MOJ
eISSN: 2379-6383


A risk assessment is an important step in protecting your workers and your business, as well as complying with the law. It helps you focus on the risks that really matter in your workplace especially the ones with the potential to cause real harm. In many instances, straightforward measures can readily control risks. There is not the only one way to do a risk assessment, but many methods that work well, particularly for more complex risks and circumstances. The overall principles of risk assessment process included Identify the hazards, Decide who might be harmed and how, Evaluate the risks and decide on precautions, Record your findings and implement them, Review your assessment and update if necessary. To streamline through this process, applicable clinical risk assessment tools required. All tools have their own challenges and strengths but, they should be intended to encourage greater use at practice level, and increased awareness and Understanding of risk assessment at all levels. Therefore, we must make assessment tools closer to clinical language in health system as most of these models originate of industry.
Keywords: assessment, adverse event, risk assessment models, healthcare
AE, adverse event; CIR, critical incident reporting; PSRS, patient safety reporting systems; FTA, fault tree analysis; RCA, root cause analysis; RBS, risk breakdown structure; FHA, functional hazard analysis; SSHA, subsystem hazard analysis
Risk assessment is the processes used to determine risk management priorities by evaluating and comparing the level of risk against organizational standards, predetermined target risk levels or other criteria. According to this definition by Australian Pocket Guide to Clinical Risk Management, risk assessment contained two parts of risk analysis and risk evaluation. Risk analysis determines severity and probability of risks. Risk evaluation prioritized list of risks for further action.1,2 The output of risk assessment leads clinical management to focus on critical point of any process or systems. Healthcare organizations could increase the extent to which they learn from defects. This could be obtained by risk assessment models. Both prospective retrospective tools could be applied for risk assessment and learning from defects. Johns Hopkins Hospital defined learning as reducing the probability that a future patient will be harmed. As known before, clinicians often recover from mistakes by reducing risks to the patient who suffered a defect. They believe that while we need to recover, we also need to learn or reduce risk to future patients.3,4 The purpose of clinical risk assessment tools is to provide a structured approach to help caregivers and administrators to identify the types of systems that contributed to the defect and follow-up to ensure safety improvements are achieved. For example clinical practice guidelines and risk assessment models are some useful tools to bring medical evidences into our daily clinical practice.4,5
Through risk assessment, we can understand factors contributing to an adverse event (AE), all potential AE and types, obstetrics and defensive barriers and potential prevention strategies like Training and supervision, guidelines and pathways, human factors engineering in medical device and medical system errors and finally creating and maintaining safe systems of medical care.6 But complexity of health care industry requires risk managers to have a multifaceted vision. This means risk assessment should be done respect to all dimension and details both in retrospectives models like reporting systems or root cause analysis and prospective models like FMEA. These important dimensions have shown in Figure 1. Although emphasis of this figure is on reporting systems, but we recommend that all risk assessment tools require to follow such this comprehensive framework to identify and analyzing risk.3,7 Depends on systems which you plan to assessing risk, complexity of risk management would be varied. Information infrastructure is the main criteria to choose complicity level of analyzing.
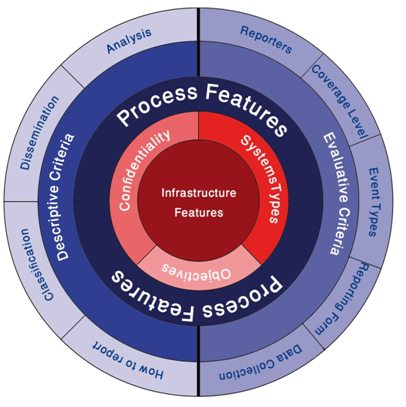
In the past two decades, researchers have emphasized the need for a system to gather information and improve hospital systems to minimize errors in health care. To achieve this goal several techniques discovered focused on Critical Incidents Survey.8 For example critical Incident Reporting (CIR) systems are any structured report of untoward and preventable outcomes of event’s which could lead to patient injury or harm. In healthcare systems, we cannot just rely on CIRs, because all incident reports’ deal with human lives. So, we need to apply a more extended scope of report systems, namely, Patient Safety Reporting Systems (PSRS). PSRS are systems used to identify safety hazards, prioritize where to focus resources, develop interventions to mitigate these hazards, and evaluate whether the interventions reduced harm. These systems can be used depending on the conditions of each country at national or regional or local level.7,9 Although most reporting systems are experienced in data collection and confidentiality of reports, more attention is required to analyzing results. As we know the most important part is risk assessment, reporting events without applying any risk assessment technique make them less valuable. Would health organization recommended analysis tools in order to assessing reported events. These are Systems analysis, Causal analysis, Risk analysis, Correlations, Trend and cluster analysis, Summaries and descriptions and Hazard identification.1 For implementing each of these analysis tools several professional techniques developed by countries, organization and other institutions. For example, John Hopkins hospital developed a risk assessment tools could be used all over the world with some changes in details to make it localized. The strength of their risk assessment tool is turning quality issues into quantifiable issues. They first of all determined what happened, then Why did it happen (Different factors contributing negatively or positively and importance of them to current and future events), Then how will you reduce the likelihood of this defect happening again (Ability to mitigate the contributing factor and Teams belief that the intervention will be implemented and executed, Strength of selected Interventions, Who will lead this effort, Follow up date), Finally, How will you know the risk is reduced (Intervention was effectively implemented and Intervention reduced the likelihood of recurrence).10 Thus, such these risk assessment tools clarify more detail for decision makers and more acceptable for caregivers.
It’s important to point out that you’re always not required to design a new methodology for risk assessment. The general framework of the methodology has proven before, but you need to consider changes made it more applicable for your organization. These methodologies are Root Cause Analysis (RCA), Failure Modes and Effective Analysis (FMEA), Fault Tree Analysis (FTA), Preliminary Hazard Analysis (PHA), Operating and Support Hazard Analysis (O&SHA), Subsystem Hazard Analysis (SSHA), Functional Hazard Analysis (FHA) and Human Error Analysis (HEA).11 The most applied risk assessment methodologies in healthcare systems are RCA and FMEA approved by JCAHO to be used as a risk assessment model.12,13 RCA is like an umbrella covering other methodologies and tools for retrospective and structured investigation of adverse incidents, near misses and sentinel events. More than 40 RCA techniques identified, such as brainstorming, cause-effect charts, “five whys” diagrams and fault trees and CIR which provide different forms of analysis.14,15 More recently, new methods has been invented which integrate both FMEA, RCA, ABS (Activity Breakdown Structure), RBS (Risk Breakdown Structure) and FMEA–waste analysis to make them applicable especially in health system as most of these models originate of industry.3,15
What is important for future studies to be focus on are information systems and safety culture. Information system has an important role in determining risk assessment tools. Countries with developed incident reporting systems have greater potentiality to applying retrospective risk assessment methods. If there is no formal and coherent errors registration system, you can be satisfied only by prospective risk assessment methods. In addition, if there is no safety culture, retrospective risk assessment methods will have deficient and unsustainable information. Thus, it’s important to provide basic infrastructure in health system for applying retrospective methods. But, we can’t also wait for AE happening and then deal with them especially in healthcare systems. So, prospective assessment tools were cost effective methods as they prevent AE happening, if they could help you to propose and implement required interventions. Finally, previous researches healthcare systems lead us to notify that both prospective retrospective risk assessment methods are essential to prevent AE happening.
None.
The author declares no conflict of interest.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.