MOJ
eISSN: 2381-179X


Case Report Volume 13 Issue 3
1First Moscow State Medical University named after IM Sechenov, Clinic of Aortic and Cardiovascular Surgery, Russia
2Federal State Autonomous Educational Institution of Higher Education “Russian National Research Medical University named after NI Pirogov” of the Ministry of Health of the Russian Federation, Russia
3Department of Operative Surgery and Topographic Anatomy of the First Moscow State Medical University named after I.M. Sechenov, Russia
4State Budgetary Institution of Health Care “Chelyabinsk Regional Clinical Hospital”, Russia
Correspondence: MI Tkachev, Clinic of Aortic and Cardiovascular Surgery, Sechenov University, Trubetskaya street, 8, Moscow, Russia, 119048, Tel +79621716698
Received: August 04, 2023 | Published: August 17, 2023
Citation: Komarov RN, Tsaregorodtsev AV, Tkachev MI, et al. Monoblock aorto-mitral homograft: surgical technique and results. MOJ Clin Med Case Rep. 2023;13(3):58-60. DOI: 10.15406/mojcr.2023.13.00438
The treatment of infective endocarditis of a prosthetic valve remains a big problem to this day. It is even more difficult to make an informed decision when two prosthetic valves are infected simultaneously with the inclusion of the mitral-aortic contact and the formation of an aortic root abscess. Occurrence of infectious endocarditis of a prosthetic valve is 1-6% and equally concerns both mechanical and biological prostheses. According to the literature, the hospital mortality rate ranges from 20 to 55%. In this situation, excision of both infected valves and adjacent structures is necessary. To compensate for the tissue deficit arising after valve dissection and mitral-aortic contact, the choice was made in favor of surgery using monobloc aortic-mitral homograft.
Aortic and mitral valve surgery remains a challenge for cardiac surgeons and cardiologists. If reconstructive valve surgery is not possible, replacement with a commercially available mechanical or xenogeneic valve is required. The aortomitral homograft can be another alternative with rational patient selection and the right surgical technique can be an excellent solution, in terms of survival rate and quality of life of patients.
A number of studies have shown the benefits of homograft valves, when used with the correct surgical technique, to ensure a good postoperative result and improve the patient's quality of life. They caused virtually no risk of thromboembolism and bleeding associated with anticoagulant therapy; they also had an advantage in use in the presence of active infection,1 since they have been shown to be associated with a low risk of early and late endocarditis. The main advantages of the method are the absence of synthetic material and the absence of the need for constant anticoagulation. Another potential advantage of the aortomitral homograft is the preservation of mobility of the supporting apparatus, which may be important for the long-term functioning of the homograft.
Although successful experience of valve replacement by aortomitral homograft implantation has already been documented, this operation is now rare. In our opinion, the factors preventing stable positive results of the operation are: the complexity of implantation of homografts, difficulty in selecting the right size homografts, the probability of early suture failure in the place of papillary muscle implantation.
The decision to use homografts for valve replacement is made individually for each patient, taking into account the prognosis of results and complications. So far this method of valve replacement has not been widely used, but in the treatment of complex valve endocarditis, the use of aortomitral homograft may sometimes be the only method of treatment that can provide an acceptable result.
We present the experience of aortomitral homograft implantation performed in May 2022 in a patient with subacute infective endocarditis (presumably from September 2021) with aortic (severe failure) and mitral (severe failure) valve involvement and aortic root abscess. We believe that this operation is a good alternative to the Commando operation (UFO), which consists in separate prosthetics of the mitral and aortic valve with proprietary prostheses, using two patches of treated bovine pericardium for reconstruction of the central fibrous body and the roof of the left atrium.2
The operation was performed under combined endotracheal anesthesia, artificial circulation, hypothermia and pharmaco-cold cardioplegia. Access to the heart was performed from the median sternotomy. We followed the implantation technique described below. This technique was used in 1 patient in our clinic https://www.youtube.com/watch?v=wCNrSQfIlYU.
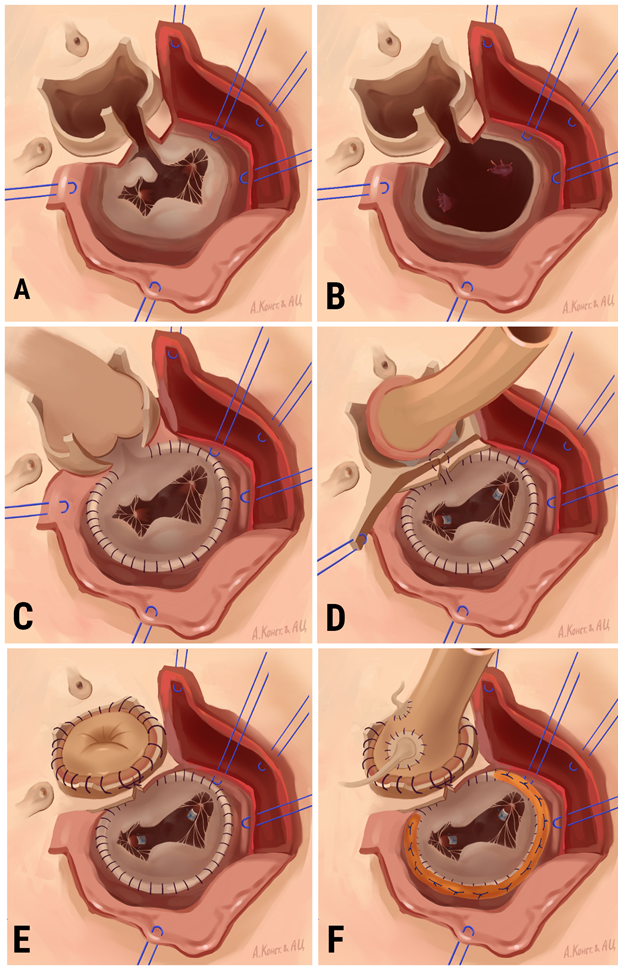
Figure 1 Sequence of aorto-mitral homograft implantation A, The aortic valve is dissected. An incision of the non-coronary sinus with transition to the anterior mitral valve leaflet was performed. Mitral valve dissection is started; B, Mitral valve dissected, revision and sanation of the left heart; C, Suturing of the anterior half ring; D, Implantation of the mitral portion of the aortomitral homograft; E, Implantation of the aortic portion of the aortomitral homograft; F, Implantation of coronary artery ostium on buttons.
