MOJ
eISSN: 2575-9094


Review Article Volume 2 Issue 3
1Department of Human Reproduction, National medical university, India
2Department of Neurologist, National medical university, India
Correspondence: Kulvinder Kochar Kaur, Department of Human Reproduction, National medical university, India, Tel 91-181-9501358180
Received: April 30, 2018 | Published: May 23, 2018
Citation: Kaur KK, Allahbadia G, Singh M. Existing and prospective pathways for intervention in treatment of obesity in a novel way-a review. MOJ Drug Des Develop Ther. 2018;2(3):94-104. DOI: 10.15406/mojddt.2018.02.00035
Medical treatment of obesity is difficult as newer drugs get elucidated and may show promise but as they undergo phase trials some or other side effects appear. Initially sibutramine was used for some years and then abandoned in view of CVS side effects. Rimonabant an endocannabaninoid receptor antagonist showed great promise as far as its antiobesity properties were concerned but had to be abandoned in trial phase due to appearance of severe psychiatric side effects including suicidal effects. Now although Qsymia is approved but its use is with caution due to the effects of fetal side effects like oral cleft lip, palate and propensity for CVS side effects. Similarly lorcarserin has now been categorized as schedule IV drug in view of it causing tumor development. Tesofensine has started showing severe side effects in initial phases of phase trials. The only drug approved is orlistat for long term obesity treatment. Antidiuretic drugs like GLP1R agonists like liraglutide are other drugs of promise. Newer discoveries resulting from genetic studies of obesity and MS have identified novel molecules which act on hunger and satiety peptidergic signaling of the gut‒hypothalamic axis like ghrelin, PYY, NPY or the melanocortin system of the brain and are promising targets for future drug development. Thus the aim is to initiate drug development which not only treats obesity but also has a favorable impact on associated traits.
Keywords: antiobesity drugs, orlistat, liraglutide, rimonabant, qsymia, lorcarserin, peptidergic signaling pathways
MS, metabolic syndrome; DM, diabetes mellitus; BAT, brown adipose tissue
The incidence of obesity in developed countries has increased rapidly with parallel increase in developing countries like India and China having the highest population.1 Hence need for having effective therapy for same is important because of the associated morbidities like metabolic syndrome (MS), Diabetes mellitus type 2 (DM), hyperlipidemia and associated CVS morbidity and mortality. In our earlier reviews we have covered the medical management of obesity extensively,2,3 and further covered the novel therapies available besides the use of targeting brown adipose tissue (BAT).4‒6 Here we further update the advances in our understanding of improving medical treatment although till now one does not have any effective permanent medical therapy in contrast to the stable and long lasting effects of weight loss achieved by bariatric surgery.
Antiobesity drugs
There are various options for pharmacotherapy of obesity. Use of obesity drugs is approved for patients having a BM>30Kg/m2 or BMI>27kg/m2 along with one or more com or bidities like high BP or type 2DM. On combination with lifestyle modifications drug therapy can improve weight loss by 3‒5kg over placebo. While this is modest loss it maybe pharmacotherapy of use to add it in patients who find a plateau in losing weight with lifestyle changes alone. Importantly for this long term therapy is needed as weight loss attributed to this drug therapy is regained when the drug is discontinued.
Withdrawn drugs
Many antiobesity drugs have been introduced but despite showing promise lot had to be withdrawn. The serotonin releasing agent fenfluramine index fenfluramine were more potent obesity drugs which had to be withdrawn from the market due to reports of increased cardiac alular disease after the use of these drugs.7 Sibutramine which also affects the 5HT system and raised BP and heart rate had to be withdrawn because of association with high incidence of CVS events and stroke.8 Similarly rimonabant which showed great promise as an cannabinoids receptor antagonist in weight less had to be removed because of severe psychiatric defects including suicidal tendencies.9‒11 The new theory is that drug therapy is needed lifelong and maybe useful in weight maintenance. Tolerance can get developed and weight gain occurs even with the continued drug regimen. Intermittent dosing is being examined as a potential strategy for preventing tolerance during long term treatment.
Drugs still in use
The initial drug for obesity still in use is amphetamine derivatives like phentermine, desoxy ephedrine and diethylpropion. These drugs are centrally acting sympathomimetics with undesired effects on CNS such as agitation, hallucinations, uncontrolled movements, dizziness, difficulty in sleeping, irritability, nausea, vomiting.12 Tolerance develops very fast to these agents. As such they are approved for 12wks treatment only. As increased HR can be an adverse effect therapy with this drug class alone is not optimal for obese patients.
