eISSN: 2378-3176


Research Article Volume 14 Issue 1
Department of Urology, Consultant Urological Surgeon, University Hospital of North Tees and Hartlepool NHS Foundation Trust, United Kingdom
Correspondence: Ashwini Gaur, MB.BS(Delhi), F.MAS, CCST(Urology)Moldova, FEBU, FRCS(Urol), DLS(Strasbourg), Department of Urology, Consultant Urological Surgeon, University Hospital of North Tees and Hartlepool NHS Foundation Trust, Stockton on Tees, TS19 8PE, United Kingdom, Tel 441642617617
Received: January 29, 2026 | Published: February 13, 2026
Citation: Ashwini Gaur. High-power, high-frequency holmium laser versus low-power low-frequency holmium laser in retrograde intrarenal surgery: a comparative single surgeon, single centre study on efficacy, safety, and stone clearance outcomes. Urol Nephrol Open Access J. 2026;14(1):8‒14. DOI: 10.15406/unoaj.2026.14.00365
Objective: To compare the stone-free rates (SFR), procedural efficiency, and safety of high-power (100 W) and low-power (50 W) Holmium: YAG laser systems in retrograde intrarenal surgery (RIRS) for renal stones.
Materials and Methods: This comparative study included 204 patients (102 in each group) undergoing RIRS with either a 100 W high-power laser or a 50 W low-power laser. Continuous variables (age, stone size, density, operative time) were analysed using Welch's t-test, and categorical outcomes (overall and subgroup SFRs) were compared using the Chi-square test. Fisher's exact test was used for small subgroups (stones ≥15 mm). All analyses were two-tailed, with p < 0.05 considered statistically significant. Results were reported as Relative Risk (RR) and Odds Ratio (OR) with 95% Confidence Intervals (CI).
Results: The overall SFR was higher in the 100 W group (90.2%) than the 50 W group (81.4%, p = 0.021). Subgroup analysis showed significant improvements for hard stones (HU ≥900) (p = 0.043) and stones ≥10 mm (p = 0.026). For lower-pole stones, clearance was higher in the 100 W group (p = 0.050, two-tailed; however significant (p < 0.05) on one-tailed directional analysis). Operative times and complications were comparable (p > 0.05).
Conclusion: The 100 W Holmium laser achieved higher overall stone clearance rates without increasing operative time or complications. Benefits are most pronounced for stones ≥10 mm and hard stones, with a positive trend for lower-pole stones confirming the safety and efficiency of high-power laser lithotripsy.
Keywords: retrograde intrarenal surgery (rirs), holmium:yag laser, high-power laser lithotripsy, low-power laser, stone-free rate (sfr), renal calculi, hard stones, flexible ureterorenoscopy, intrarenal temperature
Flexible ureterorenoscopy (RIRS) has transformed renal stone management. Advances in Holmium: YAG laser design allow high-power, pulse-modulated systems to operate at frequencies up to 100 Hz, improving dusting and fragmentation efficiency while minimizing retropulsion.
Earlier ≤ 50 W systems, limited to 10–15 Hz, often produced slower fragmentation and larger residual fragments. Newer 100 W dual-paddle designs permit long-pulse dusting and fine fragmentation at low energy, enhancing procedural control.
However, comparative data on the clinical efficacy and safety of high-power versus low-power lasers remain limited. This study compares clinical outcomes between 100 W and 50 W Holmium: YAG lasers used by a single surgeon in a District General Hospital, analysing efficacy, procedural efficiency, and safety. Particular emphasis is placed on overall renal stones, hard renal stones, lower-pole calculi, and larger renal stones (≥10 mm and ≥15 mm).
