MOJ
eISSN: 2471-139X


Review Article Volume 5 Issue 5
1Department of Molecular Biology, School of Exact, Physical-Chemical and Natural Sciences, Argentina
2Department of Animal Anatomy, School of Agronomy and Veterinary, National University of Rio Cuarto, Argentina
Correspondence: Nancy Rodriguez, Molecular Biology Department, Faculty of Exact Physical Chemistry and Natural Sciences, National University of Rio Cuarto, Rout 8 Km 601, X5804BYA Río Cuarto, Cordoba, Argentina
Received: June 01, 2017 | Published: September 18, 2018
Citation: Liaudat AC, Mayer NI, Vivas A, et al. Attenuation effects of prenatal stress by early postnatal stimulation in different research models. MOJ Anat & Physiol. 2018;5(5):284–290. DOI: 10.15406/mojap.2018.05.00211
Prenatal stress alters the pattern of corticoid secretion and affects transplacentally the fetus hypothalamic-pituitary-adrenal axis (HPA) in different animal models. Early postnatal stimulation or handling results in an increase in maternal care to the offspring what reverts the effects of prenatal stress on the alterations produced in the HPA axis. In this revision article we describe and discuss the underlying mechanism present and discuss the mechanisms leading to causing imbalance produced by prenatal stress and how it is related to immune system disorders. The general effects that postnatal early stimulation has on the parameters altered by prenatal stress and the attenuation produced on prenatal stress immune system negative effects will also be discussed.
Keywords: handling, stress, immune system, hypothalamic-pituitary-adrenocortical axis
The prenatal and early postnatal life stages are both dynamic and vulnerable phases during mammalian development. Exposure to adverse factors that interfere with this critical sequence of events places the exposed individual to a higher risk of developing various disorders in adult life.1–3 For instance, high blood glucocorticoids concentration in pregnant stressed females crossing the placenta and the fetal blood-brain barriers,4 can affect brain development, birth weight and HPA axis function in offspring.5
In mammals, the mother is not only the source of infant nutrition but also thermal, olfactory, visual and auditory stimuli6 and a complex bidirectional communication between the mother and offspring is established immediately after parturition. For instance, arched back nursing behavior and milk ejection in the rat are dependent on the combined suckling stimulus of several neonatal pups.7 Numerous studies demonstrated that prenatal stress-induced alterations can be reverted by handling.8 Using the rat as model organism, Zhang et al.9 demonstrated that neonatal handling could improve the spatial learning and memory ability previously impaired by prenatal stress.
In this review, we describe effects of prenatal stress on different aspects of animal physiology and analyze the possible mechanisms underlying prenatal stress-associated alterations, in particular on immune system function. Then, the general effects that postnatal early stimulation has on the parameters altered by prenatal stress and finally, the attenuation that handling produced on prenatal stress immune system negative effects.
Early life stress triggers hypothalamic-pituitary-adrenocortical (HPA) axis activation and the associated neurochemical reactions following glucocorticoid (GC) release are accompanied by a rapid physiological response. An excessive response may affect the developed of different organs/systems such as the brain, resulting in neurobehavioral and neurochemical changes which are evident in later life.10 The embryo and fetus are highly responsive and vulnerable to the gestational environment. Glucocorticoids represent a major class of developmental cues and are crucial for normal brain development. In animal models, GC exposure during early development has been associated with transcriptomic and epigenomic changes that influence behavior, HPA function and growth.11 Repeated exposure to high levels of GC during pregnancy suppresses 5α-reductase expression and allopregnanolone levels in the fetus and results in reduced myelination. Both fetal growth restriction and prenatal maternal stress lead to increased cortisol concentrations in the maternal and fetal circulation. As a consequence, prenatal stress results in reduced expression of γ-aminobutyric acid (GABAA) receptor subunits that normally enhance neural cells sensitivity.12
A defense mechanism against high hormone levels is the expression of 11β-hydroxysteroid dehydrogenase type-2 (11β-HSD2) that catalyzes the conversion of corticosterone into inactive metabolite, 11-dehydrocorticosterone,13 limiting in this way fetal exposure to maternal GC. Repeated exposition to stressors during pregnancy significantly reduces the placental expression of 11β-HSD2 and its activity,14,15 potentially increasing the exposure of the fetuses to maternal GC. Similarly, Fujioka et al.16 demonstrated that adrenal glands from 16-day old rat fetuses can secrete corticosterone. Moreover, rat fetuses exposed to prenatal stress exhibited decreased body, adrenal, pancreas and testis weights. These alterations were associated with reduced plasma levels of glucose, growth hormone and ACTH, whereas insulin, IGF-1 and plasma corticosteroid binding globulin levels were unaffected.14 Contrary to these results, it was reported that increased peripheral sympathetic nerve activity may contribute to the attenuation of glucose uptake and thus reduce insulin sensitivity.17 Del Cerro et al.18 found that prenatal stressed rats had higher ACTH values than that in the unstressed controls.