Role of qsymia (phen/top)
We had reviewed the reason why Qsymia TM (a combination of combined immediate release phentermine (PHEN) hydro chloride and delayed release topiramate beads (TPM) via it med release capsule got approved in July and got marketed in September 2012 but the European medicine agency (EMA) rejected the marketing authorization of PHEN/TPM because of long term cardiovascular and central nervous side effects, besides the teratogenic potential of TPM seen when used for prophylaxis of migraine patients alone and the possibility of continuation in patients in whom this combination is not recommended). Reviewed in great detail including various trials. FDA approved it and marketed it under the tradename Qsymia.13,14
Role of iorcaserin
Lorcaserin believed to act as an agonist at central serotonin subset 2C (5HT2C) receptors located on hypothalamic POMC neurons. As an Agonist of 5HT2C receptors, it is believed to reduce food intake and increase satiety relating to appetite suppression and weight loss, being a drug having serotonergic properties with trade name Belviq.15 It was initially rejected by FDA because of concerns over tumour eight losses. Ln is similar In mechanism of action to fenfluramine and dexfluramine except that it is free of any heart or heart valve side effects. 3US Trials were published for Ln‒Blossom, BLOOM, BLOOM‒DM.2,15 Despite getting rejected by the FDA earlier for fear of tumor development growth in early studies it got approved ultimately. Yet it comes as a Schedule IV drug under the Controlled Substances Act by the Drug Enforcement administration since 2013. Its marketing rights however were withdrawn by the Arena Pharman by ceuticals from Europe because of risks of tumor growth, besides development of psychiatric disorders and alular disease. Also its estimated cost of 2200$/yr makes it an expensive choice as an antiobesity agent.
Role of orlistat
Orlistat is a commonly used drug for obesity which is FDA approved .It is also approved by EMEA and is available in Europe. Orlistat inhibits pancreatic lipase, =>decreased fat uptake by the gut.16 Because it lacks a central effect on appetite and energy expenditure its effects on weight loss is relatively modest.17 Yet it has significant effects on decreasing CVS risk by reducing plasma lipids, glucose, Fatty liver disease along with systemic BP.18,19 Also it is not centrally acting and has fewer side effects in contrast to other antiobesity drugs. These side effects are mainly GIT symptoms like dyspepsia, flatulence, abdominal pain and diarrhea, but can be reduced with consumption of a low fat diet (Table 1).20
Weight Loss Drugs |
||
|---|---|---|
Drug |
Dosage |
Current Status |
Symphathomimetics Noradrenergic Agents(Amphetamine Related Drugs |
||
Diethylpropion (Tenuate)12 |
25mg before meals ,75mg slow release in morning |
Still in use |
Phentermine12 |
8mg before meals,15or 37.5mg in morning |
Still in use |
Phendimetrazine (Bontrl)12 |
35mg before meals ,105mg slow release form daily |
Still in use |
Bezphetamine (Didrex)12 |
25‒50mg 1‒3times daily in morning |
Still in use |
Noradrenergic and Serotonergic Agents |
||
Sibutramine (Meridia) |
10‒15mg daily |
withdrawn |
Tesofensine21 |
0.25mg,0.5mg,1.0mg |
in clinical trial |
Lipase Inhibitors |
||
Orlistat (Xenical)17 |
120mg tied |
in use long term |
Cannabinoid receptor antagonists |
||
Rimonabant11 |
5 or20mg |
withdrawn(psycsideeff |
Combinational Agents |
||
Phentermine/Topiramate13,14 |
3.75mg/23mg(low) ,7.5mg/46mg(mid),15/92(high) EQUIP,EQUATE,CONQ |
in use cautiously |
Bupropion/Naltrexone (Contrave) |
360/32,400/46 |
in use |
Serotonergic Agents |
||
Fenfluramine7 |
withdrawn |
|
Dex fenfluramine7 |
withdrawn‒CVS se |
|
Lorcaserin15 |
still in use, grIV category |
|
GLP1R Agonists |
||
Liraglutide24,25 |
3mg |
in use |
GLP: Glucagagon coagonist4 |
only animal studies |
|
Mc4 R Agonists autonomic side effect |
||
RM 49334,35 |
xceptRM493,trial |
|
Observational drugs |
||
PPAR β/δ46 |
Clinical trials needed |
|
CDDO‒Im47 |
animal studies only |
|
RIP14028,29 |
Animal studies, Cyt R&D dev |
|
SMRT30 |
Animal studies only |
|
Table 1 These side effects are mainly GIT symptoms like dyspepsia, flatulence, abdominal pain and diarrhea, but can be reduced with consumption of a low fat diet
Role of tesofensine
Tesofensine (NS2330) and its active metabolite (NS2360) inhibit presynaptic reuptake of the neurotransmitters noradrenalin, dopamine and 5HT and also stimulates cholinergic system indirectly. Its main mechanism of action was to reduce appetite sensations and enhance satiety in early part of weight loss besides possibly causing increased resting energy expenditure. It is a sympathomimetic of the family of sibutramine, initially investigated during the treatment of Alzheimer’s disease and parkinsonism. Tesofensine has completed phase 1 and2 trials. Astrup et al presented the results of 2 trial which showed showed that proportion of pts achieving >=5kg(49% was 59%, 87% and 91% for 0.25, 0.5 and 1mg groups respectively as compared to 29% of controls. This weight reduction was much more than those which was obtained by any of the existing drugs at least in the initial phase appetite suppression was much more.21 Neuroresearch initiated phase 3 trials, with 0.5 and 0.25mg doses lieu of serious adverse effects.22 Which was the only one endorsed by FDA and EMA. They planned a trial of 6000 patients. The commonest side effects include increased HR, hypertension and greater frequency of mood changes.
