MOJ
eISSN: 2379-6162


Case Report Volume 11 Issue 2
1Continuous Admission Unit. High Specialty Medical Unit Hospital of Specialties “Dr. Antonio Fraga Mouret”, National Medical Center “La Raza” Mexican Institute of Social Security, Mexico
2Intensive Care Unit. High Specialty Medical Unit Infectology Hospital “Dr. Daniel Mendez Hernandez, National Medical Center “La Raza” Mexican Institute of Social Security. Mexico City
3Emergency Unit. High Specialty Medical Unit General Hospital “Dr. Gaudencio González Garza”. National Medical Center “La Raza” Mexican Institute of Social Security, Mexico
4Emergency Unit. General Hospital of Zone 197 Texcoco. Mexican Social Security Institute, Mexico
5Emergency Unit. ISSSTE TACUBA General Hospital. Institute of Security and Social Services for State Workers, Mexico
Correspondence: Ignacio Alberto Mendez de Jesus, Address: Calle lago chapa number 34, colonia Anahuac I, Mexico City, ZIP Code: 11320, Tel 55 35 06 93 69
Received: April 25, 2023 | Published: May 8, 2023
Citation: Gutierrez EMP, de Jesus IAM, Sanabria-Cordero D, et al. Approach to hypoxemia in the emergency department in a clinical case of Eisenmenger Syndrome. MOJ Surg. 2023;11(2):58-61. DOI: 10.15406/mojs.2023.11.00223
There is a wide variety of pathologies that can cause hypoxia with different pathophysiological mechanisms, and therefore with different response to treatment with supplemental oxygen, it is essential for emergency care physicians to know the different pathologies causing hypoxia to be able to adequately and direct the treatment , in this paper we discuss the case of a patient with syncope and hypoxia who reaches the emergency department, which mechanism of hypoxia is a consequence of an interatrial short circuit, reaching the conclusion of Eisenmenger syndrome, which is a pulmonary vascular disease, with elevation of pulmonary resistance in patients with a history of heart disease with short circuit from left to right. This pathology is of vital importance that its identification is made in initial stages, with the aim of preventing evolution to the point where medical treatment is insufficient, and as in the case to mention, the only remaining option would be lung and heart transplantation.
Keywords: Eisenmenger syndrome, congenital heart disease, hypoxia, diagnosis, treatment
There are different physiopathological mechanisms that cause hypoxia, each one with a variable response to the administration of supplemental oxygen, they are grouped into causes that cause a decrease in the fraction of inspired oxygen, those that produce hypoventilation, alteration in diffusion, in the ventilation-perfusion ratio, short circuits, whether at the pulmonary or cardiac level, and finally alterations in oxygen transport. Whenever a patient enters the emergency area from the observation of the patient, interrogation and physical examination, causes of hypoxia must be ruled out, in order to reach an adequate diagnosis and therefore an adequate response to the treatment established. The clinical case of a patient who was admitted with syncope and hypoxia in the midst of a health contingency caused by SARS-COV-2 who did not improve oxygen saturation by oximetry despite supplementary intake with a 10-liter reservoir mask is presented, where by questioning , physical examination and ultrasound scanning, probable causes of hypoxia are ruled out until the cause of cardiac shunt is suspected as the etiology of the clinical picture, later it is confirmed by means of contrasted tomography, electrocardiogram, lung scintigraphy and echocardiogram, it is concluded that a Eisenmenger's syndrome.
We present a 29-year-old female patient, with no history of chronic-degenerative diseases, 12-year-old Gynecology-Obstetric history, pregnancies: 2, cesarean section: 1, Abortion: 1 in February 2021, refers to a murmur detected since childhood that was never cause of complications or study. She began her condition in February 2021 with palpitations, progressive edema of the lower limbs, progressive dyspnea up to paroxysmal nocturnal dyspnea and orthopnea. Upon directed questioning, she denies angina, palpitations, fainting, or syncope. He was admitted by the Cardiology service in a tertiary hospital with a 3-day stay (April 7 to 9, 2021) where a transthoracic echocardiogram was performed (04/08/21) with the following results: Mitral and aortic valve: without insufficiencies, leaflets with normal characteristics, tricuspid regurgitation velocity 503 cm/sec (5.03 m/sec), Pulmonary Valve: ring 33 mm, trunk 36 mm, right branch 19 mm, left branch 20 mm, moderate insufficiency Maximum velocity 133 cm/sec, Maximum gradient 7.14 mmHg, Left ventricle: normal global and segmental motion at rest, concentric remodeling geometry, 70% ejection fraction, Right ventricle: TAPSE (tricuspid annular plane systolic displacement) 16 mm, dilated and hypertrophic ventricle with wall hypokinesis free, systolic dysfunction, right atrium 51x41, area 19.3 cm. Pulmonary artery systolic pressure 106 mmHg.
