MOJ
eISSN: 2374-6939


Case Report Volume 15 Issue 6
Department of Orthopaedic Surgery and Sports Medicine, University of Kentucky College of Medicine, USA
Correspondence: Srinath Kamineni MD, Department of Orthopaedic Surgery and Sports Medicine, Kentucky Clinic, Department of Orthopaedic Surgery, 740 S. Limestone, Suite K401, Lexington, KY 40536-0284, USA, Tel +1 (859) 806 7703
Received: September 21, 2023 | Published: November 27, 2023
Citation: Gary U, Srinath K. Reverse shoulder arthroplasty in dialysis amyloidosis: a case report. MOJ Orthop Rheumatol. 2023;15(6):229-231. DOI: 10.15406/mojor.2023.15.00651
A 65-year-old male with end-stage renal disease (ESRD) on hemodialysis (HD) for 32 years presented with chronic intractable pain and progressive loss of motion in his right shoulder. The patient underwent a reverse total shoulder arthroplasty (RTSA) based on his glenohumeral arthritis, rotator cuff pathology, and debilitating symptoms. Intraoperative inspection revealed a large soft tissue mass in the area of the supraspinatus footprint and abnormal appearances of the humeral head, soft tissues, glenoid rim, and labrum. Histological specimens confirmed the diagnosis of amyloidosis and showed focal calcium pyrophosphate deposition. The patient reported improvement in pain and shoulder range of motion following the procedure in follow-up. We conclude that in the setting of long-term HD, amyloid can deposit into periarticular bone and soft tissue to contribute to patient pathology.
Level of Evidence: Level V.
Keywords: amyloidosis, shoulder arthroplasty, beta-2 microglobulin, hemodialysis, case report
DRA, dialysis-related amyloidosis; B2M, beta-2 microglobulin; HD, hemodialysis ESRD, end-stage renal disease; RTSA, reverse total shoulder arthroplasty; DIPJ, distal interphalangeal joint; TTR, transthyretin; Apo A-I, apolipoprotein A-I
Dialysis-related amyloidosis (DRA) from beta-2 microglobulin (B2M) retention and systemic amyloid fibril deposition is a rare occurrence with the use of high-flux biocompatible membranes.1 Prolonged survival on hemodialysis (HD) increases the risk for DRA, with most patients requiring surgical intervention for complications by 30 years.2 DRA skeletal complications such as bone cysts, tendinopathy, and fractures significantly impact patients' quality of life. These complications challenge surgeons to make treatment decisions for a patient population in which little information exists regarding surgical outcomes. The patient provided written informed consent for the publication of the case.
A 65-year-old male with end-stage renal disease (ESRD) on HD for 32 years presented with chronic intractable pain and progressive loss of motion in his right (R) shoulder in 2020. Radiographs and MRI identified significant glenohumeral osteoarthritis, severe tendinopathy, and a large rotator cuff tear. His medical/surgical history was extensive and included multiple comorbidities such as atrial fibrillation, pulmonary hypertension, hepatitis C, R nephrectomy for renal cell carcinoma superimposed on acquired renal cystic disease, severe secondary hyperparathyroidism, and chronic anemia. Orthopedic history included bilateral carpal tunnel releases, bilateral shoulder pain and arthritis, trochanteric bursitis, bilateral hip arthroplasties for osteoarthritis and avascular necrosis, distal interphalangeal joint (DIPJ) fusion for mallet finger, internal fixation of a fifth metatarsal fracture with non-union, and spinal stenosis.
In 2008, the patient underwent right shoulder diagnostic arthroscopy, subacromial decompression, labral debridement, subscapularis debridement, and biceps tenodesis. Arthroscopic visualization at this time revealed mild degenerative changes of the rotator cuff, absence of a full thickness cuff tear, and extensive glenoid cartilage loss with a normal appearing humeral head. Later that year, the same basic procedure was performed for his left side. Multiple rounds of bilateral subacromial steroid injections were administered for refractory pain in the years following the shoulder operations. In 2019, his pain became resistant to injection therapy and an MRI of his right shoulder was ordered. The MRI demonstrated diffuse tendinopathy throughout his rotator cuff with a full thickness tear and glenohumeral arthritis.