Role of glucagon like peptide 1 receptor (GLP1R) agonists
Earlier we had reviewed the role of glucagon like peptide 1receptor (GLP1R)agonists like liraglutide and eventide in detail especially the importance of using liraglutide 3mg in obesity besides their use for treatment of T2DM. GLP1 is a gut hormone which increases secretion of insulin in pancreatic β cells, gets secreted by the endocrine L cells after food intake. It has double mechanism of action i) on GIT and ii) on brain, i.e to increase the secretion of lepton leading to suppression of appetite, energy intake and a delay in gastric emptying. Long term effect is to decrease HbA1c levels and systolic BP.23 Large doses(3mg) of GLP1R agonist liraglutide causes satiety in CNS besides getting considerable long term weight loss and improve insulin sensitivity.24,25 Patients treated with liraglutide also showed a significant decrease in BP and prevalence of pre DM (84‒96%). Further we had emphasized on the co‒agonistic properties which had helped in the development of new generation of antiobesity molecules like GLP: Glucagon unimolecular coagonist. This molecule reduced body weight in obese animals to almost 30% of initial body weight in only one month. Also leptin induced body weight loss has also been shown to be considerably potentiated by the GLP: Glucagon unimolecular coagonist. Chronic administration of leptin and GLP: Glucagon give a 50% weight loss in obese mice over one month along with normalizing glucose intolerance. Further we had reviewed how the anorexigenic effects of GLPR’is obtained both through hypothalamic and two key mesolimbic reward systems namely VTA and nucleus accumbens.
Role of receptor interacting protein 140(RIP140)
Receptor interacting protein 140 (RIP140) also known as NCOR2 is a nuclear hormone co repressor, which regulates fat accumulation. It regulates with nuclear receptors like estrogen, thyroid hormone and retinoic acid receptors through C terminal receptor interacting domains (RID). It serves as a scaffold protein to recruit histone deacetylase complexes and chromatin remodeling factors.26,27 Mice with global RIP140 knockout are lean and resistant to high fat DIO and fatty acid liver disease. Silencing RIP140 in animal models cause long lasting weight loss, resistance to DIO and increased metabolic rate RNAi against RIP140 is being developed by the Cyt R&D Company for treatment of obesity and T2DM.28,29
Role of SMRT (silencing mediator of retinoid and thyroid hormone)
SMRT is another nuclear hormone co repressor. Disruption of molecular interactions between SMRT and nuclear hormone receptors=>increased adiposity and a decreased MR in genetically engineered mice.30 These studies suggest that targeting the molecular interaction between nuclear hormone receptors and then regulating cofactors may=>development of novel therapeutics which can control obesity.
Role of Mc4 receptor agonists
Mc 4 receptors have been thought to be the initial targets for efficacious therapy in monitoring obesity Various MC4 Ragonists were described which produce mark decrease in adiposity in humans and monkey.31‒35 Yet MC4 Ragonists are not used in normal therapeutic arena because of potential side effects, especially those affecting the autonomic nervous system. E.g MC4Ragonists like Melatonin II may=>hypertension and priapism.36,37 These side effects are caused by the stimulation of preganglionic autonomic neurons expressing Mc4Receptors.38 Still all MC4R agonists may not similarly change autonomic function. A small peptide RM‒493 has been shown to effectively induce weight loss in non human primates as wells humans, with no marked effects on cardiovascular function. It is in clinical trials for genetic obesity. Mc4Recept ors may be modulated by the melanocortin receptor accessory proteins (Mrap 1and Mrap 2).39,40 These Mraps may give something to differentially target the beneficial effect of Mc4Receptors actively on energy expenditure, glucose utilization and other metabolic arameters, while minimizing the adverse side effects. Recent shork has also highlighted a potential role for the melaocortin system to control BAT thermo genesis along with brown /beiging of WAT.41‒45 It is not clear how much the thermo genesis aids in regulation of body composition and glucose homeostasis, yet this data may help in the development of MC4Ragonists to fight obesity and DM.
Role of peroxisome proliferator of activated receptors beta/delta (PPARβ/δ
PPAR β/δ is coming up as a potential target for the pharmacotherapy of metabolic syndrome. Activation of this nuclear receptor seems to improve both insulin sensitivity and plasma lipid profile without causing weight gain. The limitations in use of TZD drugs include the risk of salt retention and heart failure and osteoporosis especially in elderly women.