Study design and setting
This work was registered with the hospital Clinical Effectiveness Department as a clinical audit (Audit no. 2579), using retrospectively analyzed, prospectively collected routine clinical data. Formal NHS Research Ethics Committee review was not sought, in line with local governance procedures for clinical audit. All patients signed a generic consent form for their operation which also indicated that the data can be used for research purpose. Prospectively maintained data was reviewed retrospectively comparing RIRS outcomes using high-power (100 W) and low-power (50 W) Holmium:YAG lasers. All procedures were performed by the author in a District General Hospital setting between September 2017 and December 2023.
Group A: 100 W Lumenis Pulse-modulated Dual Paddle (December 2020 to December 2023)
Group B: 50 W Auriga XL (September 2017 to December 2020)
Most procedures were performed under general anaesthesia; spinal anaesthesia was used for patients with significant comorbidities unsuitable for general anaesthesia.
Data availability statement
The authors confirm the availability of, and access to, all original data reported in this study. Data are available from the corresponding author upon reasonable request.
Inclusion and exclusion criteria
Inclusion: Pre-stented patients with renal stones and concomitant obstructive ureteric stones, and non-pre-stented patients with renal calculi. Stones 5–30 mm (single) or total length ≤60 mm (multiple) in Group A and 5–40 mm (including multiple stones). Longest linear length was used for measurement (not volume). Anatomical variants were included.
Exclusion: Active urinary tract infection, uncorrected coagulopathy, or need for combined percutaneous/open approach.
Pre-operative preparation: Urine culture was obtained only from symptomatic patients. Antibiotics at induction consisted of gentamicin 240 mg with co-amoxiclav 1.2 g or gentamicin 240 mg intravenously or based on any recent urine culture results. Gentamycin was omitted in patients with EGFR < 30 and instead Tazocin was given. Serum creatinine was recorded pre-operatively; urate and calcium were checked for all. All patients underwent non-contrast CT for stone size, location, and Hounsfield Units (HU). Stones with HU ≥900 were classified as hard.
Surgical technique
Access: After cystoscopy, a 0.035-inch sensor-tip guidewire was placed. Rigid ureteroscopy was performed only in pre-stented patients with concomitant ureteric stones (Olympus or Wolf 7.5 Fr). A Boston Scientific ureteral access sheath (11/13 Fr × 36 cm in females and 46 cm in males) was inserted under fluoroscopy below the ureteropelvic junction or to the proximal ureter.
Flexible ureterorenoscope: Olympus URF-V3 digital ureteroscope (8.4 Fr) with irrigation from a pressure bag. Room temperature or cold saline was used for high-power laser lithotripsy, and warm saline was used for low-power laser lithotripsy.
Laser lithotripsy parameters: The 100 W Lumenis laser (200 µm fibre, pulse energy 0.3–1.0 J, frequency 30–53 Hz, maximum power in kidney ≤53 W) (intermittent firing of the laser was used) and 50 W Auriga XL laser (235 µm fibre, pulse energy 0.4–1.0 J, frequency ≤12 Hz, maximum power in kidney ≤12 W) were used according to treatment strategy.
Dusting was performed using low energy and high frequency mode (0.3 J and 30–53 Hz for high-power laser; 0.4 J and 12 Hz for low-power laser). Fragmentation, popcorning, and pop-dusting using intermittent firing were performed with high energy (1 J and 30–53 Hz for high-power laser; 1 J and 12 Hz for low-power laser). Fragments <2 mm were left for spontaneous passage; larger fragments were retrieved with a basket.
Intrarenal urine was sent for culture when urine appeared cloudy or the collecting system was distended.
Post-operative care and imaging: Nearly all patients received a double-J ureteric stent post-operatively (5 Fr or 6 Fr multilength, or 6 Fr × 26 cm magnetic). Discharge was same-day if the patient was afebrile, pain-free, and voiding normally; otherwise, discharge occurred the next day or when medically indicated. Stone-free status was assessed as follows:
Stones ≤15 mm: Ultrasound ± KUB X-ray at 4–6 weeks post-stent removal
Stones >15 mm: Non-contrast CT pre-stent removal (if deemed necessary) or ultrasound± x ray Kub at 4–6 weeks post-stent removal.
