MOJ
eISSN: 2381-179X


Case Report Volume 13 Issue 3
1Department of Radiology Mother and Child, CHU Hassan II Fez, Sidi Mohammed Ben Abdellah University, Morocco
2Department of Radiology and Interventional Imaging, CHU Hassan II Fez, Sidi Mohammed Ben Abdellah University, Morocco
Correspondence: Sara El Malih, Department of radiology Mother and Child, CHU Hassan II Fez, Sidi Mohammed Ben Abdellah University, Fez, Morocco, Tel +212628244889
Received: August 08, 2023 | Published: August 18, 2023
Citation: El-Malih S, El-Mounssefe G, Akammar A, et al. Spontaneous intraventricular rupture of a pineal teratoma in a 12-year-old infant. MOJ Med Clin Case Rep. 2023;13(3):62-64. DOI: 10.15406/mojcr.2023.13.00439
Pineal teratoma is a subtype of germ cell tumors that develops in the pineal gland and represents the second most common pineal region tumor. Although they are typically seen in a mature benign form, teratomas can sometimes present as immature fetal appearing tissue and have the potential for malignant transformation. On imaging, teratomas show typical appearance with areas of mixed CSF, lipid and calcifications. In this paper we illustrate the case of a typical mature pineal teratoma with a spontaneous rupture in the fourth ventricule in a 12-year-old infant. The patient was referred to our institution for chronic headache, and recent onset of projectile vomiting. A CT scan revealed significant hydrocephalus and a mixed density lesion in the pineal region with hypo dense lesions in bilateral lateral ventricles corresponding to fat component, very suggestive for the diagnosis. The patient underwent a ventriculo-peritoneal shunting for his hydrocephalus then he was referred for chemotherapy and radiation before surgery but unfortunately, he died unexpectedly before any intervention was carried out.
Keywords: pineal, teratoma, hydrocephalus
Intracranial teratomas constitute up to 50% of fetal brain neoplasms;1 in neonates, they account for 33% of intracranial tumors, whereas in infants under 15 years, they account for only 2-4% of intracranial tumors.2-4 They have a male preponderance that ranges from 2:1 to 8:1 with an overall survival rate of 90-100%.5
Intracranial teratomas often develop from the pineal gland which is a frequent site, and affect the third ventricle,6 nevertheless, intraventricular ruptured teratomas are extremely uncommon, multiple mechanisms of rupture have been identified, including physical injury, ischemic necrosis, and enzymatic degradation, nonetheless, the pathophysiology of this occurrence within the cerebral compartment is still unknown. Once the rupture occurs, the patient may rapidly worsen, developing hydrocephalus-like symptoms such as headache, nausea, vomiting, and papilledema. Early detection of a ruptured teratoma is crucial for effective and optimum patient care.
In this case, we report a patient who presents with a ruptured intracranial teratoma originating from the pineal region, with fat content migrating superiorly into the bilateral lateral ventricles.
A 12-year-old male patient with a 2 months history of intermittent headaches, presented with acute onset nausea, headache, and projectile vomiting while sitting at school. After seeking medical attention, he was found to be neurologically intact on physical exam. CT scan of the head was performed and demonstrated a mixed density mass in the pineal region with central calcifications, and soft tissue components (Figure 1). Hypodense lesions were seen in the lateral ventricles, third ventricle, and quadrigeminal cistern. The lesions in the lateral ventricles demonstrated hypo attenuations as low as -93 Hounsfield Units, consistent with fat with frontal horn level (Figure 2 & Figure 3). Ventriculomegaly was also evident, due to the obstruction of the aqueduct of Sylvius.

Figure 1 Non-enhanced cerebral CT scan (a), shows a mixed heterogeneous mass extending from the pineal region anteriorly into the cistern of the velum interpositum. The mass contains a central calcification with tissue component (enhancement) on both the axial (b) and the coronal (C) post contrast CT scans.