Role of CDDO‒imidazolide inhibitor
Another inhibitor of adipogenesis in preclinical trials is CDDO‒Imidzolide (CDDO‒Im).CDDO‒Im activates the nuclear factor (erythroid derived 2) like 2‒Nrf2 pathway and results in increased mitochondrial biogenesis, decreased adipogenesis and increased energy metabolism. This drug has been shown to decrease total body weight and body fat and decreases hepatic lipid accumulation in rodents. there novel targets for obesity treatment are Peptidergic Signaling Pathway with advancement of knowledge regarding signaling of hunger and satiety from the GIT, which is mediated by GLP1,cholecystokinin (CCK), peptide YY(PYY) and ghrelin and the homeostatic mechanism of leptin and its upstream pathways there is pavement for drug development of obesity using hypothalamic pathway (Figure 1).
Role of ghrelin
Ghrelin is the only gastrointestinal (GI) hormone known to stimulate appetite is made up of 28 amino and is secreted by the stomach. Ghrelin levels were shown to be markedly increased in cases having Prader Willi Syndrome (PWS), relative to obese children PWS children had lower fasting insulin and higher fasting ghrelin and ghrelin/PYY. It is released both centrally (arcuate nucleus of hypothalamus) and peripherally (stomach) and its antisatiating properties may be due to its biological effects to increase GI motility and decrease insulin secretion. Ghrelin level increase in the absence of central intake and decrease right after meal initiation and exact macronutrient inducing greatest suppression is not well defined. In arcuate nucleus it prevents the activity of POMC neurons by stimulating NPY and AgRP. Ghrelin concentrations were found to be greater in obese in contrast to to normal weight children.40 Examining the effects on GLP1,PYY and ghrelin levels in 16 women before and up to 1 year after RYGB surgery , Beckman et al found that Ghrelin levels got reduced considerably in contrast to no changes found following after diet induced weight loss, while GLP1 increases and fasting leptin decreased in both groups and was lower in fat‒beverage group as compared with protein–beverage group after 1 year. Thus reason for greater efficacy of RYGBP surgery is obtained more effective in getting long term weight loss in contrast to diet alone treatment. Because of greater risk of surgery in morbidly obese patients having some metabolic complications, need for obtaining drugs which counter ghrelin or GHS‒R is there. Somatostatin infusion has been shown to suppress ghrelin levels. Also short term administration of octreotide, a somatostatin agonist was found to reduce ghrelin levels in children with PWS by though long term octreotide reduced acyl and desacyl ghrelin concentrations, De Waele et al found it did not have much impact on appetite and body weight. Schellekens et al.40 showed how different ligands acting on the growth hormone stimulating receptor(GHS‒R) have been developed including a vaccine made up of antibody against ghrelin which impaired the ghrelin action on CNS developed anti ghrelin RNA Spiegelmer NOX‒B11‒3 which blocked ghrelin –but not fasting–induced neuronal activation in the hypothalamic arcuate nucleus besides promoting development of antibodies against ghrelin, ghrelin enantiomers which can neutralize ghrelin and decrease acyl ghrelin through inhibiting ghrelin O‒acetyl transferase (GOAT),which belongs to a family of membrane bound‒O acyl transferases(MBOATS).46 Despite considerable interest one has not found answers using antighrelin drugs for achieving long term weight loss.
Role of peptide YY (PYY)
PYY, a gut hormone peptide is a 36 amino acid peptide which is synthesized in endocrine L cells in the distal GIT and is released into the circulation in response to food intake. This peptide has a number of tyrosine residues at the C and N terminus because of which it was named PYY.47 Along with NPY and pancreatic polypeptide (PP) it belongs to a family of peptides which share a common characteristic U shaped tertiary structure called the PP fold.48 They mediate effects via 5subtypes of a 7 transmembrane GPCR called Y receptor (Y1,Y2,Y4,Y5&Y6).YY3‒36 has the greatest affinity for the Y2 receptor subtype followed by Y1 and Y5 subtypes.49 The activation of presynaptic Y2 receptors by NPY3‒36(also NPY and PP)=>to the inhibition of neurotransmitter release.50 In comparison Y4 receptors have greater affinity for PP and modulates its anorexigenic signaling as well as the vasovagal reflex.51,52 PYY levels are low in the fasting states but peak after a meal and remain increased for various hours.53 Peripheral infusion of YY3‒36 has dose dependent anorectic effects in rodents.54‒56 Peripheral infusion of PYY1‒36 has weaker anorectic effects which likely result from its conversion to PYY3‒36 by DPPIV.57,58 Mice having global PYY knockout are hyperplasic, show increase weight gain and has increased visceral fat which are rescued by exogenous replacement of PYY3‒36 by exogenous replacement of PYY3‒36.59 Unfortunately PYY1‒36 has failed to show inhibitory effect on food intake in humans.60 DPPIV inhibitors used for treatment of T2DM may have mild anorectic effects by increasing the circulating levels of PYY3‒36. They regulate proliferation and differentiation of adipocytes.J104870 is a selective inhibitor of PYY. It has high affinity for the Y1 receptor and inhibits the NPY dependent Ca influx.61 Intracerebroventricular and intraperitoneal administration of J 104870 crosses the BBB and its intraperitoneal injection increases the drug concentration in brain. Long term oral administration of PYY antagonists also decreased the food intake in obese fa/fa zucker rats ,albeit only transiently.62 Others have shown that high dose systemic PYY antagonist decrease body weight while low doses decrease adiposity hypertrophy without effects on plasma lipids, glucose, insulin and total body weight.63
