Journal of
eISSN: 2475-5540


Research Article Volume 6 Issue 2
European Wellness Academy, Germany
Correspondence: Dr. Volodymyr Chernykh, European Wellness Academy, Klosterstrasse 205, Edenkoben, Germany
Received: February 12, 2020 | Published: March 17, 2020
Citation: Chernykh V, Nallenthiran L, Yefimov S, et al. Novel technique of aesthetic skin rejuvenation with cellular extracted compounds enriched with peptides and hyaluronic acid. J Stem Cell Res Ther. 2020;6(2):64-68. DOI: 10.15406/jsrt.2020.06.00142
The structural composition of the skin is dynamic, exhibiting various signs of aging such as the decrease in collagen and elastin content leading to increase in tactile and visual roughness, dryness, wrinkles, fine lines and stiffness as age advances. This study was done to elicit the efficacy and potency of mesotherapy using a cocktail of triple strength nano-sized peptides from the skin, placenta, mesenchyme fortified with collagen and hyaluronic acid (HA) in the revitalisation of the skin of the dorsum of the hand. Ten male subjects suffering from mild signs of aging of the skin were treated with the formulation of MF+ SPMC + HA over the dorsum of both hands. The treatment course spanned over eight weeks with once a week application. Baseline assessment was done before the treatment and at the end of the first and second month and 90 days post-therapy, respectively, using photography, ultrasound comparison and biophysical parameters of the skin. The results proved the application of SPMC + HA rendered long-term hydration and improved the viscoelasticity of the treated area. The study concluded that mesotherapy application of MF+ SPMC extracts/peptides in combination with hyaluronic acid could be considered as a safe and effective method for rejuvenation of the skin of the dorsum of hands.
Keywords: mesotherapy, skin rejuvenation, cell therapy, stem cells, aesthetic medicine, cellular extracts
Beauty may lie within the skin’s depth, but there is more to the skin than it appears on the surface. The physiological function of human skin is numerous, and some diseases of the skin might not just be skin deep.1 Being the largest organ in the body, the skin is an essential flag of systemic illnesses.2 The skin is engineered with the complex anatomical composition, which caters numerous vital functions.2
The pathologies of skin are vast, and aging is one of the most common and inevitable conditions which displays both pathological and cosmetic symptoms (visible and tactile) such as dryness and roughness. These could be explained by the age-related decrease in collagen and elastin content of the skin, leading to impaired structural integrity and eventually the formation of deep and superficial creases and fine lines.3 Microscopic analysis of aging skin would reveal common pathological changes among which includes significant exhaustion of the epidermal HA, flattening of the epidermal-dermal junction, reduced cell turnover rate, decreased melanocytes and their unusual activities.4 These along with intracellular matrix and collagen dysfunction lead to physically visible conditions including dehydration, loss of skin turgor, skin atrophy, pigmentations, lentigos and keratosis. Skin peptides and HA have been implicated in age-related skin conditions.5‒9 Well-hydrated and youthful skin retains its turgor, elasticity and suppleness. The water content of the skin is a pivotal factor to regulate and maintain optimal physiological function.10 The ultimate pathophysiology in aging has been explained by cellular and DNA damage by free radicals as well as mitochondrial dysfunction.11
Hyaluronic acid
Hyaluronic acid is most abundantly found in skin, accounting to about 50% of the total body content. 12 followed by the vitreous of the eye,13 umbilical cord14 and the synovial fluid.15 The HA or hyaluronate is a unique non-sulphated glycosaminoglycan, a relatively large molecule weighing up to 7 million Dalton 16,17 found commonly in connective, epithelial and neural tissues.14,15 The repeating polymeric disaccharides of D-glucuronic acid and N-acetyl-D-glucosamine linked by a glucuronidic β(1→3) bond makes HA a unique molecule that forms numerous different types of stable polymers with an array of physiological properties, depending upon the configurations and shapes, size, salt concentration, pH and associated cations.18 In the human body, hyaluronic acid is produced by the mesenchymal cells, primarily the peripheral connective tissues. The key feature of this molecule is its ability to release the high number of water molecules even at low concentration and high viscosity state.19 As skin moisturisation is vital in achieving skin beauty, commercial skin care products should include active compounds such as hyaluronic acid, which can bind water to attain water retention.20
The era of cellular extracts