Discharged home with a diagnosis of NYHA III heart failure and pulmonary hypertension of etiology to be determined, with treatment based on sildenafil 25 mg orally every 24 hours, furosemide 40 mg orally every 12 hours, and rivaroxaban 15 mg orally every 24 hours , with reference to the cardiology clinic to continue the study protocol externally. However, on 05/20/21 he was admitted directly to the shock area due to syncope, where the following vital signs were obtained: BP 113/73 mmHg, HR 52 bpm, RR 22 rpm, SpO2 70%. Accompanying relatives refer prior to loss of alertness, I present general malaise with palpitations.
On directed physical examination, no neurological alterations, grade I jugular venous engorgement, reinforcement of the second sound in the pulmonary focus, at the pulmonary level with adequate respiratory noise, without rales or wheezing, without integration of pleuropulmonary syndrome, initial oximetry saturation of 70%, with oxygen by nasal cannulas at 3 liters per minute increased to 75%, managing to increase up to 85% when using oxygen at 10 liters/minute, tachypnea, without use of respiratory accessory muscles, fluent speech, extremities without edema, capillary refill in 4 seconds.
Due to the presence of hypoxemia, non-responsive to supplemental oxygen, in the context of the SARS COV 2 pandemic, the BLUE protocol was applied, where the findings were evidenced: Pulmonary window with adequate pleural sliding in the 4 explored regions of each hemithorax, presence of lines A in the 4 regions of each hemithorax with the presence of 1 to 3 B lines, absence of PLAPS (posterolateral alveolar pleural syndrome), right ventricular dilation in the cardiac window in greater relation to the left ventricle and hypokinesia of the right ventricle free wall, absence of pericardial effusion, absence of intracavitary thrombi and in the pelvic limbs without data suggestive of acute or chronic venous thrombosis.
As part of the diagnostic approach for syncope, an electrocardiogram is obtained (Figure 1).1 A chest x-ray is also obtained (Figure 2).

Figure 1 12-lead electrocardiogram with sinus rhythm, electrical axis 160°, HR 59 bpm, PR segments 160 msec, QRS 120 msec, QT 456 msec, rsR morphology in V1 to V3, negative T wave of asymmetric branches from V1 to V6, incomplete right bundle branch block.
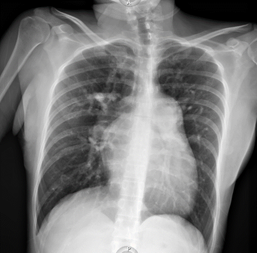
Figure 2 Anteroposterior chest X-ray: elongated arch of the right atrium, elongated pulmonary arch, dilated right ventricle, LUPI index 34 mm, prominence of the right pulmonary bronchus, venous capillary hypertension, absence of pleural effusion.
Upon regaining alertness, the patient comments that a study was carried out that has not been assessed by the treating physician: Contrasted cardiac tomography on 05/19/21 with a report of: atrial septal defect, patent foramen ovale 9 x 7 mm, interatrial septum defect 28x 25x 29 mm, pulmonary artery with a dilated appearance and a caliber of 44 mm, coronary arteries with little value due to movement and lack of dilation, secondary increase in lung vascularity (Figure 3).