Figure 2 Cerebral CT scan in axial (a) and sagittal reconstruction (b) showing hypodense lesions in the lateral ventricles consistent with fat content forming a supernatant lipid layer floating on the CSF in the superior portions of both lateral ventricles, very suggestive of intra-ventricular rupture..
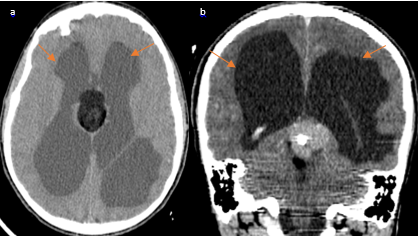
Figure 3 (a) NECT in axial view and postcontrast coronal (view) (B) show hydrocephalus secondary to the obstruction of the aqueduct of Sylvius by the pineal teratoma.
The patient was referred to the pediatric neurosurgery department where he benefited first from a ventriculo-peritoneal shunting for the management of the hydrocephalus, then he underwent a steriotaxic biopsy. Immunohistochemistry (IHC) staining revealed positivity for CD117 and SALL4 which were consistent with the diagnosis of germ cell tumor. The patient was referred for chemotherapy and radiation before surgery but unfortunately, he died unexpectedly before any intervention was carried out.
Teratomas are one of six varieties of germ cell cancers identified by the World Health Organization.7 They arise from all three-germ cell layers.8 Histologically, teratomas are classified into three types: (1) mature tumors, the most usually found form, which have completely differentiated tissue; (2) immature tumors, which consist of a mix of fetal and mature-type tissue elements, and (3) teratomas with malignant transformation, which have malignant degeneration of mature tissue. Teratomas are neoplasms characterized by multipotent cells that revert to normal organogenesis, typically forming tissues that represent a mix of two or more embryological layers, including ectoderm, mesoderm, and endoderm, frequently found elements include fat, bone, cartilage, teeth, and hair.2,8 Pineal teratomas can be encapsulated either completely or partially, but they can also be unencapsulated and locally invasive.
The pathophysiology of germ cell tumors is still unclear. While the most commonly recognized hypothesis holds that these tumors are the consequence of a primordial totipotent germ cell, Sano provided an alternate viewpoint, he postulated that embryonic cells at various stages of development may go misdirected during primitive streak formation and eventually become incorrectly enfolded into the brain during neural tube formation.9
Intracranial teratomas have been reported to occur in the lateral ventricles, pineal region, suprasellar regions, and the posterior fossa, but the pineal location is the most frequent.7 The clinical manifestation of tumors arising in the pineal area is frequently that of hydrocephalus or Parinaud's Syndrome.10 Those in the suprasellar area are more prone to endocrine (dysfunction) or visual problems.11
On imaging CT or MRI scans are characteristic for intracranial teratoma diagnosis. Teratomas are often visible as a solitary irregular mass with or without multilocularity and mixed signals from various tissues, such as fat tissue, calcification, cystic elements, hair follicles, cellular debris, and keratinocytes. Though MR imaging is insensitive for detecting small calcifications, CT could easily demonstrate regions of calcification in teratomas. Teratomas may also demonstrate contrast material enhancement on both CT and MR images. Enhancement is usually heterogeneous, either limited to the solid-tissue areas or along the walls lining the cystic spaces. Perilesional cerebral edema is uncommon due to the existence of the tumor capsule.
Pineal teratomas may rupture spontaneously or at surgery, spilling their varied contents, causing a chemical meningitis or, rarely, implanting along the meninges, an intraventricular or subarachnoid low density lesion should raise suspicion for a ruptured teratoma. MRI imaging can easily recognize intraventricular free fat and separate it from other pathologies such as pneumocephalus or pneumoventricle, because of the high T1 signal and fat suppression.
So far, teratoma treatment options have remained (controversial). When possible, treatment comprises radical excision. If more than 90% of the tumor has been eliminated, the excision is called complete.12 Because mature teratomas are radioresistant, complete tumor excision is the preferred therapy.13 The pathophysiology determines the clinical outcome of patients with primary intracranial teratomas. Recurrence after incomplete excision has been observed earlier, because of the possibility of residual immature or malignant tissue.
While patients normally recover without incident, they should be followed for a longer length of time since malignant teratomas or new teratomas could appear intracranially or extracranially.
None.
Authors declare that there is no conflict of interest.

©2023 El-Malih, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.