Complete SFR was defined as no visible residual fragment; acceptable SFR was defined as residual fragment <5 mm. Stone analysis was retrieved from the hospital ICE reporting system for metabolic guidance.
Data collection and outcomes: Patient demographics, stone characteristics, operative time, hospital stay, and postoperative complications were recorded. Subgroup analyses were performed for hard stones (HU ≥900 or calcium oxalate monohydrate, or mixed with calcium oxalate monohydrate predominating, or cystine stones), lower-pole stones, stones ≥10 mm, and stones ≥15 mm.
Sample size calculation: A convenient sample of 204 patients (102 in each group) was used based on the total number of eligible RIRS procedures performed with each laser system during the study period. No formal a priori sample size calculation was performed due to the retrospective design and predetermined patient cohort availability. The sample size was sufficient to detect clinically meaningful differences in stone-free rates between laser systems based on observed frequencies.
Statistical analysis
Data were analyzed using two different software that used IBM SPSS Statistics, Version 28 (IBM Corp., Armonk, NY, USA). Continuous variables (age, stone size, Hounsfield units, operative time) were analysed using Welch's t-test and expressed as mean ± standard deviation (SD). Categorical outcomes (overall and subgroup SFRs) were analysed using the Chi-square test, which is mathematically equivalent to the pooled two-proportion Z-test for large balanced samples. Subgroups with small sample sizes (stones ≥15 mm) were analysed using Fisher's exact test. Effect sizes were presented as Relative Risk (RR), Odds Ratio (OR), and 95% Confidence Intervals (CI). All hypothesis testing was two-tailed, with p < 0.05 considered statistically significant. Directional (one-tailed) analysis was initially explored for superiority hypotheses; these results are noted where clinically relevant. This statistical framework ensures precision and reproducibility in line with journal standards.
Baseline characteristics were comparable between groups, with no statistically significant differences in age, stone size, or operative time (p > 0.05).
Over all demographics and procedural details are mentioned in Table 1. In Group A, 102 patients underwent 113 procedures with 132 renal stones: 56 stones (42.42%) in the lower pole, 14 (10.6%) in the upper pole, 21 (15.9%) in the mid pole, and 41 (31.06%) in the renal pelvis. Twenty-four patients (24/102) had multiple stones in multiple calyces. In Group B, 102 patients underwent 115 procedures with 130 renal stones: 46 stones (35.3%) in the lower pole, 36 (27.6%) in the renal pelvis or ureteropelvic junction, 24 (18.4%) in the mid pole, and 24 (20%) in other locations. Twenty-six patients (26/102) had multiple stones in multiple calyces. Details of stone distribution by location and subgroup is shown in Table 2.
|
Parameter |
Group A (100 W) |
Group B (50 W) |
P-value |
|
Number of Patients/Procedures |
102/113 |
102/115 |
NA |
|
Mean Age (Years) |
57.17±16.4 |
61.7±15.7 |
>0.05 |
|
Mean Stone Size (mm) |
14.8±9.08 |
13.8±8.13 |
>0.05 |
|
Mean Operative Time (min) |
79.4±39.0 |
73.0±31.9 |
>0.05 |
Table 1 Overall demographics
*The two groups were comparable in baseline parameters. All differences were statistically non-significant
|
Subgroup |
n (Group A, 100 W) |
n (Group B, 50 W) |
P-value |
|
Hard Stones (HU ≥900) |
59 |
50 |
>0.05 |
|
Lower-pole Stones |
54 |
42 |
>0.05 |
|
Stones ≥10 mm |
84 |
75 |
>0.05 |
|
Stones ≥15 mm |
41 |
35 |
>0.05 |
Table 2 Subgroup demographics
* Mean stone size, HU, and operative times did not differ significantly in any subgroup, confirming comparability. HU = Hounsfield units.
