MOJ
eISSN: 2374-6939


Case Report Volume 5 Issue 3
Department of Rheumatology, University of Qom, Iran
Correspondence: Masoumi M, Assistant professor of Rheumatology, University of Qom, Iran
Received: June 27, 2016 | Published: July 29, 2016
Citation: Masoumi M, Mollaabasi F, Noorbakhsh M, Gharib B, Ansari K,et al (2016) Wegener’s Granulomatosis and a Very Rare Complication of Cyclophosphamide: Alveolar Hemorrhage. MOJ Orthop Rheumatol 5(3): 00181. DOI: 10.15406/mojor.2016.05.00181
Granulomatosis with polyangiitis (GPA) previously known as Wegner’s granulomatosis is a small vessel vasculitis that preferentially involves capillaries, arterioles and venules, presenting as multisystemic disease classically with alveolar hemorrhage and renal insufficiency. We report a case of GPA diagnosed on history, clinical findings and supported by imaging and very high levels of Cytoplasmic antineutrophil antibodies(C-ANCA). Lung and nose biopsies confirmed the typical Histopathological findings. After taking cyclophosphamide pulse, very rare complication of cyclophosphamide has happened to the patient.
Keywords: Granulomatosis, Antineutrophil, Cyclophosphamide, Rhinoplasty, Purpura, Creatinine, Bronchoscopy
GPA, Granulomatosis with Polyangitis; ANCA, Antineutrophil Cytoplasmic Antibodies; IF, Immune Fluorescence; EIA, Enzyme Immunoassay; NIH, National Institutes of Health; CYC, Cyclophosphamide; RTX, Rituximab
A 38 year-old woman with a history of two-year refractory chronic sinusitis and nasal involvement presents to emergency department with persistent nausea and vomiting. Because the high level of Creatinine and BUN (Cr: 11.5 and Bun: 167) two-times of dialysis is performed. The biopsy of her nasal mucus was done before and it showed just some inflammatory cells. She also has a rhinoplasty 6 months ago. She complains so she complains multiple joint pain during 6 months ago that developed weakness, fatigue and bite-like lesion and petechia and palpable purpura in her extremities in 2 weeks ago before admission. She reports a nasal bloody discharge following rhinoplasty too.
She is pale, ill and toxic in general appearance and Examinations reveals vasculitis lesion on her whole body and petechial and non-palpable purpura. A mild arthritis in wrists and +3 edema in the lower limbs are significant too. Her lung HRCT-scan shows infiltration in the lower lobe of left lung so she is candidated for bronchoscopy.
In reviewing of nasal biopsy from open biopsy, the squamous mucosa is ulceronecrotic replaced by fibrinoleucocytic exudates. The submucosa represents evidence of marked vasculitis. Some vessels have thick wall, showing endothelial proliferation, occasional with occluded Lumina, tendency to fibrinoid change is also evident in an occasional vessel wall. Intervening is abundant lymphomononuclear in filtration, mixed with scattered plasma-cells and eosinophils. No granulomatous and giant-cell reaction identified (Table 1).
|
WBC:9600-PMN: 70% |
ANA: neg |
C-ANCA: >300(>18) |
P-ANCA: neg(<1.5) |
|
Hb: 5.1 |
C3: 134(90-180) |
||
|
Plt: 168000 |
ESR:114 |
UA24h: vol: 700 cc |
U/A: |
|
PT: NL |
RF:82(>20) |
B2GPl(IgG, IgM): neg |
U/C: neg |
|
Antiphospholipid panel: neg |
PPD test: neg |
UA24h: vol: 700 cc |
Table 1 Laboratory testing yields the following results
The lung biopsy taken at brochoscopic biopsy represents evidence of prominent vasculitis with fibrinoid change and interstitial inflammatory infiltration, like that of nasal mucosa, but with fewer eosinophils. The EMG-NCV is normal. According to high level of C-ANCA(more than 300), lower & upper respiratory tracts and renal involvement (RPGN[rapidly progressive glomerulonephritis]); the treatment of Wegner’s vasculitis is starting to pulse-therapy of Methyl-Prednisolone in 5 times and fallow by Prednisolone 1mg/kg/day and cyclophosphamide pulse(500mg) furthermore the level of serum creatinine is decreased.
