eISSN: 2378-3176


Case Report Volume 5 Issue 6
1Resident of Urology, Pontificia Universidad Javeriana, Colombia
2Universidad del Rosario, Colombia
3Department of Pathology, Hospital Universitario San Ignacio, Colombia
Correspondence: Maria A Ocampo, Carrera 24 No 63C-69, Transversal 2 #67-22 apto 1005, Bogota, Colombia, Tel (57) 3207272080, Fax (57) 3207272080
Received: November 19, 2017 | Published: December 7, 2017
Citation: Chavarriaga J, Torres L, Ocampo MA, Meek E (2017) Segmental Testicular Infarction Mimicking Testicular Seminoma. Urol Nephrol Open Access J 5(6): 00189. DOI: 10.15406/unoaj.2017.05.00189
Segmental testicular infarction could present as a hypoechoic well-rounded mass, with or without vascular flow and negative tumor markers mimicking testicular seminoma. We aim to present a case of segmental testicular infarction of the upper pole of the testis, the microscopic pathological assessment and a state of the art of the current management and diagnosis of this rare entity.
Keywords: testis, testicular neoplasm, seminoma, infarction, orchiectomy, pathology, periodic acid shiff reaction, staining and labeling
Segmental testicular infarction (STI) is a rare entity, the etiology is unclear, although it is thought to be caused due to arterial flow obstruction secondary to venous thrombosis; there has been several predisposing factors described: polycythaemia, epididymo-orchitis, trauma, vasculitis, intimal fibroplasia of the spermatic artery, hypercoagulability disorders and sickle cell disease.1–4 Usually occurs between the third and fourth decade of life, the most common symptom is acute onset orchialgia; Scrotal US remains the first assessment tool, it is described as a wedge-shaped or rounded area of hypoechogenicity within the testis, at doppler evaluation with markedly decreased or absent, although it could have well preserved flow and negative tumor markers, mimicking testicular seminoma.1,3 We aim to present the case of a 33 year-old male with presumptive diagnosis of testicular seminoma, with upper pole STI confirmed by microscopic pathological evaluation.
A 33 year-old male was admitted to the emergency department with a 5 -day complaint of left testicular pain, without other symptoms. At physical exam the left testis was in normal anatomic position and location, the upper pole was tender and mildly swollen with no palpable mass, past medical history was unremarkable, he denied testicular trauma or risky conducts for sexually transmitted disease. Complete blood cell count, creatinine, urinalysis were within normal range; Color Doppler ultrasound revealed a hypoechoic heterogeneous mass with a volume of 2,9 cc and preserved flow, in the upper pole of the left testis, the remaining testis exhibited normal echogenicity and vascularity (Figure 1). Tumor markers (Lactate dehydrogenase 230 U/L, β-HCG 0,10 mlu/mL, Alpha-fetoprotein 2,20 ng/ml) were negative, Abdomen CT and chest X rays did not show metastasis. Our assessment pointed to testicular neoplasia, seminoma. We scheduled the patient for radical orchiectomy; it was carried out without complications.
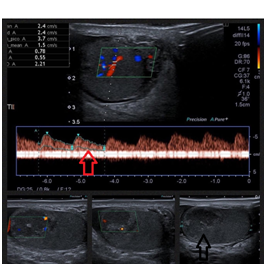
Figure 1 Hypoechoic heterogeneous mass located in the upper pole of the left testis of 17 x 17 x 17 mm, (L x W x H) with a volume of 2,9 cc. Black Arrow pointed to hypoechoic área on the upper pole. Color doppler US showed preserved flow within the mass, the remaining testis exhibited normal echogenicity and vascularity. Red Arrow shows preserved peak systolic velocity within the hypoechoic área of the upper pole of the left testis.
At gross anatomy no visible mass or infarcted area was seen on the upper pole, the specimen was processed by the pathology department following the quality standards of our institution. Microscopically in the hematoxylin and eosin stain no tumoral cells were observed, instead larger areas of pale eosinophilic seminiferous tubules characteristic of coagulation necrosis, accompanied by a slight interstitial hemorrhage and mild inflammatory response. We performed two special stains: Periodic acid-Schiff (PAS) that is a staining method used to detect polysaccharides such as glycogen and mucosubstances such as glycoproteins and glycolipids; In seminoma and intratubular germ cell neoplasia (ITGCN) the majority of tumor cells are positive for PAS stain, we performed to help us clarify the diagnosis given that testicular seminoma was suspected; PAS stain showed us thickening of the basement membrane of the seminiferous tubule without evidence of tumor cells. The other stain was Prussian blue, it is used to detect the iron focal deposits, in our case was positive for siderophages that are hemosiderin containing macrophages corresponding to old-established diffuse interstitial hemorrhage in the infarcted area, although this last featured it’s not pathognomonic of testicular infarction, usually it could be seen within the infarcted area, the Prussian blue stain help us confirmed the diagnosis of STI and in our practice we recommend to use it in case the hematoxylin-eosin microscopic evaluation it´s not conclusive (Figure 2). Routine follow up was carried, the patient came for post-operative visit 8 days after the procedure; He presented asymptomatic, the physical exam was unremarkable (Table 1).