The consumption of placenta for cosmetic and therapeutic purposes is dated. Laboratory trials of placental extracts date back to the 30s by Filatov V.P, who demonstrated the halt of disease progression and rapid progress of healing by using placental extract.21 Since the ’60s, women in China and Thailand have been documented to consume placenta from young mothers, which they believed is rich in an array of bioactive compounds that are therapeutically beneficial.22 Subsequently, in the 1970s, the same practice had been found in a small number of women in the United States of America and Mexico. 23,24 published excellent results with fetal mesenchyme – even in long-lasting defects, which were not successfully treated by other therapies. Based on his experience in large scalds and burns, F. Schmid developed a fetal tissue ultrafiltrate for long-term topical applications, which was prepared by the extraction of fetal connective tissue, fetal skin placenta and adrenal gland. Two multicentric trials were designed to determine the list of indications. The following indications were listed for the fetal cutaneous extract: burns, scalds, irradiation burns on the skin injuries, explosion injuries, keloids, hyperkeratosis, anhidrosis syndromes, ulcus cruris, senile atrophy of the skin, trophic disturbances in circulatory trouble, Lyell syndrome, cosmetic lesions, keloids, sclerodermia.24,25
Four decades of research and development of cell extracts witnessed a breakthrough in the area. The therapeutic properties of cell extracts were further expanded in the world of aesthetics and anti-ageing and were seen to be widely used in India, Japan, Korea, Thailand and the United States of America.26‒30 Subsequent years saw a different group of researchers and clinicians documenting positive therapeutic outcome using various kinds of placental preparations and components.31‒34 These include placental tissue, amniotic and chorionic membranes, amniotic fluid, umbilical cord, placental extracts and lyophilizates and cord blood serum, as well as number of differentiated cells and stem cells administered through different routes such as oral, subcutaneous and intramuscular.35‒41
The biological properties of placental peptides have been studied according to its active component. Instead of using the tissue, researchers have lysed placenta of full-term to obtain the bioactive components, an array of proteins, minerals, amino acids, and steroid hormones.31 Besides cosmetic reasons, these extracts have been successfully used in the treatment of wounds, non-healing ulcers, and burns. Researchers documented an accelerated rate of reepithelialisation and reduced infiltration and pain syndrome.36,42 The past two decades are marked by invention and vast application of aesthetic formulations of organ-specific extracts from skin, placenta, and mesenchyme enriched with collagen via cold-enzymatic processing with further nano-filtration of biologically active ingredients since 2003. Nano-peptides show high bioavailability, biocompatibility and injectability due to the linear size of the peptides below ten nm and molecular weight below ten kDa. The abovementioned advantages not only increase the biomedical applications of the nano-sized product but provide a unique rejuvenating and revitalising effect of this compelling tissue therapy, which can be applied either via mesotherapy or topically.
The new dawn
Aewsthetic dermatology has been of significant interest in recent years and it expands beyond the facial skin rejuvenation. The entire human body garnered much attention in this regard, with an increasing concern on the hands, especially the skin of the dorsum. A casual handshake can reveal the state of one’s skin. Rough, wrinkly and dry skin of the dorsum remains a major concern despite the emergence of store-available hand care products. As such, the revolutionary discovery of cellular extracts is promising to address the abovementioned issues. The application of peptides and HA has been proven to successfully reverse the aging manifestation of the skin.43‒45 This combination works synergistically in halting aging process as well as to stimulate regeneration of the tissue. Various types of skin peptides embedded in a meshwork of hyaluronic network provide an optimal micro-environment for bioactive molecules.46‒48 An important key feature of the peptides in this cocktail is the ability to stimulate regeneration of fibroblasts and keratinocytes as well as collagen, elastin and extracellular matrix synthesis.49,50 Besides being an excellent hydrating agent, the HA used in this preparation renders the structural integrity of dermal collagen.51 HA has also been successfully used as a vector for targeted drug delivery, ensuring a more extended bioavailability.52
Objective
The objective of this study was to identify the efficacy, potency and safety of mesotherapy with nano-sized peptides from the skin, placenta and mesenchyme fortified with collagen and HA “SPMC + HA” in treating aging signs of the surface of the dorsum of the hand.
This case study was performed at the clinical research facilities of the European Wellness centres. A total of 10 male patients with no significant concurrent illness were invited in the study. All 10 underwent eight treatment sessions using the cellular extracts concoction known as “SPMC + HA” (MF+ brand, Germany) and took part in the follow-up session. All patients were Asian; the mean age standard deviation was 48 ± 5,6 years old. The used formulation “SPMC + HA” by MF+ brand (Germany) contains cells extracts from the skin, placenta, and mesenchyme with addition of pro-collagen peptides blended with HA. These compounds are dispersed in physiological saline solution (pH 7.0). Treatment sessions were performed across eight weeks at weekly interval.