After one-month, the pulse-therapy of cyclophosphamide is repeated, in the same day, she presented by hemorrhagic cystitis (U/A: blood: +4, RBC: many, WBC: 2-3) and one-day later she complains dyspnea and hemoptesis. Also on CBC has been pancytopenic (WBC: 2300, Hb: 4.9, MCV: 94, Plt: 42000). So she’s admitted to hospital with probability relapsing and she takes the one-time pulse of Methyl-Prednisolone and plasmaphresis is done. CXR, CT-scan and bronchoscopy are repeated and bronchoscopy reveals the hemorrhagic alveolar pattern (Figure 1).
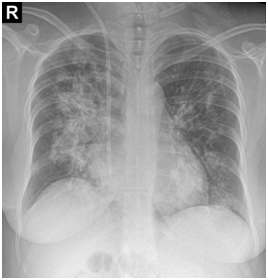
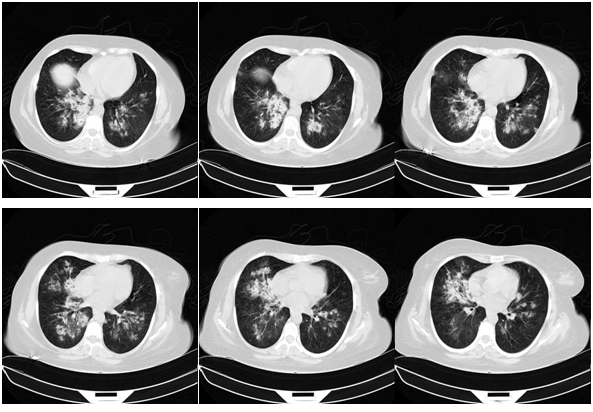
Figure 1 CXR, CT-scan and bronchoscopy are repeated and bronchoscopy reveals the hemorrhagic alveolar pattern.
Laboratory testing results show severe pancytopenia in BM, negative C-ANCA titer and normal ESR and normal CRP so the relapsing is ruled out and cyclophosphamide toxicity is considered so cyclophsphamide is stopped. One-month later we prescribe rituximab (Mabthera) 1gr (0-15d). In follow up, patient had dramatic responding to treatment and decreasing of serum creatinine level (1.3) during 6 months, being negative of C-ANCA in serum, normal ERP & CRP in recent 2 weeks, and we able to tapper Prednisolone to 7.5 mg per day.
Granulomatosis with Polyangitis (GPA), is a granulomatous disorder, often associated fibrinoid necrotizing vasculitis. The pathology of GPA comprises granulomas and necrosis, as well as the vasculitis. Pulmonary vasculitis in GPA may involve arteries, veins, and capillaries, with or without granulomatous features. Vascular necrosis begin as clusters of neutrophils within the blood vessel wall, which degenerate and become surrounded by histiocytes.1,2
The clinical features of GPA are driven by a predilection for the upper and lower respiratory tracts, as well as for kidneys. GPA may occur as a disease limited to the respiratory tract without evidence of systemic involvement, when it is referred to as limited GPA; such presentation is usually as a granulomatous disorder without vasculitis features.3 Upper airway disease is the most common presenting feature of GPA, occurring in more than 70% and eventually being present in more than 90% of patients.4,5 Nasal involvement causes mucosal swelling with obstruction, crusting, septal perforations, serosanguineous discharge, and epistaxis; a saddle nose deformity due to collapse of the cartilaginous portion of the nasal septum may develop. Sinusitis is common and will occur in more than 80% at some point during the illness.6
Pulmonary involvement will affect around 90% of patients at some point during the course of disease.4 Patients with GPA may have pleuroparenchymal lung involvement causing cough, dyspnea, hemoptysis, and/or pleuritic chest pain. The pulmonary manifestation of GPA ranges from asymptomatic lung nodules and pulmonary infiltrates to fulminant pulmonary bleeding.7 Some patients may have asymptomatic disease that is detected only after imaging.