<p>Role of NPY receptors as potential targets for antiobesity drug development In contrast to PYY, the related 36amino acid NPY is mainly expressed in the brain and mainly activates Y1 receptors, which results in inhibition of adenylate cyclase and cAMP generation, and thereby exerting orexigenic effects.64‒66 Surprisingly, NPY deletion alone does not significantly reduce feeding and body weight in mice.67 However ob/ob mice deficient for NPY are less obese and less severely affected by DM.68 This finding has led to the postulation that the major role of neuromodulators is maintaining a minimum body weight as opposed to adjusting body weight. The Y1 receptor mediates the increase in total body weight and total fat accumulation without hyperphagia.69 Consistent with its peripheral effects it promotes rat 3T‒L1 preadipocyte proliferation in vitro.70 Pharmacological inhibition of the Y1 receptors by central administration of the Y1 antagonists, however results in significant attenuation of feeding in rodents, suggesting both peripheral and central effects.71 Interestingly Y1 receptor polymorphisms in the non coding region of the gene has been associated with lower fasting plasma triglycerides and higher plasma HDL concentrations.72
Wnt and low density lipoproteins receptor related protein 6(LRP6)
Wnt proteins are one class of endogenous factors which repress adipogenesis and WNT pathway has been well characterized in embryogenesis and tumor genesis. It is also a nutrient sensing pathway which can be activated by sensing glucose and via insulin signaling.73 Impaired Wnt /LRP6 signaling is associated with hyperlipidemia which gets reversed by Wnt 3a rescue.74 It has been shown that the Wnt/LRP6/TCF7L2 axis is a regulator of glucose metabolism and potential herapeutic to target for insulin resistance.75‒77 19 different ligands activate either canonical or no canonical pathways resulting in regulation of transcription, calcium signaling, and cell polarity. tried to explain the mechanism by which Wnt binding to its receptors Frizzled and low density lipoprotein receptor –related protein 6 (LRP6) triggers downstream signaling events .Signaling through frizzled results in the stabilization and nuclear translocation of β–catenin where it associates with the members of T‒cell factor/Lymphoid enhancer (TCF/LEF) family and the resulting complex targets key genes to mediate the Wnt response.78 The canonical Wnt signaling pathway comprises a cascade of events initiated by binding of a Wnt–protein ligand to a Frizzled family receptor and phosphorylation of its coreceptors LRP5/6. This causes inactivation of GSK3β, breakdown of Axin, Dishevelled, and APC complex and subsequent stabilization of β‒catenin, which translocates to the nucleus following that, where it interacts with TCF/LEF family of transcriptional activators for promoting gene expression. GSK β gets inhibited by insulin also and mediates the crosstalk between insulin and leptin –signaling pathways. Takada et al showed how the no canonical Wnt signaling through Ca MKII‒TAK1‒TAB2‒NLK (Nemo Like kinase transcriptionally represses PPAR γ Transactivation and induces Runx2 expression, promoting osteoblstogenic proliferation in preference to adipogenesisin bone marrow mesenchymal progenitors. Wnt 5a activates nemo like kinase (NLK) which in turn phosphorylates a histone methyl transferase SETDB1=>formation of a corresseppor complex that inactivates PPAR γ function through histone H3‒K9 methylation. They also showed that Wnt5a‒Ror2 (receptor tyrosine kinase‒likeorphan receptor proteins signals enhanced receptor activation of nuclear factor Κb (RANK), thereby inducing Rank ligand (RANKL)induced osteoclastogenesis. Latest research has shown that inducers of osteoblastogenesis (such as bone morphogenetic protein 2 and Wnt ligands) use different mechanisms to suppress transactivation function of PPAR γ during osteoblastogenesis from MSC’s, Signaling via the canonical Wnt‒beta catenin pathway inhibits PPAR γ mRNA expression, whereby signaling via the non canonical Wnt pathway results in activation of a histone methyl transferase SETDB1 which represses PPAR gamma transactivation through histone H3‒K9 methylation of target genes. This occurs in part by controlling important preadipogenic transcription factor, peroxisome proiferator activated factor receptor gamma PPAR γ.79 Showed that Wnt/beta catenin signaling promotes the expression of chicken ovalbumin upstream promoter ‒transcription factor COUP‒TFII, which is a potent adipogenic factor. They further showed that COUP‒TFII results the silencing mediator