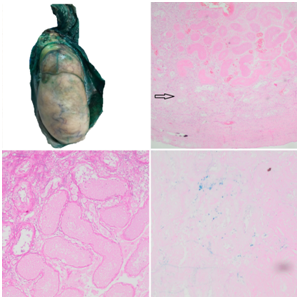
Figure 2 (A) Surgical specimen showing normal gross testicular anatomy, without evidence of tumor or visible areas of infarction. (B) Hematoxylin-eosin Stain showing diffuse interstitial hemorrhage of the upper pole with larger areas of pale eosinophilic regions and coagulation necrosis within the seminiferous tubules, no germ cells were visible. The black arrow shows an área of non infarcted seminiferous tubules. (C) PAS stain showing thickening of the basement membrane in the infarcted área. No PAS positive tumor cells were identified. (D) Prussian blue stain showing siderophages in the testicular interstitium surrounded by necrosis, characteristic of STI of the testis.
|
Segmental testicular infarction |
Testicular seminoma |
Clinical Presentation |
Acute onset of scrotal pain |
Painless mass (Rarely presents with pain), the patient notice a slow growing mass |
Physical examination |
Pain localized to the infarcted area with/without signs of inflammation. |
Palpable painless solid mass |
Color doppler ultrasound |
Wedge-shaped or round hypoechoic lesion |
Hypoechoic lesion |
B-mode ultrasound |
Focal low reflective area with no posterior acoustic enhancement |
Focal area of varying reflectivity |
Tumor markers |
Negative |
Positive in 30% of cases (β-HCG, AFP, LDH) |
Management |
Conservative management |
Partial orchiectomy, Radical inguinal orchiectomy |
Pathology |
Gross: no visible mass or infarcted area |
Gross: brown to pale yellow solid, well-circumscribed mass |
Table 1 Differences between segmental testicular infarction and testicular seminoma
The differential diagnosis of STI are testicular hematoma, epidermoid cyst, orchitis with or without abscess, adrenal rests, sarcoidosis, sex cord-stromal tumor, lymphoma and germ cell tumors of the testis that in 55% of the cases accounts for testicular seminoma. The common finding of all these entities is that at US evaluation are described as well-rounded hypoechoic lesions with or without arterial flow at Doppler US and without elevation of tumor markers that often misleads the diagnosis towards testicular seminoma.1,3,5 Segmental testicular infarction is an unusual diagnosis and it is often confused with testicular neoplasm, fewer than 50 cases have been reported, the current standard for diagnosis is US, magnetic resonance imaging (MRI) showed no advantage over US evaluation. It is often managed with scrotal exploration that ends up in radical or partial orchiectomy because it is misdiagnosed with testicular neoplasia; conservative management has proven to be feasible, Madaan et al. reported on a case series of 19 patients with STI in which 16 of them were successfully managed with watchful waiting, documenting regression of the lesion in 9 of them. The improvements in Color doppler US (CDUS) can help differentiate malignant lesions from a STI allowing to avoid unnecessary orchiectomy, it allows the evaluation of intratesticular blood flow, tumors are often seen with high vascularization while testicular infarctions have deficient vascularization. There are new methods like contrast-enhanced US with microbubble injections that can help differentiate a tumor by displaying an abnormal vascularization in the lesion. STI is considered on CDUS by poor or absent blood flow. There has been reported some variable manifestations in the B-mode US of segmental infarction, a low reflective area with no acoustic enhancement posteriorly or a segmental reflective mass with sacks of low reflectivity. In this case making the differential diagnosis was troublesome given the CDUS reported normal flow to the testis and within the hypoechoic area described at US evaluation; while we were unable to provide an alternative etiology to testicular seminoma, radical orchiectomy was indicated.6,7
The peak of incidence for testicular cancer is between 15 and 44 years, the most common type of testicular neoplasia are germ cell tumors and of these seminoma being the most common subtype, accounting for 55% of the cases; The clinical features are a painless, palpable, solid mass, usually seminoma does not elevate tumor markers, although up to 30% of patients with seminoma could elevate β-HCG due to the presence of syncytiotrophoblastic giant cell, but could never elevate Alpha-fetoprotein.1,8 The initial assessment of testicular seminoma includes high frequency US, typically it shows a hypoechoic, homogeneous mass with increased vascularity. At gross pathology usually a brown to pale yellow solid, well-circumscribed mass is visible, and at microscopic pathology the nuclei is large with prominent nucleoli, sheets of cells and nests are typically seen with fibrous septa disposing the tumor into lobules of tumor cells, the cells are immunoreactive for SALL4, OCT 3/4 , C-KIT, SOX-17. The aim of reviewing testicular seminoma and its clinical presentation is the same as reporting this case; we aim to prevent misdiagnosing testicular seminoma with STI in other urological practices given that if testicular seminoma is suspected radical orchiectomy is inevitable and it is considered overtreatment in the clinical scenario of segmental testicular seminoma.1,9
The segmental infarction of the testis is amongst the differential diagnosis of testicular seminoma, the urologist have to be very careful with the ultrasonographic evaluation and the clinical history to suspect this entity. Efforts have to be made to avoid radical orchiectomy in these cases given the majority of them present in young males and conservative management has proven to be a safe option.
None.
The authors have no conflict of interest to report.

©2017 Chavarriaga, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.