The patient underwent a reverse total shoulder arthroplasty (RTSA) based on his rotator cuff pathology and debilitating symptoms in 2020. A preoperative radiograph of the shoulder is portrayed in Figure 1A and B with appreciation of pathologic subacromial changes with the ultimate treatment of a RTSA depicted in Figure 1C. Intraoperative inspection revealed a large soft tissue mass, measuring 6.5 x 3.2 x 2.0 centimeters, in the area of the supraspinatus footprint (Figure 2A-2D). Additionally, abnormal appearances of the humeral head (Figure 3), soft tissues, glenoid rim, and labrum were observed. Histological specimens of intra-articular soft tissue, labrum, and synovium were obtained and confirmed the diagnosis of amyloidosis (apple-green birefringence by Congo Red Staining) and showed focal calcium pyrophosphate deposition (Figure 4A & 4B). Additionally, histology specimens of the undecalcified bone tissue showed moderate secondary hyper-parathyroid bone disease with peri-trabecular amyloid deposits (Figure 5A-5F).

Figure 1 (A) Preoperative anteroposterior, (B) preoperative Grashey, and (C) postoperative anteroposterior radiographs.
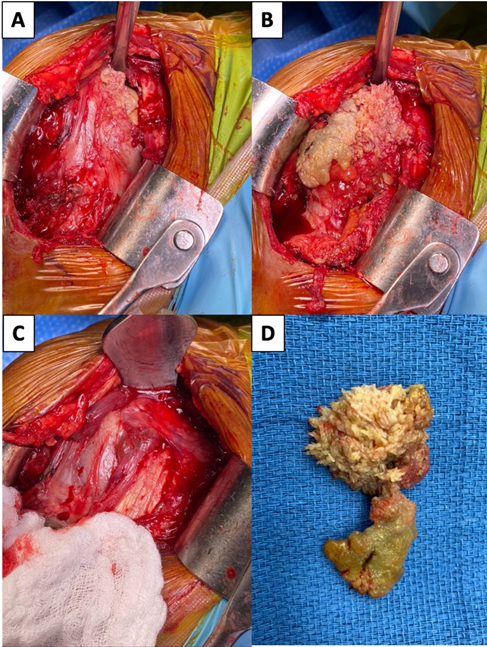
Figure 2 Intraoperative view of the pathology during (A) exposure, (B) prior to resection, (C) after resection, and (D) the specimen removed from the body.
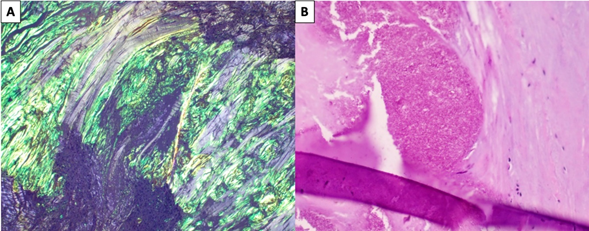
Figure 4 (A) Amyloid stained by congo red under polarized light displaying apple-green birefringence and (B) calcium-pyrophosphate crystals in soft tissue.

Figure 5 a) Undecalcified bone histology of humeral head with changes of moderate secondary hyperparathyroid bone disease, and b-f) histologic sections demonstrating thin cortex, low cancellous bone volume, low trabecular thickness and high separation; osteoid volume is increased with high osteoid surface, high osteoblast surface, very high osteoclast surface, with approximately 20% of trabecular surfaces covered by peri-trabecular fibrosis. On polarized light microscopy approximately 30% of osteoid seams exhibit woven texture, and peri-trabecular amyloid deposits are noted.
Following the procedure, the patient was prescribed non-weight bearing status in an UltraSling. In subsequent follow up visits, the patient reported improvement in pain and improvement in shoulder range of motion. Unfortunately, in 2021, the patient contracted COVID19 and developed multiorgan failure, pleural effusions, and passed away from cardiac arrest so the long-term efficacy of the rTSA could not be evaluated beyond 8 months.