In the reaction of an organism to stress situations, multiple are the factors involved in the actions produced to the maintenance of body homeostasis. There are evidences that elevated levels of circulating maternal catecholamines could impair fetal development indirectly through placental vasoconstriction that reduces oxygen and nutrient supply to the fetus/es.19,20 Romanini et al.21 concluded that the chronically stressed rats had activated the sympathetic-medullary-adrenal axis and increased the levels of catecholamine metabolite, acid 3-metoxi 4-hidroximandélico, in urines at the beginning of pregnancy. Also, high concentrations of corticotropin-releasing hormone (CRH) are found in growth-retarded fetuses and elevated maternal CRH levels are associated with decreased gestational length and increased risk of preterm delivery.22 Conversely, other authors found no effect of prenatal stress on CRH levels.23,24
In animal models, prenatal stress disrupts the normal surge of testosterone in the developing male, whereas in females, the effect on sexual steroid level differs among species studied.25 When immobilization stress is applied to pregnant rats, Mayer et al.26 observed that basal corticosterone and glucose levels were higher in prenatally stressed animals, in agreement with results reported by Vallée et al.27 A group of rats with immobilization stress showed a hyperactive hypothalamic-pituitary-gonadal axis,15,28 alterations in plasmatic LH and testosterone plasmatic levels, testis with reduced weight and higher apoptosis caused by the prenatal stress.29
In humans, exposure of the developing brain to various types of environmental stressors during intrauterine life increases the susceptibility to neuropsychiatric disorders such as autism, attention deficit, hyperactivity disorder and schizophrenia.30 In the same way, likelihood of developing anxiety, depression and learning deficits that are associated with structural alterations in the offspring hippocampus were observed in animals exposed to prenatal stress.31 Chronic social stress during adolescence has been shown to significantly increase basal corticosterone secretion with a flattened circadian rhythm of secretion and enlarged adrenal glands along with a decreased thymus weight. Furthermore, stressed animals display an increased anxiety-like behavior in the elevated plus maze and the novelty induced suppression of feeding test. Hippocampal mineralocorticoid receptor (MR) and the glucocorticoid receptor (GR) mRNA levels were significantly downregulated.32 Adult prenatally stressed rats showed high anxiety-like behavior, as indicated by an escape behavior to novelty correlated with high secretion of corticosterone in response to stress.33 Also, prenatal restraint stress modified reactive and predictive adaptation by altering circadian rhythmicity.34 In the same way, treated rats had greater levels of corticosterone at the end of the light period and especially female animals increased corticosterone values over the diurnal cycle.35
Prenatal stress has been associated with a significant decrease in placental 11β-HSD2 mRNA, increased mRNA levels of the DNA methyltransferase DNMT3a and increased DNA methylation at specific sites within the hsd11b2 gene promoter.36 Additionally, rats that were stressed across generations increased placental microRNA-181a, a marker of human preterm birth.37 Also, in prenatal stress rats, the expression of glucose transporter (GLUT) type 1 was decreased, whereas GLUT type 3 and type 4 were slightly increased.14
The immune response involves a complex and well-orchestrated series of events aimed to protect the host against disease-causing organisms, toxins and malignant cells.38 Most studies coincide that immune suppression is invariably associated with stress situations regardless of animal model or type of stress elicited.
