MOJ
eISSN: 2379-6162


Research Article Volume 4 Issue 3
College of Medicine & Dentistry, Pakistan
Correspondence: Nasir Iqbal, MS, FRCS, Fellow Thoracic Surgery, Professor and Chief of Surgery FMH and college of Medicine & Dentistry, Shadman, Lahore, Pakistan
Received: June 23, 2015 | Published: May 17, 2017
Citation: Iqbal N, Shakir J, Bokhari ST, et al. Ewing’s sarcoma chest. MOJ Surg. 2017;4(3):63-65. DOI: 10.15406/mojs.2017.04.00074
Aim: The aim of this review article was to highlight clinical presentations diagnosis and treatment approaches of thoracic cavity Ewing’s sarcoma. This was a prospective study. Ewing’s sarcoma is the second most frequent bone tumour among children and young adults. It comprises 10-15% of all bone tumours. Ewing’s sarcoma arises intramedullary. The frequency of rib involvement follows that of the femur, the pelvic bones, the fibula, the humerus and the tibia.
Materials and methods: Between January 1999 and December 2005, 5patients of Ewing’s sarcoma chest were dealt with. The age range was 6-12years.
Results: All the 5patients had huge masses compressing the lung and other vital structures in the chest. One patient was on ventilatory support preoperatively. Thoracotomy was done in 3patients prior to chemotherapy and later on after chemotherapy in 2patients. Post operative stay range was 7-15days. No hospital mortality occurred.
Keywords: ewing sarcoma; thoracotomy
Ewing’s sarcoma is named for James Ewing (1866-1943), the American pathologist who first described it.1,2 It is a malignant tumour composed of small round cells. It originates in the bone and is characterized by the translocation of chromosomes 11 and 22. Approximately 15% of all cases of Ewing’s sarcoma arise in the chest wall, most frequently from a rib (less often from the scapula).3-6 The peak incidence occurs between the ages of 10 and 15 years. There is a slight male: female preponderance (1.6:1). Clinical presentation includes pain and swelling of weeks or months duration.7 Micro-metastatic disease is present in many at the time of diagnosis that can’t always be seen on the CT & bone scans or bone marrow tests.
The general radiological appearance is a mass in the chest wall accompanied by destruction of the bone. Ewing’s sarcoma is rarely extra osseous.8 Although bone involvement is an important characteristic, the soft tissue components may be much more extensive than the bony component. Usually the Ewing’s sarcoma is treated initially with chemotherapy, followed by surgical resection with or without radiation therapy. This malignant tumour is associated with a high rate of metastasis (75% of cases) and local recurrence. The overall 5-Year survival rate is 50%.9-11
There were five patients of Ewing’s sarcoma initially diagnosed by needle biopsy between January 1999 and December 2005, who were treated. All the patients presented with chest pain, shortness of breath and in one patient there was obvious external swelling on the chest wall. There were two females and three males. The age range was 6-12years. Three patients had to be operated immediately after the percutaneous biopsy as the respiratory distress was increasing in these patients. Two were operated after the initial chemotherapy.
All the five patients had huge masses in the chest cavity compressing the lung and other vital structures. One patient was on ventilatory support preoperatively. Thoracotomy for excision of mass and the involved rib was done in all the cases. In three patients who underwent surgery prior to chemotherapy treatment had their chemotherapy treatment later on. In one patient there was recurrence after 6months. For which Thoracotomy for excision of mass and adjacent rib excision was repeated. This patient also had additional radiotherapy treatment to the chest wall. The post operative stay range was 7-15days. There were no complications. No hospital morality occurred.
Chest wall tumours of primitive neuroectodermal origin (PNET, Ewing’s sarcoma) are rare and have a poor prognosis. Multimodality therapy has improved survival results, and long-term survival is possible. Evidence suggests that roughly 75% of the patients with Ewing’s sarcoma present with localized disease. For those who present with metastatic disease the cure rate is 20 to 30 percent. The metastatic disease is to the lungs, bone and bone marrow. Complete resection of the primary tumour of the chest wall is accomplished more frequently in resections performed after initial adjuvant chemotherapy compared with primary resections. Although thoracic tumours are frequently large and primary resection is often attempted. In our study we had to do primary resection in three cases as they were having acute respiratory distress after the percutaneous biopsy from the chest. It was probably due to the bleeding in the large tumours after the biopsy, which was later confirmed at the time of thoracotomy. Delayed resection resulted in a significant decrease in the proportion of patients requiring radiation therapy to the chest. Radiation therapy has been associated with the significant incidence of secondary tumours, pulmonary fibrosis and cardiomyopathy in these patients. Evaluation of the primary and metastatic sites is performed approximately every 10-12 weeks while on chemotherapy and at 3 to 4 months intervals during the first year following the completion of therapy. Reevaluations are spaced to gradually or 5-6 years following the completion of therapy. Recurrence of primary disease is the major risk in the first 10 years from the time of diagnosis. Minimally invasive surgery has a role in planning the resection of malignant chest wall tumours in the pediatric population. 12 (Figures 1-4).
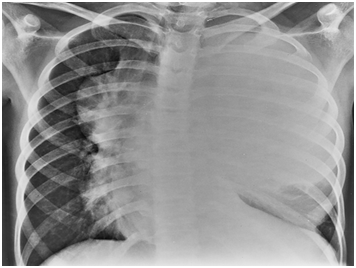
This article demonstrates the most effective and immediate treatment in case of Ewing’s sarcoma chest. The three cases that had to be operated urgently before the chemotherapy survived and received chemo-radiation later on thus signifying the importance of timely and proper surgery. Referral to proper hospital is mandatory who cater these kind of cases.
None.
The author declares no conflict of interest.

©2017 Iqbal, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.