** Stone Distribution: Group A (102 patients, 132 stones): Lower pole 56 (42.42%), upper pole 14 (10.6%), mid pole 21 (15.9%), renal pelvis 41 (31.06%). Multiple stones in multiple calyces: 24/102 (23.5%). Group B (102 patients, 130 stones): Lower pole 46 (35.3%), upper pole/renal pelvis/PUJ 36 (27.6%), mid pole 24 (18.4%), other 24 (20%). Multiple stones in multiple calyces: 26/102 (25.5%).
Stone free rates by laser type and subgroup are summarized in Table 3. The overall SFR was higher in the 100 W group (90.2%, 92/102) compared to the 50 W group (81.4%, 83/102; p = 0.021, RR 1.11, 95% CI 1.02–1.22; OR 2.07, 95% CI 1.08–3.97). Subgroup analyses revealed significant improvement with high power laser for Hard stones (> 900 HU) 83.1% (49/59) in Group A vs. 68.0% (34/50) in Group B (p = 0.043, RR 1.22, 95% CI 1.02–1.47; OR 2.21, 95% CI 1.02–4.76). For Stones ≥10 mm the 100 W laser achieved superior clearance (89.3%, 75/84) in Group A vs. (76.0%, 57/75) in Group B (p = 0.026, RR 1.17, 95% CI 1.01–1.36; OR 2.63, 95% CI 1.10–6.29). Lower-pole stones clearance showed a trend towards significance: 81.5% (44/54) in Group A vs. 64.3% (27/42) in Group B (p = 0.050 two-tailed, showing trend towards significance; one-tailed analysis p = 0.025, RR 1.27, 95% CI 1.0–1.61; OR 2.39, 95% CI 0.99–5.75). Stones ≥15 mm showed no significant difference (63.4%, 26/41) in Group A vs. 57.1% (20/35) in Group B (p = 0.56, Fisher's exact test, not statistically significant; RR 1.11, 95% CI 0.80–1.55; OR 1.29, 95% CI 0.55–3.00). Acceptable SFR (residual <5 mm) was achieved in 92% (94/102) of Group A and 84.4% (86/102) of Group B (p = 0.082, trend towards significance).
|
Subgroup |
High-Power (100 W) |
Low-Power (50 W) |
P-value |
RR (95% CI) |
OR (95% CI) |
|
Overall Complete SFR |
90.2% (92/102) |
81.4% (83/102) |
0.021 |
1.11 (1.02–1.22) |
2.07 (1.08–3.97) |
|
Acceptable SFR (<5 mm) |
92% (94/102) |
84.4% (86/102) |
0.082* |
1.09 (1.01–1.18) |
2.01 (0.98–4.12) |
|
Hard Stones (HU ≥900) |
83.1% (49/59) |
68.0% (34/50) |
0.043 |
1.22 (1.02–1.47) |
2.21 (1.02–4.76) |
|
Stones ≥10 mm |
89.3% (75/84) |
76.0% (57/75) |
0.026 |
1.17 (1.01–1.36) |
2.63 (1.10–6.29) |
|
Lower Pole |
81.5% (44/54) |
64.3% (27/42) |
0.050** |
1.27 (1.0–1.61) |
2.39 (0.99–5.75) |
|
Stones ≥15 mm |
63.4% (26/41) |
57.1% (20/35) |
0.56 |
1.11 (0.80–1.55) |
1.29 (0.55–3.00) |
|
Subgroup |
High-Power (100 W) |
Low-Power (50 W) |
P-value |
RR (95% CI) |
OR (95% CI) |
|
Overall Complete SFR |
90.2% (92/102) |
81.4% (83/102) |
0.021 |
1.11 (1.02–1.22) |
2.07 (1.08–3.97) |
|
Acceptable SFR (<5 mm) |
92% (94/102) |
84.4% (86/102) |
0.082* |
1.09 (1.01–1.18) |
2.01 (0.98–4.12) |
|
Hard Stones (HU ≥900) |
83.1% (49/59) |
68.0% (34/50) |
0.043 |
1.22 (1.02–1.47) |
2.21 (1.02–4.76) |
|
Stones ≥10 mm |
89.3% (75/84) |
76.0% (57/75) |
0.026 |
1.17 (1.01–1.36) |
2.63 (1.10–6.29) |
|
Lower Pole |
81.5% (44/54) |
64.3% (27/42) |
0.050** |
1.27 (1.0–1.61) |
2.39 (0.99–5.75) |
|
Stones ≥15 mm |
63.4% (26/41) |
57.1% (20/35) |
0.56 |
1.11 (0.80–1.55) |
1.29 (0.55–3.00) |
Table 3 Stone-free rates (sfr) by study group and subgroup analysisp
* Trending towards significance; p > 0.05 = not statistically significant.