As a diagnostic approach through physical examination and ultrasonographic monitoring with the Lung Ultrasound in Emergency (BLUE) protocol, decompensation of its functional class of heart failure was ruled out, pneumonia, pleural effusion and pericardial effusion were ruled out. Up to this point, there are 2 diagnostic possibilities due to the clinical picture that does not respond to supplemental oxygen, in the first place, pulmonary thromboembolism, however, without clinical or ultrasonographic data of venous thrombosis of extremities, with Wells and Geneva scales with a score of 0 points, for making the diagnosis unlikely; although a PESI in class IV. On the other hand, there was evidence of interatrial shunt, with dilation of the right cavities, tricuspid regurgitation and pulmonary hypertension, the diagnostic suspicion was directed towards this second option, making it necessary to rule out the first.
The following laboratories were collected: glucose 72 mg/dL, Creatinine 0.78 mg/dL, Sodium 141 mmol/L, Potassium 4 mmol/L, Chlorine 111 mmol/L, ALT 16 U/L, AST 17 U/L, LDH 352 U/L, CPK 95 UI/L, CPK-MB 35 UI/L, TP 11.5, TPT 30.7, INR 0.99, Hemoglobin 17.2 g/dL, Hematocrit 50.7%, Leukocytes 7 300 K/uL, Neutrophils 46.7%, Lymphocytes 39 %, Platelets 184 300 K/uL. To complete the study protocol in the emergency area, a chest angiotomography was performed without alterations to the passage of contrast medium in the pulmonary arteries. Lung window with preserved density and perfusion lung scintigraphy with 99 Tc-MAA (6 mCi) was performed: no sites of hypoperfusion suggestive of pulmonary thromboembolism are visualized (Figure 4).

Figure 4 Pulmonary scintigraphy, with a preserved base-vertex perfusion relationship, thus ruling out the diagnosis of pulmonary thromboembolism.
The following diagnoses were integrated: syncope of cardiogenic origin, pulmonary arterial hypertension PSAP 110 mmHg, Eisenmenger syndrome with interatrial communication with bidirectional flow predominantly from right to left (Table 1).
|
Causes of hypoxemia |
Examples |
Gradient Alveolar arterial |
Oxygen |
|
Decreased oxygen pressure |
High altitude, carbon monoxide poisoning |
Normal |
Reply |
|
Hypoventilation |
Neurological (Central nervous system disorders such as cerebral vascular event, tumors, conduction disorders such as demyelinating diseases such as Guillain Barré, neuromuscular junction disorders such as myasthenia gravis), Sedation, Obstructive sleep apnea syndrome, Obesity |
Normal |
Reply |
|
Alterations in diffusion |
Interstitial lung diseases, pulmonary edema |
increased |
partially answering |
|
Ventilation/Perfusion ratio imbalance |
Pneumonia, asthma, COPD, pulmonary embolism |
increased |
partially answering |
|
Short circuits |
Intrapulmonary (arteriovenous malformations), Intracardiac (atrial or ventricular septal defects) |
increased |
does not answer |
|
OTHER CAUSES |
Alterations in oxygen transport: anemia, alkalemia |
Variable |
Variable |
Table 1 Approach to hypoxemia
Pulmonary arterial hypertension secondary to chronic exposure of the pulmonary vasculature to a greater flow due to a pre-tricuspid lesion such as interatrial septal defect or shunts, results in the remodeling of the vascular bed, (2,3) which conditions an increase in the pulmonary vascular resistance until the Eisenmenger syndrome finally sets in Figure 5.4
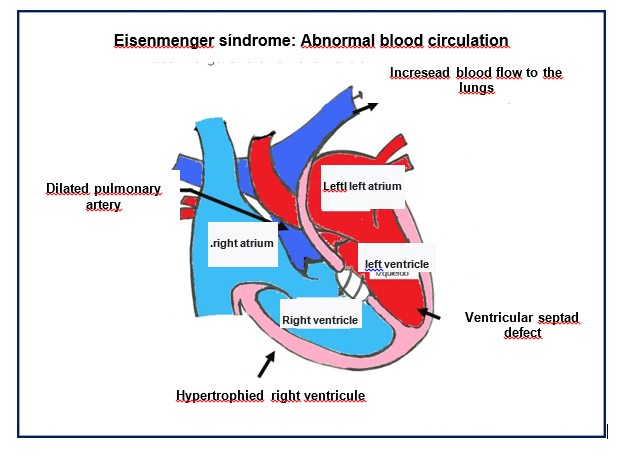
Figure 5 Main pathophysiological characteristics of Eisenmenger Syndrome (Authorized by Dr. Tabatta Gabriela Orejel Feria).