All patients were asked not to apply any local aesthetic treatments and to refrain from any form of commercial food supplements with rejuvenating and skin firming effects during the treatment. A follow-up visit was scheduled 90 days after the first treatment. A topical anaesthetic cream EMLA (Recipharm Karlskoga AB, Sweden) was applied to the area to be treated, and all patients were administered with injection in seated positions. Tiny amounts of the tested product (approximately 0.03 – 0.04 ml) were injected intradermally after the disinfection with an aqueous solution of chlorhexidine using a 33G needle (Beaumed, Korea) with a papule technique. The spacing between two injections was approximately 0,5 - to 1.0 cm apart; around 1 ml of the product was injected into the skin of the dorsum of both hands per session over an area of about 11*9 cm.
Assessments
Assessments were performed at the baseline, after the one, two months and the 90-day clinical follow-up visit. The next biophysical parameters were evaluated: stratum corneum hydration, measured using a capacitance device (the Corneometer CM825, Courage & Khazaka, Cologne, Germany); skin thickness, investigated ultrasonographically, using the Ultrascan C22 (Courage & Khazaka, Cologne, Germany)); and skin elasticity, using a Cutometer dual MPA 580 (Courage & Khazaka, Cologne, Germany). Three elasticity parameters were assessed: skin extensibility (the maximum extent to which the skin is stretched after applying a standard force, R0), gross elasticity (a measure of the ability of the skin to return to its original position after the force is off, R2), and viscoelasticity (the ratio of extensibility caused by the skin’s viscous vs elastic components, R6). The measurements were conducted under air-conditioned conditions (temperature 22.5±1.5°C; relative humidity 50±10%) after an acclimatisation period at least 20 minutes. Digital photographs were taken to evaluate treatment success at the 90-day follow-up treatment using a Canon IOS R camera (Japan). Every patient was photographed at the same distance from the camera using standardized light conditions. Patients also provided written consent for the photographs to be published. Adverse events observed by the investigator or reported by the patient were recorded. Data were summarized, and analysis of variance was used to compare means. Pair-wise comparisons controlling for the family-wise error rate were done using the Tukey test (pr≤.05).
Hydration
Moisturizing increased continuously during the study period. Statistically significant gradual increases from baseline in capacitance values were obtained during the trial period (p <.05 and p<.001) (Figure 1), indicating that treatment increased horny layer hydration. Degree of hydration increased more than 16% from baseline to the fourth treatment visit (p<.001) and more than 20% from baseline to the eighth treatment visit. They were maintained until at least 30 days after the last treatment visit (p<.05 vs baseline at D60 and D90). Dermal thickness ultrasound measurements showed a profound increase in skin thickness over the study period.
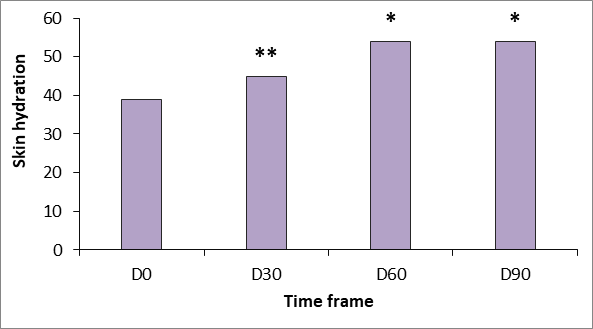
Figure 1 Changes in stratum corneum hydration measured using the Corneometer CM825 (Courage & Khazaka, Germany). *p<.05 versus baseline; **p<.01 versus baseline.
Skin thickness
Ultrasound measurements reflect a continuous increase in dermal thickness within the study period (Figures 2 and 3). Increases over baseline were statistically significant from 30 days after the first treatment (p<.05) and were highly significant (p<.001) at D60 and D90.
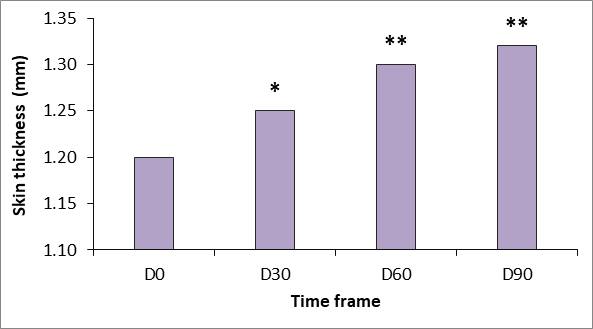
Figure 2 Changes in skin thickness measured using Ultrascan 22 (Courage & Khazaka, Cologne, Germany).* p<.05 versus baseline; **p<.01 versus baseline.
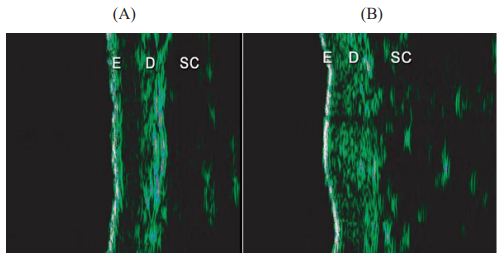
Figure 3 Ultrasound images were taken at (A) baseline (D0) and (B) 90 days after initial treatment (D90) using Ultrascan 22, (Courage & Khazaka, Cologne, Germany).