** Two-tailed p = 0.050 (borderline); one-tailed directional analysis p = 0.025 (statistically significant).
Note: SFR = Stone-free rate; RR = Relative risk; OR = Odds ratio; CI = Confidence interval; HU = Hounsfield units.
Operative times and efficiency metrics are presented in Table 4. Mean operative times were comparable between groups: overall 79.4 ± 39.0 minutes (Group A) vs. 73.0 ± 31.9 minutes (Group B, p > 0.05). No significant differences were observed in operative times for hard stones, lower-pole stones, stones ≥10 mm, or stones ≥15 mm (all p > 0.05).
|
Subgroup |
Group A (100 W) Mean±SD |
Group B (50 W) Mean±SD |
P-value |
|
Overall (range) |
79.38±38.98 (24–180) |
73.0±31.9 (14–175) |
>0.05 |
|
Hard Stones (range) |
77.4±36.0 (18–165) |
77.94±35.26 (20–172) |
>0.05 |
|
Lower Pole Stones (range) |
72.31±37.2 (15–160) |
78.99±33.83 (12–168) |
>0.05 |
|
Stones ≥10 mm (range) |
88.7±40.37 (33–180) |
85.62±30.62 (36–175) |
>0.05 |
|
Stones ≥15 mm (range) |
104±40.78 (40–180) |
100.5±28.78 (60–175) |
>0.05 |
Table 4 Operative time (minutes) by study group and subgroup
* P-value >0.05 indicates not statistically significant. Mean operative time was comparable between groups (p > 0.05) for all subgroups. SD = Standard deviation.
Postoperative outcomes and complications are summarized in Table 5. Day-case discharge rate was significantly higher in Group A (83/112, 74.1%) compared to Group B (63/115, 54.7%, p < 0.05), with a mean hospital stay of 0.4 days (range 0–7) in Group A versus 0.9 days (range 0–7) in Group B (p < 0.05). Postoperative complications were generally comparable between groups. Sepsis or fever occurred in 4/112 (3.5%) patients in Group A and 6/115 (5.2%) in Group B (p > 0.05). Intensive Care Unit admission was required in 0 patients in Group A versus 2/115 (1.7%) in Group B (p > 0.05). In Group A who had sepsis or temperature, all were female, 1 had 18mm stone and rest had ≥ 2.0cm stone and all but one took longer than 90 minutes. Also 1 had Stenotrophomonas in urine from kidney and infected 3 cm stone, 1 had recent septic shock due to infected stone 6 weeks pre op needing ITU admission, 1 had recurrent uti’s and multiple allergies. While in group B those who had sepsis or temperature 1 had VRE in the urine from the right kidney, 1 had h/o recurrent uti’s, 1 had 2 episodes of sepsis and grew ecoli in urine from left kidney twice and fungus in urine from right kidney and had h/o recurrent uti’s. 1 had high residuals and foul smelly urine in bladder and partial staghorn. In this group 4 episodes were in females and 2 in males. Readmission within 6 weeks occurred in 3/113 (2.7%) in Group A and 3/115 (2.6%) in Group B (p > 0.05). Stricture developed in 1/113 (0.88%) in Group A and 1/115 (0.8%) in Group B (p > 0.05). Deep vein thrombosis occurred in 0 patients in Group A and 1/115 (0.87%) in Group B (p > 0.05); this patient was prone to recurrent thromboembolic events. Mean stent duration was 17.06 ± 7.9 days (range 0–36) in Group A and 15.2 ± 7.2 days (range 0–36) in Group B (p > 0.05). No blood transfusions were required, and haematuria was mild or absent in both groups. All procedures were completed without conversion or device malfunction, except in one case where the high-power laser malfunctioned.