for retinoic acid receptor and thyroid receptor ( SMRT) co repressor complex to the 1st intron located downstream from the 1st exon of both PPAR‒gamma ‒1and 2introns mRNA (Figure 2).80 This maintains the local chromatin in a hypoacetylated state repressing PPAR γ gene expression to inhibit adiogenesis resulting in decreased chromatin acetylation and repressing PPARγ gene expression to inhibit adipogenesis (Figure 2). Takada et al showed that no canonical Wnt signaling via Wnt 5a suppresses PPAR γ function through chromatin inactivation triggered by recruitment of a repressing histone methyl transferase set domain bifurcated 1(SETDB1) which thus=>osteoblastic cell lineage from mesechymal stem cells although PPAR γ is the prime inducer of adipogenesis.81 Showed that glycogen synthase kinase 3β(GSK3β) a serine threonine kinase which is the key enzyme for Wnt pathway is increased in hypothalamus during obesity and exacerbates high fat diet induced weight gain as well as glucose tolerance.82 Characterized whether WNT signal in general is altered in the hypothalamus of adult mice. They studied murine arcuate nucleus and found target genes of the WNT pathway were down regulated in obese and glucose intolerant leptin deficient mice. Similarly the number of cells immunoreactive for the active phosphorylated form of the WNT–coreceptor lipoprotein receptor related protein 6(LRP6) was also decreased in leptin deficient mice. Leptin treatment normalized expression of the WNT target genes Axin 2 and cyclin D and increased the number of phosphor‒LRP‒6‒immunoreactive cells reaching levels of lean controls. Leptin also increased the levels of phosphorylated (inactive) GSK3 β in the arcuate nucleus and this effect was colocalized to NPY neurons suggesting that the inactivation of GSK3 β may contribute to the neuroendocrine control of energy homeostasis. Thus they concluded that hypothalamic WNT signaling is an important novel pathway which integrates peripheral information of the body’s energy status encoded by leptin.83 There is no Wnt antagonist in pipeline for obesity treatment currently and paradoxically, the GSK3β antagonist lithium causes weight gain.84 Metreleptin (myalept), a synthetic analog of the hormone leptin, maybe helpful in treating hyperlipidemia secondary to diabetes and obesity in patients with impaired Wnt signaling. In USA it has been approved as replacement therapy for treatment of leptin deficiency and lipodystrophy. This drug is also being currently investigated for the treatment of T1DM in a combined effort led by juvenile Diabetes Research Foundation in collaboration with Amylin pharmaceuticals and the University of Texas Southwestern Medical Centre to improve metabolism and control appetite.
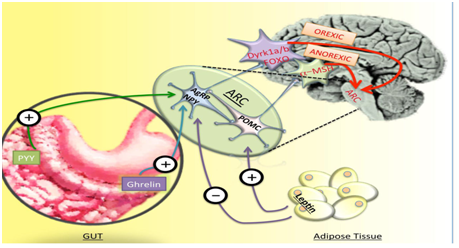
Figure 2 Courtesy ref no 126‒Schematic diagram of peptidergic pathways in interplay between gut adipose tissue hormones and central nervous system regulation of appetite.
Dyrk1 family of proteins‒novel targets for drug development
The dual specificity tyrosene regulated kinases (Dyrk) family of proteins ,belong to the larger group of kinases , which also includes CDK’s ,GSK3 and MAPKs.85‒87 Members of the Dyrk family of protein kinases, many of which have been implicated in nutrient sensing, have significant homologyin the kinase domain and an upstream sequence called the Dyrk homology domain. The Dyrk1B orthologue in yeast, YAK1 is a nutrient sensing protein which is inhibited by glucose. It increases neoglucogenesis and reduces glycolysis by inhibiting the yeast transcription factor MSN2.88‒90 The mammalian orthologue of MSN2 induced by glucose in pancreatic β cell and plays an important role in regulating insulin biosynthesis, glucose homeostasis and islet size.91,92 Dyrk kinases also act as a primary kinase for GSK β phosphorylation of NFAT’s, a group of transcription factors which contribute to skeletal muscle development and its glucose and insulin homeostasis and to pancreatic function.93,94 The Dyrk1B is transcriptionally regulated by the Rho GTPases and is ubiquitously expressed.95‒99 It contains different motifs which include a nuclear localization signal(NLS), a kinase like domain which harbours the NLS, a DYRK homology box, an N terminal autophosphorylation accessory region and kinase domain and the PEST sequence, which acts as a signal peptide for protein degradation. Cellular activity of Dyrk proteins is regulated by autophosphorylation, protein stability (via the PEST sequence) and sub cellular localization. Dyrk1A and its drosophila ortholog minibrain (mnb) regulate NPY and sNPF (the drosophila functional homology of NPY) signaling in mice and drosophila respectively. In mouse hypothalamic cells and drosophila neurons, NPY and sNPF increase the expression of Dyrk1 A and mnb by activating the PKA‒CREB pathway. Dyrk1A phosphorylation of Sirt1 causes increased Sirt1 dependent deacetylation and activation of the transcription factor FOXO. FOXO further potentiates sNPF/NPY expression and promotes food intake, effects which are neutralized by activation of insulin signaling. Dyrk1A over expression in mice causes lower FOXO aceylation and increased NPY expression in the hypothalamus leading to increased food intake (Figure 3).
