest toxic effect, observed a violation of the monolayer, the appearance of rounded intensively colored apoptotic cells (Figure 3D). Due to the influence of water packed, significant changes in the structure of the monolayer and the cells themselves were not detected in comparison with the control ones (Figure 3E). The obtained research results correspond to the data of literature. Thus, in experiments carried out in Yakubchak OM10 it is shown that the introduction into the culture of cells of chlorinated water from the water supply after 2 hours. Caused cytotoxic action. At the same time, the destruction of the monolayer, gluing, and changes in the morphological structure of cells were observed. The most sensitive to the toxic effects of the test water were human kidney cells of the HEK-293 line, which may indicate a negative effect of water on the urinary system.
Thus, the study of the cytotoxic activity of drinking water from different sources of water supply on the culture of cells in vitro allows the following conclusions:
None.
None.

©2019 Vergolyas. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.