|
Outcome |
Group A (100 W) |
Group B (50 W) |
P-value |
|
Day Case Rate |
83/112 (74.1%) |
63/115 (54.7%) |
<0.05* |
|
Extended Day Case (1 night) |
16/112 (14.2%) |
31/115 (26.9%) |
<0.05* |
|
Mean Hospital Stay (days) |
0.4 (0–7) |
0.9 (0–7) |
<0.05* |
|
Sepsis/Fever |
4/112 (3.5%) |
6/115 (5.2%) |
>0.05 |
|
ITU Admissions |
0/112 (0%) |
2/115 (1.7%) |
>0.05 |
|
Readmission (Any Cause) |
3/113 (2.7%) |
3/115 (2.6%) |
>0.05 |
|
Stricture |
1/113 (0.88%) |
1/115 (0.8%) |
>0.05 |
|
DVT |
0/113 (0%) |
1/115 (0.87%) |
>0.05 |
|
Mean Stent Duration (days) |
17.06±7.9 (0–36) |
15.2±7.2 (0–36) |
>0.05 |
Table 5 Postoperative outcomes and complications
* Statistically significant (p < 0.05).
** Hospital stay was significantly shorter in Group A. Day-case discharge rate was significantly higher in Group A. All procedures were completed without conversion or device malfunction except in one case where the high-power laser malfunctioned. No blood transfusions were required in either group. Haematuria was mild or absent in both groups. ITU, intensive care unit; DVT, deep vein thrombosis
The findings from this study reinforce the advantages of high-power, high-frequency Holmium laser in the treatment of renal stones. The ability to adjust pulse modulation and frequency settings allows for better control over fragmentation and dusting, ultimately improving stone-free rates. These laser systems offer broad energy and frequency settings (up to 100 Hz) for stone dusting, fragmentation, pop-dusting1 and pop-corning.2
Early laser machines were low-power, low-frequency systems generating frequencies of only 10–18 Hz. The 50 W Auriga XL laser used in this study generated a maximum frequency of 12 Hz and maximum energy of 1 J for fragmentation and 0.3–0.4 J at 12 Hz for dusting using a 230 µm fibre. With this system, effective dusting was difficult and impossible for hard stones; therefore, fragmentation, popcorning,3 and basketing were the primary treatment modalities.
The advent of high-power laser technology (100–120 W) with pulse modulation has revolutionized stone management, enabling significant improvement in stone dusting by utilising low energy (0.2–0.4 J) and high frequency settings (50–100 Hz) as demonstrated in earlier studies.4 Retropulsion of stones has been reduced through pulse modulation. Long-pulse-width technology further reduces stone retropulsion and facilitates laser efficacy. MOSES technology exemplifies this advance, providing split-pulse energy delivery with creation of a small vapour bubble around the stone through which the second laser pulse travels, resulting in further reduced retropulsion and improved fragmentation.5
In this study, significantly improved stone clearance was achieved with the high-power, high-frequency laser machine while maintaining low complication rates and quick recovery. High-frequency settings of 30–53 Hz and energy of 0.3 J were used for dusting, and 1 J at 30–53 Hz was used for fragmentation and pop-dusting, resulting in superior overall SFR (90.2% vs. 81.4%, p = 0.021).