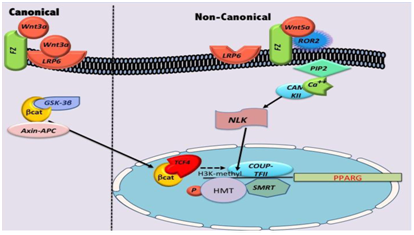
Figure 3 Courtesy ref no.126‒Schematic diagram of canonical and non canonical want regulation of PPAR gamma (PPARG) transcription in adiposeness.
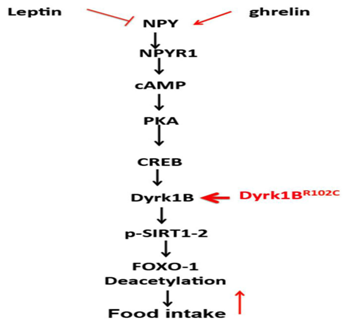
Figure 4 Courtesy ref no 126‒Schematic diagram of interplay between leptin, ghrelin, NPY and Dyrk1A (and possibly B) in central regulation of FOXO1 and food intake. The effects of R102C mutation on these pathways are shown in red.
Role of dyrk1B in truncal obesity
Recently identified Dyrk 1B as the disease genes in three large families with coinheritance of early onset coronary artery disease (CAD) and central obesity in South west iron. Founder mutation was seen in DYRK 1B, substituting cysteine for arginine in position 102 in the highly conserved kinase like domain. Functional characterization of the diseases gene showed that premutant protein encoded by DYRK1B inhibits the SHH (Sonic hedgehog) and Wnt signaling pathways and thus enhanced adipogenesis. Also DYRK1B promoted the expression of key gluconeogenic enzyme G‒6‒P. The R102C allele showed gain off function activities by potentiating these effects. A second mutation, substituting proline for histidine 90, was found to cosegregate with similar syndrome in an ethnically distinct family .Thus they concluded a role for DYRK1B exists in adipogenesis and glucose homeostasis and associate its altered function with an inherited form of metabolic syndrome. Dyrk1B protein is a dual specificity kinase with its tyrosine phosphorylation activity mainly involved in autophosphorylation and an arginine directed serine/threonine kinase activity. Its kinase activities are promoted by signaling from Rho‒Rac1. Its expression gets inhibited by RAS‒MEK‒ERK. Loss of function mutations in the pathways were recently linked to obesity and insulin resistance in humans and mice.99‒101 Targets for its serine/threonine phosphorylation include HNF‒1α, glycogen synthase.102 FOXO and SIRT1/2,103,104 all of which are proteins involved in glucose metabolism and transcription of insulin GLUT2, glucokinase and G‒6‒Pase. Dyrk1B is a pan cellular protein. It is localized in the nucleus in malignant cell lines, but found in cytoplasm in human skeletal muscle.105‒108 Hence its functions in malignant cells can’t be assumed automatically for normal cells. There is a suggestion that it acts as a cell cycle regulator in the nucleus, mainly by increasing the turnover of p27kip (CDKN1B).109 Mice deficient for p27kip are protected against atherosclerosis, obesity and insulin resistance.110 In cytosol, DYRK1B may act as an ant apoptotic factor by stabilizing p21cip.111‒114
Interaction between leptin, NPY, dyrk family of proteins
Mnb and Dyrk 1A and B get markedly expressed in arcuate nucleus. Both Dyrk 1Aand B have CREB binding motifs in their promoter’s. Thus the stimulatory effects of NPY can similarly effect Dyrk1B expression and FOXO1 activation. These findings suggest a potential role of Dyrk1B in regulation of appetite. Whether Dyrk1B mutations and/or over expression increase food intake and weight gain is being currently investigated.