Pulmonary arterial hypertension (PAH) is defined as mean pulmonary arterial pressure (mPAP) > 20 mmHg at rest.5 The risk of developing Eisenmenger syndrome varies depending on the type of heart disease; in patients with uncorrected interatrial septal defects it is 10 to 17%, and 50% in patients with interventricular septal defects.2
The clinical presentation of Eisenmenger Syndrome is an advanced form of congenital heart disease associated with pulmonary arterial hypertension (PAH), characterized by multiorgan effects due to chronic hypoxemia, such as cyanosis and hematological changes (erythrocytosis and thrombocytopenia). The main symptoms are fatigue, dyspnea, and syncope. Hemoptysis, chest pain, and cerebrovascular accidents up to sudden death may also occur.5
Pulmonary arterial hypertension is the result of various stimuli that cause endothelial dysfunction, inducing the contraction and proliferation of smooth muscle cells. Sustained vasoconstriction of the vessels causes dysfunction of the pulmonary artery endothelial cells, causing a reduction in the production of vasodilators such as prostacyclin and nitric oxide and an increase in the production of endothelin-1 (ET-1) and thromboxane A2.6
The vasoconstriction, in situ thrombosis and the abnormal vascular remodeling described condition the obstruction of the pulmonary vessels at the microvascular level, which increases precapillary vascular resistance and pulmonary arterial pressure,2 chronic right ventricular failure occurs due to increased preload, increased wall tension, decreased contractility, increased ventricular interdependence, decreased left ventricular filling, ultimately leading to decreased cardiac output, organ dysfunction, and death.7
Supplemental oxygen does not modify the survival of patients with Eisenmenger syndrome,2 it can be used in cases in which arterial oxygen saturation increases and symptoms decrease, although its level of evidence is IIa C.5
Pregnancy is not recommended in women with this condition, since its presence is associated with a high incidence of abortion, premature births and a maternal mortality of 50%,2 contraceptive methods with levonorgestrel should be recommended, avoiding estrogens due to its association with thromboembolic events or methods such as the intrauterine device.5 Its objective is the dilation of the pulmonary vascular system (Table 2).8–10
|
Mechanism of action |
Calcium antagonists |
Prostacyclin analogs |
Endothelin receptor antagonists |
Phosphodiesterase inhibitors |
Guanylate cyclase stimulators |
Prostaglandin E1 |
|
Route of administration |
orally |
inhaled iloprost Intravenous epoprostenol |
orally |
orally |
orally |
intravenous |
|
Dose |
Nifedipine 120-240 mg/day Diltiazem 240-720 mg/day |
Epoprostenol (by central route 2 - 40 ng/kg/min) Iloprost 20 mcg/1 ml 6 times a day |
125 mg every 12 hours |
20 mg every 8 hours |
2.5 mg every 8 hours |
Start: 0.05 – 0.1 mcg/kg/min. Maximum 0.4 mg/kg/min
Maintenance: 0.01 - 0.05 mcg/kg7min |
|
Risk in pregnancy |
Amlodipine (C) Nifedipine (C) Diltiazem (C) |
Epoprostenol(B) iloprost |
Bosentan (X) |
Sildenafil and Tadalafil (Lack of data, recommended not to use) |
Riociguat (Lack of data, recommended not to use) |
Alprostadil (B) |
Table 2 Treatment with drugs
Congenital heart diseases such as Eisenmenger Syndrome usually condition the appearance of pulmonary arterial hypertension due to flow overload due to the shunt, which leads to right ventricular cardiac dysfunction, the patient's condition is triggered by pregnancy since plasma volume increases, increasing cardiac output and the refore worsening ventricular cardiac dysfunction leading to decompensation of their heart failure and the rest of the complications already mentioned, the prognosis in patients with this pathology depends on diagnosis and treatment from early phases, since in advanced phases the only treatment is a heart and/or lung transplant, but in advanced cases the treatment is only symptomatic; in the case of young women, the recommendation to avoid pregnancy due to a high risk of morbidity and mortality.
None.
The authors declare no Conflicts of interest.

©2023 Gutierrez, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.