While the overall SFR improvement was significant, the lower-pole subgroup showed a borderline two-tailed p-value (0.050); this reached significance under directional (one-tailed) hypothesis testing, consistent with the expected superiority of the 100 W laser. This pattern suggests a genuine effect likely limited by subgroup sample size. From a statistical perspective, the combined use of Welch's t-test, Chi-square, and Fisher's exact tests ensures robust, valid inference for both continuous and categorical data, aligning with established biostatistical standards.
These results affirm the role of high-power Holmium lasers as the preferred technology for all renal stones, especially complex cases. Although operative time reduction was not demonstrated, the high-power laser generated finer fragments and dust compared to the low-power laser, resulting in better stone clearance and reduced basketing requirements.
The rise in intrarenal temperature during high-power laser lithotripsy has been a topic of discussion in recent years. High intrarenal temperature can adversely affect the kidney through destruction of nephrons.6,7 Earlier studies have shown that the risk of temperature increase can be reduced by intermittent laser firing and increased irrigation flow with room temperature or cold saline.4 However, increased irrigation flow can elevate intrarenal pressure, potentially resulting in intrarenal reflux or forniceal ruptures—a risk that can be mitigated by using an ureteral access sheath of adequate diameter to permit continuous outflow of irrigation fluid.8
Compared to traditional ureteral access sheaths, suctioning ureteral access sheaths offer advantages including higher SFR at 1 day post-operatively, lower incidence of infectious complications, and shorter operative times.9 A temperature threshold of 43°C has been suggested for cellular thermal injury,10 with temperatures above 56°C causing cell death.10,11 The novel addition of flexible and navigable suction-assisted ureteral access sheaths allows active control of intrarenal pressure by adjusting negative vacuum pressure and vent opening, maintaining clear visibility and reducing thermal and intrarenal pressure-related complications.12 This technology may be effective even with low-power lasers by reducing residual stone fragments (Figure 1– 6).
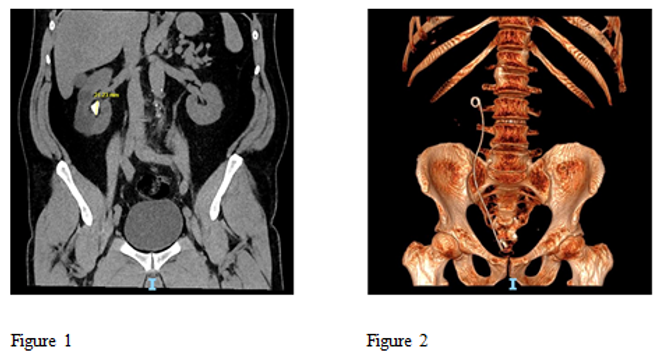
Figure 1 Pre op Large 21 mm Partial staghorn stone and Figure 2 Post op 3 D reconstructed CT scan showing complete clearance of stone after single procedure using High Power Holmium Laser.
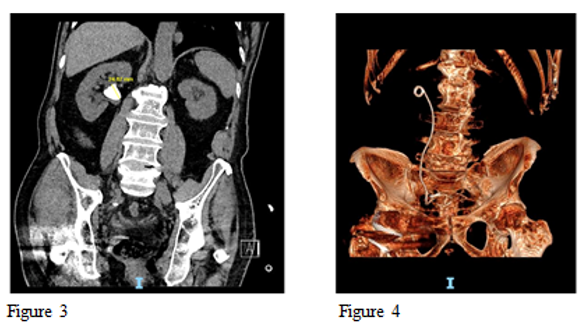
Figure 3 Pre op Large renal pelvic stone 24mm and Figure 4 Post op 3d reconstructed CT scan showing complete clearance of the stone after single procedure using High Power Holmium Laser.