The artificial circulation time was 235 minutes; myocardial ischemia time was 210 minutes. The patient was in the intensive care unit for 3 days and in the hospital for 12 days. In the postoperative period, the patient underwent echocardiography to assess homograft function; the results are shown in the table 1.
EchoCG |
Before the surgery |
1 year after the surgery |
Aortic valve insufficiency |
2 degree |
0,5 degree |
Mitral valve insufficiency |
1 degree |
0,5 degree |
Tricuspid valve insufficiency |
3 degree |
1 degree |
Systolic pulmonary artery pressure |
50 mm Hg. |
28 mm Hg. |
End-diastolic volume |
155 mL. |
120 mL |
Ejection fraction |
58% |
60% |
Table 1 Immediate results of the operation
Patient's condition 12 months after surgery
Six months after the operation, the patient is in functional class I according to NYHA, he has no complaints from the cardiovascular system. The patient underwent a cardiac MRI. There are no signs of infective endocarditis (Figure 2).
In the PubMed database, we found not so many clinical cases of aortomitral homograft implantation, which were presented in the form of a table (Table 2). Analyzing the world experience of this operation, we came to the conclusion that this operation is quite rare. This is due both to the complexity of performing and providing the operation, and to the relatively small number of such "neglected" patients in our time.
|
Year |
Article |
Number of observations |
Indication for surgery |
Observation period (prosthesis – dependent complications= +/-) |
|
2007 |
1 |
Relapse of infective endocarditis |
11 months (-) |
|
|
1 |
Relapse of infective endocarditis |
6 months (-) |
||
|
2001 |
1 |
Para-aortic abscess, fistula between the aorta and the right atrium |
8 months (-) |
|
|
2006 |
3 |
Recurrent infective endocarditis |
5.5 years (-)* |
|
|
2015 |
1 |
Prosthetic infective endocarditis |
9 months (-) |
|
|
2022 |
1 |
Relapse of infective endocarditis |
20 days (-) |
Table 2 Сlinical cases of aortomitral homograft implantation
*1 patient survived, remains asymptomatic
This surgery is an alternative to Commando surgery, which is used for correction of double-valve lesions with involvement of the central fibrous body. There are no systematic direct results of aortomitral homograft implantation in the works found. However, it is difficult to think of a more effective method of treatment in such severe cases as extensive infectious endocarditis of the aortic and mitral valve with involvement of the fibrous skeleton of the heart, abscess of the aortic root, fibrous body fistulas.
The aortomitral homograft in such a case is an ideal implant because it allows: to reconstruct the fibrous body much better than with a biological or synthetic patch (1), to replace 2 affected valves (2), to increase the survival and curability of patients with recurrent prosthetic endocarditis compared to the group in which biological or mechanical prosthesis were used (Commando operation).8 So far, there has been no evidence of late reinfection of the homograft.9
Implantation of a monobloc aorto-mitral homograft is a reliable method of treatment. However, several key issues need to be addressed: (1) myocardial protection using both antegrade and retrograde modes of delivery; (2) proper surgical training of the staff to work in the conditions of the limited ischemic time of the heart; (3) availability of monoblock homografts in the homograft bank; (4) avoiding disturbance of the geometry of the structures of the implanted aortomitral complex; (8) careful work in the area between the noncoronary and the right coronary aortic sinus, taking into account the danger of damage to the conduction pathways in this location; (9) treatment of such severe patients only in specialized departments with good surgical experience.
The use of aortomitral homograft gives a huge number of options for further management of the patient, which is certainly another advantage: the possibility of reconstructive interventions on the homograft in case of its dysfunction, the possibility of subsequent interventional valve prosthetics in case of homograft dysfunction. We also consider it necessary to mention that another solution is also possible: the use of not only homografts, but also neocuspidization of the valve apparatus with autopericardium, because autopericardium has high resistance, which allows preventing the recurrent development of endocarditis.10
We believe that the use of aortomitral homograft is one of the promising options for correction of aorto-mitral pathology in infectious endocarditis. Further research in this area is necessary and important.
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Informed consent was obtained from all individual participants included in the study in accordance with the Helsinki Declaration.
The study design was approved by the ethics committees of the participating centers. All participants were provided with the complete information about the design of the study and potential risks.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
None.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

©2023 Komarov, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.