The increased echogenicity that was evidenced at the end of the treatment is related to an increased density of dermal collagen fibres resulting from treatment.
Elasticity
Generally there was a good clinical response to the tested product “SPMC + HA” characterized by dramatic improvement of the gross elasticity of the skin: after pressure was removed from the suction device, the skin comes back to its original position much faster after treatment (p<.05 vs baseline at D 90). Skin extensibility showed a significant reduction from D30 to D90 (p<.05). Dermal viscoelasticity was significantly decreased throughout the trial (p<.05 at all-time points vs baseline), indicating that the skin behaved more like an utterly elastic substance in response to being stretched (Figure 4-6).
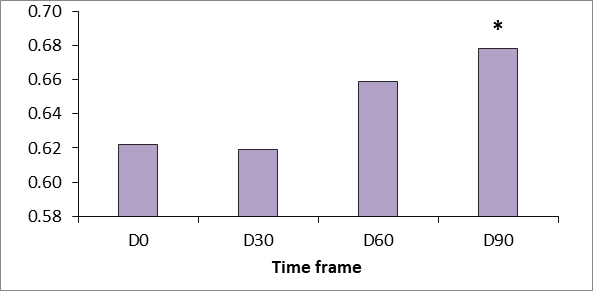
Figure 4 Gross elasticity measured using the Cutometer dual MPA 580 (Courage & Khazaka, Germany). p <0.5.
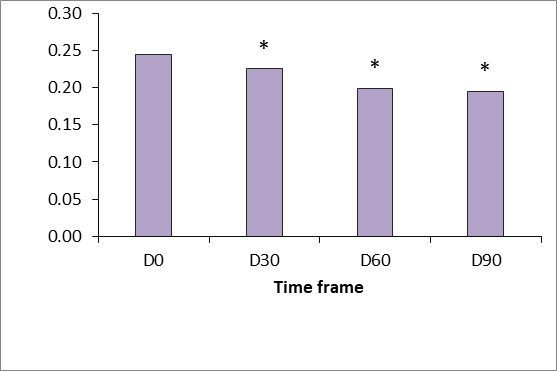
Figure 5 Gross viscoelasticity measured using the Cutometer dual MPA 580 (Courage & Khazaka, Germany). p <0.5.
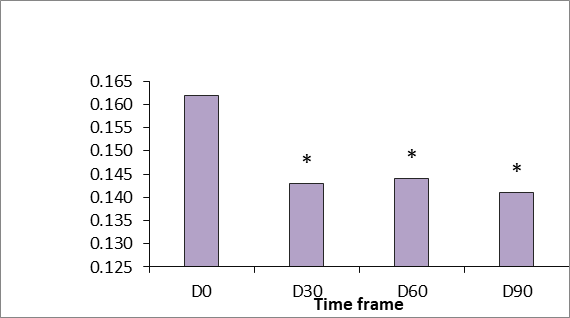
Figure 6 Gross skin extensibility measured using the Cutometer dual MPA 580 (Courage & Khazaka, Germany). p <0.5.
Clinical photographic evaluation
Comparisons of photographs taken before and after treatment showed that the skin on the backside of both hands looked much more hydrated, radiant and smoother.
Adverse events
Product was well tolerated. Unexpected adverse events weren’t reported. All expected treatment-related adverse events, such as pain and discomfort at the injection site, bruising, and the hematoma were transient.
Overall the achieved outcomes of the “SPMC + HA” clinical application were therapeutically and cosmetically laudable. The clinical outcome of wrinkle-free, supple, vitalised as well as rejuvenated and pigment-free skin of the hand’s dorsum is the expected result of this novel strategy. Treatment resulted in a gradual thickening of the dermis, and increased hydration of the horny layer and the enhancement in the gross elasticity of the skin was enhanced. These biophysical changes transformed into visible changes with the decrease in skin wrinkles during the clinical follow-up period. The tested product “SPMC + HA” (MF+, Germany) provides long-lasting and promising results of mesotherapy. This combination should be made available as one of the primary therapeutic approach in treating aging changes of the skin, taking into consideration the existing guidelines. Further studies to investigate and demonstrate the longer-term effects of “SPMC + HA” for facial rejuvenation with multiple treatment cycles and investigate the efficacy of combining treatment modalities would be of interest.
Authors declare no conflict of interests and no direct commercial benefit from this publication.

©2020 Chernykh, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.