Pregnancy is known to induce a transient depression of maternal cell mediated immunity to prevent rejection of the fetus which is considered an allograft, while at the same time it keeps adequate maternal humoral defense mechanisms to fight infections. It was found that pregnancy appears to be associated with depressed cell immunity, as evidenced by a significant inhibition of lymphocyte proliferation.39 Couret et al.40 found that prenatal stress did not affect the levels of IgG of piglets but decreased the total numbers of white blood cells, lymphocytes and granulocytes, CD4+/CD8+ T cell ratio and lipopolysaccharide (LPS) induced-tumor necrosis factor-α (TNFα) production. Another study conducted by Coussons-Read et al.41 showed that psychosocial and social stress led to higher maternal levels of the proinflammatory cytokines IL-6 and TNF-α and with low levels of the antiinflammatory cytokine IL-10 in pregnant women. In the same line, Elftman ME et al.42 demonstrated that dendritic cells exposed to corticosterone in vivo remained phenotypically and functionally immature after stimulation with LPS with impaired production of interleukin IL-6, IL-12 and TNFα. As a consequence, these effects reduced the ability of dendritic cells to prime naive CD8+ T cells. Studies conducted in our laboratory43 showed that leucocyte, lymphocyte and neutrophil levels and lymphocyte T proliferation were negatively affected in prenatally stress male rats compare to control animals.
Immunity depression was also reported for animals exposed to different types of stressor agents. Hu et al.44 demonstrated that plasmatic ACTH, epinephrine (EPI), angiotensin-II (ANG-II), and IL-10 concentrations were significantly increased in animals exposed to low temperatures (4 and -12°C). On the other hand, cold exposure caused a reduction of IFN-γ, IL-2 and IL-4, the Toll-like receptor (TLR) 4 plasmatic concentration and the percentage of CD4+ CD25+ Foxp3+ Treg cells.
Pascuan et al.45 described that prenatally restraint stressed mice had modified immune homeostasis based on their increased vulnerability to infection and disease. Corticosterone inhibitory effect was higher on lymphocytes from prenatally stress mice, what may be explained at least partially by an increase in protein levels and mRNA expression of GR in lymphoid cells. In the same way, Silberman et al.46 suggested that impaired T-cell dependent humoral response in a model of chronic mild stress is correlated with an increased T-cell sensitivity to stress hormones. Moreover, chronic stress was reported to accelerate the natural ageing of the immune system.47 Also, Li et al.48 described a possible mechanism involved in stress immunosuppression. Corticosterone levels were significantly decreased in TLR 9 deficient mice subjected to chronic stress. When immune parameters were evaluated in this system, they found that TLR9 deficiency blocked the chronic stress induced lymphocyte apoptosis, the imbalance in T helper 1 and T helper 2 cytokine levels. On the contrary, Zhang et al.49 demonstrated that TLR4 is activated by a chronic stressor in mice.
Postnatal early stimulation (PS) involves hand picking the pup and isolated from the mother in a small compartment for several minutes. This practice is repeated daily from birth to weaning.50 The effect of PS appears to be mediated by the influence of maternal licking and grooming on the development of central corticotropin-releasing hormone (CRH) systems, which regulate the expression of behavioral, endocrine and autonomic responses to stress through activation of forebrain noradrenergic systems.51 The effect of PS on hippocampal GR expression appears to be mediated by the activation of the pituitary–thyroid system that leads to an increase in activity of serotonergic pathway during the first week of life.52 In this way, postnatal stress increases plasma levels of triiodothyronine, which potentiates serotonin turnover in the hippocampus and frontal cortex, areas where GR expression is altered by prenatal stress.53,54 Thus, these findings suggest that postnatal PS might alter GR gene expression via cyclic AMP - Protein kinase A (cAMP-PKA) pathways involving the activation of nerve growth factor gene (NGFI-A) and activating protein 2 (AP-2), both factors implicated in the regulation of this receptor during development.55
Active maternal care and environmental stress exert independent effects on HPA reactivity and fearfulness of the offspring. The results obtained by Macrì & Würbel56 suggested that the down regulation of the HPA in response to stressful maternal environments could reflect adaptive developmental plasticity based on the increasing reactivity associated with progressively stressful conditions. Specific experimental manipulations of the rat postnatal environment have been shown to exert robust and marked effects on neurobiological, physiological and behavioral phenotypes in adulthood.57 For example, Chen et al.29 demonstrated that postnatal PS returned the levels of LH and testosterone to normal values and reduced apoptotic index observed in male testes from prenatal stressed animals.