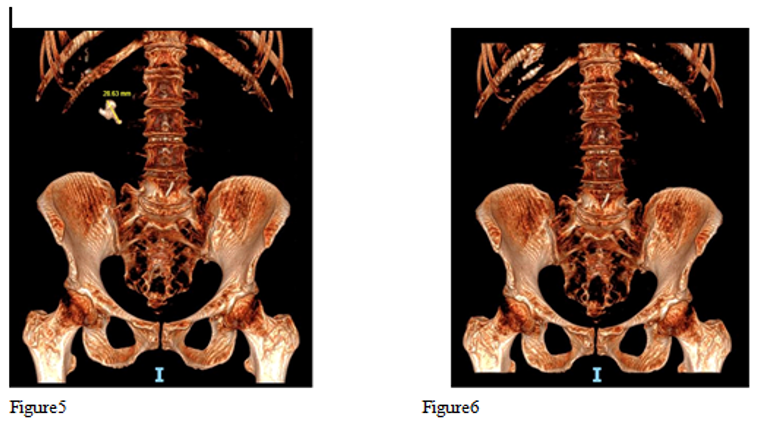
Figure 5 Pre op 3D Reconstructed CT scan image showing large partial staghorn stone and Figure 6 Post op 3D reconstructed image showing complete clearance after single procedure using High Power Holmium Lase
In this series, high-power, high-frequency laser RIRS was also used for larger stones greater than 2 cm with favourable anatomy of the pyelocalyceal system or in patients unsuitable for percutaneous nephrolithotomy. When multiple stones were present, the total length was calculated (Figure 7,8). Although the maximum combined stone length treated was 65 mm, careful patient selection based on pyelocalyceal anatomy, infundibulo-pelvic angle (especially for lower-pole stones), and patient fitness is strongly recommended for any single stone ≥2 cm.
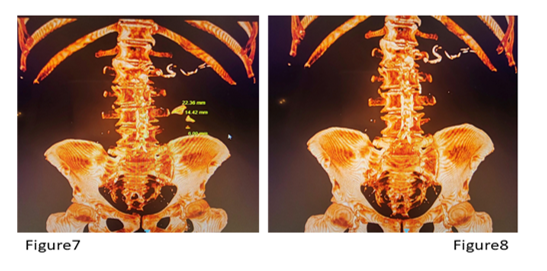
Figure 7 shows 3 dimentional Pre op CT scan with multiple stones with longest length added for total length and Figur 8 shows post op clearence following high power, high frequency laser laser.
In this study stone-free rate for stones ≥15 mm was higher in the 100 W group (63.4%) versus the 50 W group (57.1%), the 6.3% absolute difference did not reach statistical significance, likely due to small subgroup sample size. Clinically, this modest improvement may be meaningful in certain patient populations or practice settings, especially if associated with reduced need for secondary procedures. The previously published data though does suggest statistically significant stone free rate can be achieved in less time with High Power 120 W Ho:Yag laser.13
Strengths and Limitations: Strengths of this study include single-surgeon and single-centre uniformity and consistent long-term follow-up over six years. Limitations include the non-randomised, retrospective analysis and smaller subgroup sizes, particularly for stones ≥15 mm. Nevertheless, the data represent real-world outcomes in a district general hospital setting and validate high-power laser efficacy and safety.
This work was registered with the hospital Clinical Effectiveness Department as a clinical audit (Audit no. 2579), using retrospectively analyzed, prospectively collected routine clinical data. Formal NHS Research Ethics Committee review was not sought, in line with local governance procedures for clinical audit. All patients signed a generic consent form for their operation.
Prior Presentation: This manuscript is original and unpublished, and is not under consideration elsewhere. A limited version of the abstract, focusing only on overall and lower-pole stones without statistical analysis or subgroup stratification, was previously presented at the SIU Congress. The current manuscript includes comprehensive subgroup analyses and statistical evaluation not included in those presentations. Formal permission has been taken from the SIU scientific office to reproduce part of the abstract published in SIU congress 2021, 2024 and 2025.
None.
The authors of this article confirmed the absence conflict of interests, financial or any other support which should be reported
None.

©2026 Ashwini. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.