Nearly 80% of patients with GPA, develop renal disease at some point during the course of their disease. The most feared clinical presentation is a rapidly progressive glomerulonephritis (RPGN) that manifests as proteinuria (usually non nephrotic), dysmorphic haematuria, urine cellular casts, and rapidly rising serum creatinine level.4,8
Antineutrophil cytoplasmic antibodies (ANCA) testing, has become an essential part of the diagnostic evaluation of part of the diagnostic evaluation of patients with small vessel vasculitis.9 Two types of assays of these antibodies, immune fluorescence (IF) and enzyme immunoassay (EIA), are now in common use. When immune fluorescence (IF) and enzyme immunoassay (EIA) results are considered together, ANCAs are seen in approximately 90% to 95% of generalized GPA cases during active disease, and in around 70% to 80% of cases of the limited forms.10
The classic National Institutes of Health (NIH) treatment, which consisted of 1 year of oral cyclophosphamide (CYC) (2 mg/kg daily) in combination with Prednisone (1mg/kg daily tapered over 6 to 12 months), has been modified in the last decade in an attempt to spare glucocorticoids and minimize the exposure to alkylating agents [4]. Furthermore, controlled trials have demonstrated that CYC-based and rituximab (RTX)-based regimens have comparable efficacy for the treatment of generalized GPA. Since 2010, B-cell depletion strategies have emerged as a strongly preferable alternative to CYC for treatment of GPA.11,12 Of note, among patients with relapsing disease at baseline, RTX was more efficacious than CYC. Toxicity of cyclophosphamide is in the wide range and including in Hematologic toxicity, infection, Urologic complications, malignancy, and pulmonary toxicity.13
The bladder toxicities of cyclophosphamide, hemorrhagic cystitis, and bladder cancer are related to route of administration, duration of therapy, and cumulative cyclophosphamide dose. Bladder toxicity, a particular problem with long-term oral cyclophosphamide, is largely due to acrolein, a metabolite of cyclophosphamide. It is commonly accepted that bladder toxicity can be minimized in patients receiving pulse doses of IV cyclophosphamide by administering mesna, a sulfhydryl compound that binds acrolein in the urine and inactivates it the bladder toxicities of cyclophosphamide, hemorrhagic cystitis, and bladder cancer are related to route of administration, duration of therapy, and cumulative cyclophosphamide dose. Bladder toxicity, a particular problem with long-term oral cyclophosphamide, is largely due to acrolein, a metabolite of cyclophosphamide. It is commonly accepted that bladder toxicity can be minimized in patients receiving pulse doses of IV cyclophosphamide by administering mesna, a sulfhydryl compound that binds acrolein in the urine and inactivates it.14
Cyclophosphamide-induced pulmonary toxicity occurs in less than 1% of patients. Early-onset pneumonitis 1 to 6 months after exposure to cyclophosphamide may respond to withdrawal of the drug and treatment with corticosteroids. A more insidious, irreversible, late-onset pneumonitis and fibrosis with radiographic findings of diffuse reticular or reticulonodular infiltrates may occur after treatment with oral cyclophosphamide for 1 to 13 years.15
Our patient is a case of GPA with a very rare complication of cyclophosphamide as per defined alveolar hemorrhage.16 After receiving CYC, she rapidly occurred to hemorrhagic cystitis and then in <24 hours had alveolar hemorrhage and severe pancytopenia, due to the acute and severe toxicity of cyclophosphamide, we decided to change of this regime to RTX and then she had good response.
None.
None.

©2016 Masoumi, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.