Other studies indicates that PS of neonatal rat pups decreased hypothalamic CRH secretion, enhanced hippocampal GR expression in adult animals and reduced the stress hormone response during acute stress.58,59 Interestingly, PS induces a distinct temporal pattern of gene expression, with reduction in CRH expression from paraventricular nuclei. The same pattern of adult gene expression was induced by postnatal administration of a CRH-receptor antagonist60 implicating that CRH gene is a important molecular mechanism in the emergence of early life programming of the HPA axis function. Adult male rats exposed to early PS exhibit lower basal CRF mRNA and CRF levels in the hypothalamus compared to control animals. The remaining amount of CRF in the median eminence (part of the hypothalamus where regulatory hormones are released) after exposure to 20 min of restraint stress was lower in control animals than those in animals of PS group.59 Another possible mechanism involved in the disruption of corticosterone levels observed in prenatally stressed rats was the altered parameters of adrenal gland physiology. As Liaudat et al.61 showed, the cortex:medulla ratio, apoptotic index and caspase 3 expression of adrenal gland returned to control levels when the prenatally stress animals were handled. Figure 1 shows the timeline of experimental events in a research model aimed to evaluate the effects of prenatal stress and PS on endocrine and immune systems.
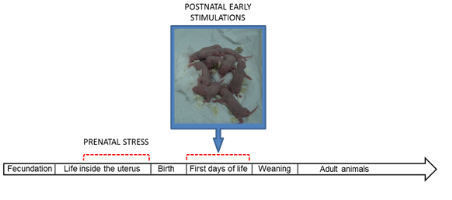
Figure 1Research strategy to study the effects of prenatal stress and postnatal early stimulations on endocrine and immune systems: timeline of experimental events. Prenatal stress in the rat: a model of early programming of stress-related diseases.43
Studies of behavior characteristics conducted in different laboratories rendered contradictory results. Adult handled rats exhibited low anxiety-like conduct, expressed as high exploratory behavior correlated with low secretion of corticosterone in response to stress. Neither prenatal stress nor PS changed spatial learning or memory performance.33 Moreover, Colman et al.62 demonstrated that variations in the neonatal environment affect both behavioral responses and amygdala neuroadaptation to acute withdraw from a palatable diet. Millstein & Holmes63 concluded that in all mice strains studied, maternal separation produced an increase in pups maternal care. The data demonstrated that the maternal separation did not provide a robust model of early life stress effects on the anxiety and depression related behaviors in the mouse strains tested.
On the other hand, Cannizzaro et al.64 reported that long-lasting PS in adult male rats was able to over compensate the increased behavioral stress reactivity induced by the prenatal exposure to diazepam and alprazolam.
There is experimental evidence that supports that the response of the immune system to conditions of chronic stress is associated with increased corticosterone secretion.65 Although Different researcher analyzed the immune parameters altered by PS but the effects produced by this treatment on immune system are currently poorly defined.
One of the first reports was published by Lown & Dutka66 These authors showed that preweaning handling significantly enhanced B- and T-cell proliferative responses. Similarly, our research group has demonstrated that postnatal PS treatment reversed the effect of prenatal stress on leukocyte total numbers as well as lymphocyte and neutrophil percentages. Moreover, when prenatally stressed animals were postnatally handled, the lymphocyte T proliferation was reverted to control levels.43
Another model of early stimulation showed that mild maternal separation in early life increased the stress-resistant phenotype of adult female BALB/c mice which shows reduced repeated stress-induced immune suppression and weight loss. These findings were linked to reduced release of GC after stress exposure.67 Additionally, Milde et al.68 described that brief maternal separation had a protective effect on adult stress exposure and protects the animals from chemically induced colitis. They also observed that handling protects the animals from dextran sulfate sodium induced colitis. Table 1 summarizes the effect of prenatal stress and postnatal handling on selected endocrine and immune physiological parameters.