Role of autophagy dysfunction in obesity
In animals chronic intake of HFD causes DIO which =>insulin and leptin resistance in hypothalamic neurons.115‒117 Increased inflammation in the hypothalamus was identified to mediate the development of obesity and the pathways which include IKKB/NFKB pathway and upstream inputs such as MYD88,endoplasmic reticulum stress and JNK signaling.118‒124 Chronic inflammatory stimuli can also =>neuronal apoptosis which is important for the anorexigenic response.125,126 Recently neuroimaging studies revealed that dysfunctional and neuronal loss were associated with obesity in the hypothalamus of humans and rodents.127‒129 Besides having effects on food intake and energy expenditure hypothalamic inflammation seems to impair systemic glucose metabolism. Genetic and pharmacological modulation of the ER stress and inflammatory pathways in the hypothalamus affected liver gluconeogenesis.130‒132 Inflammatory inhibition of TLR4 or TNFα signaling in the hypothalamus impaired improved insulin signal transduction in the liver and reduced hepatic glucose production. These studies suggest that hypothalamic inflammation plays a role in weight gain and systemic dysfunction of glycaemic control. Meng and Cai have showed that neuronal autophagy is compromised under conditions of chronic excess fatty acids in the diet. In chow feeding mice, the site specific inhibition of ATG7 in the mediobasal hypothalamus =>autophagy inhibition, impairment of hypothalamic control of energy balance, obesity and hypothalamic inflammation through IKB activation. In HFD these metabolic changes got increased along with progression of insulin and leptin resistance.133 Normally autophagy is a homeostatic process that occurs in alleukaryotic cells and is needed for degrading damaged proteins as well as organelles. It also sequesters the cytoplasmic components in the double membrane vesicles known as autophagosomes.134 These autophagosomes thus fuse with lysosomes where the damaged proteins and organelles are degraded by lysosomal proteases and recycled.134,135 If this autophagy is impaired it may cause inflammation suggesting that autophagy helps in inhibition of inflammatory response.136,137 Portovedo hypothesized that obesity may=>impairment in hypothalamic autophagy in mice. They examined the hypothalamic distribution and content of autophagic proteins in animals with obesity induced by 8or 16wks HFD to induced obesity and in response to icv injection of palmitic acid. They showed that chronic exposure to a HFD =>an increased expression of inflammatory markers and down regulation of autophagic proteins. In obese mice autophagic induction =>the down regulation of proteins like JNK and Bax which are involved in the stress pathways. In neuron cell lines palmitate has a direct effect on autophagy even without inflammation activity. Thus understanding the cellular and molecular basis of autophagy is important in finding new diagnostic and therapeutic targets for obesity.138
Many antiobesity drugs have been developed and had to be removed from the market due to the associated side effects .Drugs like rimonabant showed great promise but having severe psychiatric effects including suicidal effects led to the termination of future studies. Though monotherapeutic approaches were not found to be optimally effective even the drug which got approved like lorcarserin in on the verge of getting removed in view of its antitumor side effects. Same is the case for tesofensine, which has already started showing severe side effects in phase 3 trials itself. Although combination therapies have been more effective like phentermine/topiramate (Qsymia) and bupropion/naltrexone (Contrive) which got FDA approval in 2012and 2014 respectively ,there are still some dilemma regarding use of Qsymia in lieu of fetal effects like cleft lip, palate, needing regular pregnancy tests and some incapacitating CVS side effects. Though Mc4 receptors remain important targets most drugs developed have severe autonomic side effects including priapism except for RM 493.CDDO‒Im is still in experimental stage where clinical trials done only in animals and yet no human trial done or effective drug developed. Similarly RIO trials done only in animals although Cyt R &D is trying to develop some drugs in this line. Bariatric surgery still offers much more advantage over medical therapy–reviewed in ref 3, giving 8‒10% better loss and more sustained over these medical therapies. Similarly trials on RIO only done in animals and efficacy in human beings yet to be proven, as is the case for SMRT targets. Although inhibitors of pancreatic lipase like orlistat and serotonergic drugs are the few which got initial approval orlistat is the only one approved for long term treatment of obesity in view of development of tolerance to those amphetamine like drugs. Antidiuretic drugs like glucagon like peptide‒1 (GLP1R) agonists and DPPIV inhibitors are being evaluated for their effects on obesity and metabolic traits. Liraglutide is slightly more efficacious acting both on homeostatic feeding centers as well as hedonic mesolimbic areas got approved in 2012, being most effective in 3mg dosage. Genetic studies of obesity and metabolic syndrome have sorted out novel molecules acting on hunger and satiety peptidergic signaling on the gut–hypothalamic axis or the melanocortin system of the brain which promises to be important targets for future drug development. Aim is to develop drugs which not only treat obesity but also have impacts on the associated traits favourably. Of the ghrelin pathway which was found to be important molecule which got suppressed following bariatric surgery, although lot of efforts have been done to develop vaccines, antibodies or ligands for GHS‒receptors still no effective therapy has emerged .Similarly PYY is another molecule involved in energy homeostasis and found to be raised following BS, only animal studies have been carried out where PYY3‒36 has shown efficacy but no effective molecule developed as yet I humans. Regarding Wnt pathway, although important role found in energy homeostasis still no WNT molecule developed in humans, the only applicationis use of Metreleptin in patients with Wnt deficiency in human beings. Similarly though DYRK mutations have been associated with some forms of obesity and metabolic syndrome in humans along with its role in glucose metabolism mostly studies have been done in animals and yet it has to be worked out if these pathways can be targeted regarding development of antiobesity therapies. As far as autophagy is concerned although importance shown in role in hypothalamic inflammation still experiments only carried out in animals. Yet lot of work is further required if any of these pathways can be utilized for developing effective antiobesity therapies. One is still far from developing an effective long term strategy for obesity medically without side effects or tolerance developing.
None.
Author declares that there is no conflict of interest.

©2018 Kaur, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.