In review of the literature, it is evident that B2M has a predilection for depositing into articular surfaces, synovium, and periarticular bone.3 The orthopedic clinical manifestations that arise in long-term hemodialysis patients are likely correlated to B2M deposition and need to be treated appropriately.
B2M has been shown to play a disruptive role in the cellular signaling pathway. Studies have indicated that B2M has a concentration dependent inhibition of in vitro chondrocyte growth and influences the upregulation of multiple genes leading to the formation of type III collagen, a predominant collagen found in osteoarthritis.3 Moe et al.4 described the destructive effect of B2M in synovial tissue, primarily due to the increased secretion of metalloproteinases, COX-2, VCAM-1, and other cytokines by synovial fibroblasts. Additionally, B2M has been shown to induce macrophages to upregulate secretion of TNF-alpha, IL-1, and IL-6, which offers a likely explanation for the periarticular bony destruction and cystic lesions found in long-term hemodialysis.5 Other musculoskeletal complications of B2M are broad and include deposition into vertebral discs, carpal tunnel, and articular joint spaces. Patient presentation is predictable and often involves neural compression (both in the spine and carpal tunnel) and significant joint pain and effusion.6 Crawford et al.7 localized B2M buildup in the pseudocapsule and pseudomembrane of the hip in five uremic patients on HD whose implants failed and required revision. Accelerated loosening of implants is seen in the uremic population, suggesting B2M concentration in the tissues may play a disruptive role.7 Bardin et al.8 identified B2M in articular and periarticular soft tissue in 14 patients. The clinical presentation of these patients included 10 with carpal tunnel syndrome, 3 periarticular cysts and erosions leading to fracture, and 6 effusions in the large joints of the lower limb. Amyloid deposits were consistently found in the synovium, capsule, ligaments, cartilage, and bone of the samples collected.
In regard to non-dialysis induced amyloid, Yanagisawa et al.9 found amyloidogenic proteins, transthyretin (TTR), and apolipoprotein A-I (Apo A-I) in the knee joints and soft tissues, particularly the meniscus, of patients with osteoarthritis. Interestingly, their findings show a direct positive correlation of TTR content and negative correlation with Apo A-I with age in the knee. TTRs have a detrimental impact on chondrocytes, inducing an inflammatory cascade which leads to cell death. Although OA is a complex multivariable pathology, this indicates a potential correlation between the buildup of amyloid proteins and degenerative OA.
B2M amyloidosis is an inevitable consequence of ESRD treated with long-term HD and occurs primarily due to limited extrarenal clearance.10 While techniques to improve clearance of B2M during dialysis have been proposed, none are used regularly, and none are 100% efficient. Diagnosis can only be confirmed after surgical tissue biopsy and a high index of suspicion is needed in the presence of suggestive symptoms in a long-term HD patient. A variety of symptoms are possible and can include pain in multiple joints, carpal tunnel syndrome, radiographic findings of bone cysts, fractures, and spinal cord involvement.10
Treatment options are limited and typically include symptomatic care, especially in patients unable to obtain a kidney transplant. Physicians are routinely tasked with treating the clinical manifestations of B2M amyloidosis, which have a severe impact on the patient’s quality of life. We present a case of shoulder pain, recalcitrant to conservative management, with arthritis and significant rotator cuff pathology, that improved following a reverse total shoulder arthroplasty.
Amyloid deposition is a common phenomenon in long-term HD patients. Persistent pain resulting from deposition into periarticular bone and soft tissue should be taken seriously and treated appropriately. In this particular case, the presence of articular and peri-articular changes on preoperative radiographs in the subacromion should raise suspicion for amyloid induced arthropathy. Joint pain and paresthesia are debilitating and surgical measures can be considered in patients with such DRA complications. This case of dialysis induced B2M amyloidosis of the shoulder, along with other joints, illustrates the clinical picture and surgical progression of these rare and complex patients.
None.
No potential conflict of interest relevant to this article was reported for all authors. Each author certifies that he or she has no commercial associations (e.g., consultancies, stock ownership, equity interest, patent/licensing arrangements, etc.) that might pose a conflict of interest in connection with the submitted article.

©2023 Gary, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.