Parameter |
Prenatal stress |
References |
Postnatal early stimulations |
References |
Dysregulation of HPA axis |
↑ |
Weinstock et al.69 |
↓ |
Smythe et al.70 |
|
|
Lesage et al. 15 |
|
Macrís et al.56 |
|
|
Romanini et al. 21 |
|
Liaudat et al.43 |
|
|
Mayer et al.26 |
|
Desplats et al.71 |
|
|
Schroeder et al.28 |
|
|
Adrenal gland cortex: medulla |
↑ |
Liaudat et al.61 |
↓ |
Liaudat et al.61 |
Adrenal gland apoptosis |
↑ |
Liaudat et al. 61 |
↓ |
Liaudat et al.61 |
Body weight |
↓ |
Pollard et al.72 |
↑ |
Vallé et al.27 |
|
|
Vallée et al.27 |
|
Kiank et al.67 |
|
|
Mairesse et al.14 |
|
|
|
|
García-Cáceres et al.73 |
|
|
Testis weight |
↓ |
Mairesse et al.14 |
↑ |
Chen et al.29 |
|
|
Rodriguez et al.74 |
|
|
|
|
Chen et al.29 |
|
|
Testis apoptosis |
↑ |
Chen et al.29 |
↓ |
Chen et al.29 |
Glucose levels |
↑ |
Vallée et al.27 |
↑ |
Vallé et al.27 |
|
|
Mayer et al.26 |
|
|
|
↓ |
Mairesseet al.14 |
|
|
CRH levels |
↑ |
Wadhwa et al.22 |
↓ |
Plotsky et al.59 |
|
|
|
|
Smythe et al.70 |
|
̶ |
Williams et al.23 |
|
Avishai-Eliner et al.58 |
|
|
Jezová et al.24 |
|
|
ACTH levels |
↑ |
Del Cerro et al.18 |
↓ |
Dobrakovová et al.75 |
|
↓ |
Mairesse et al.14 |
|
|
Testosterone levels |
↓ |
Rodriguez et al.75 |
↑ |
Chen et al.29 |
|
|
Chen et al.29 |
|
|
|
Del Cerro et al.18 |
|
|
|
|
↑ |
Williams et al.23 |
|
|
Glucocorticoid receptors expression |
↓ |
Neeley et al.76 |
↑ |
Smythe et al.70 |
|
|
Maccari et al.34 |
|
Meaney et al.55 |
|
|
Weinstock et al.69 |
|
McCormick et al.77 |
|
|
Smythe et al.70 |
|
Fenoglio et al.60 |
Total leucocytes |
↓ |
Mayer et al.26 |
↑ |
Liaudat et al.43 |
|
|
Liaudat et al.43 |
|
|
|
|
Llorente et al.78 |
|
|
|
|
Dhabbar et al.79 |
|
|
Lymphocytes number |
↓ |
Llorente et al.78 |
↑ |
Liaudat et al.43 |
|
|
Laviola et al.80 |
|
|
|
|
Mayer et al.26 |
|
|
|
|
Liaudat et al.43 |
|
|
|
|
|
|
|
Granulocytes number |
↓ |
Coussons-Read et al.41 |
_ |
Liaudat et al.43 |
|
|
Mayer et al.26 |
|
|
|
|
Liaudat et al.43 |
|
|
|
↑ |
Llorente et al.78 |
|
|
Lymphocytes proliferation |
↓ |
Kay et al.82 |
↑ |
Lown et al.66 |
|
|
Liaudat et al.43 |
|
Liaudat et al.43 |
|
|
Dhabbar et al.79 |
|
|
|
|
Klein et al.81 |
|
|
Table 1 Effect of prenatal stress and postnatal handling on endocrine and immune physiological parameters
It is well known that prenatal stress causes long term modifications in the HPA axis as well as physiological and behavioral alterations in offspring. In particular, immune suppression associated to prenatal stress has been demonstrated by many groups. Interestingly, treatments such as early stimulation or handling during the first week of life were able to induce a long lasting reduction of stress responses in laboratory animal models. For the majority of parameters studied, handling of offspring was able to revert the alterations triggered by prenatal stress; however a few studies were unable to demonstrate a positive effect of handling. Based on the enormous impact of stress-associated diseases in modern life, there is great interest in pinpointing the underlying molecular mechanism of stress. This understanding would lead to development of strategies to prevent and/or treat stress-associated disorders. Information gathered from animals models of prenatal stress may prove to be valuable to identifying interventions, like handling, to reduce the impact of intrauterine stress on physiology and behavior during adult life.
None.
The author declares no conflict of interest.

©2018 Liaudat, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.