Journal of
eISSN: 2373-6437


Research Article Volume 17 Issue 3
1Anesthesiologist at various hospitals, Rio de Janeiro, Brazil
2Anesthesiologist at the National Cancer Institute (INCA), Brazil
3Head of the Anesthesiology Service of the National Cancer Institute (INCA) Anesthesiologist, Responsible for the CET-SBA of the National Cancer Institute, Brazil
4Professor of Anesthesiology at the UFG Faculty of Medicine, Responsible for the CET-SBA of the HCUFG, Brazil
5Graduated from Pontifical Catholic University of Goiás, Brazil. Fellowship in Integrative Medicine at the University of Arizona, USA
6Master’s in labor economics, UFPB, João Pessoa-PB, PhD student in applied mathematics and modeling, Universidade Aberta de Lisboa, Portugal
7Resident in Anesthesiology and Pediatrics, Brazil
Correspondence: Dr. Luiz Eduardo Imbelloni, Anesthesiologist at various hospitals, Av. Epitácio Pessoa, 2356/203, Lagoa, 22411-072- Rio de Janeiro, RJ – Brazil, Tel + 55.11.99429-3637
Received: July 11, 2025 | Published: July 23, 2025
Citation: Imbelloni LE, Rivoli ALC, Neto SVL, et al. Evaluation of the density of 0.5% Bupivacaine and its association with various adjuvants in the same syringe. an experimental study in laboratory. J Anesth Crit Care Open Acces. 2025;17(2):96-100. DOI: 10.15406/jaccoa.2025.17.00627
Background: All drugs have their own density. The baricity of the solutions injected into spinal anesthesia is an important factor for their dispersion. Various adjuvants are added to local anesthetic solutions for spinal anesthesia in the same syringe. The objective of this laboratory study using a DMA 4500 densimeter was to determine the density of the mixture obtained from anesthetic and adjuvant of various anesthetists who use it in plastic surgeries at 20°C, 25°C, and 37°C. Second objective to compare with the baricity of the CSF, through the published media.
Methods: The density of the mixtures was evaluated at three temperatures, the solution being hyperbaric if its density exceeds 1.00099, hypobaric when the density is below 1.00019 and isobaric when the density is greater than 1.00019 and less than 1.00099. For each mixture, a sufficient volume was obtained to make two measurements of the densities, and the average density value was obtained with two evaluations. The second part of the study was to evaluate the baricity (δ drug÷δ CSF of 37oC), compared with the density of the CSF. The final concentration of LA and glucose in the mixture were evaluated.
Results: Nine different mixtures were found. All mixtures exhibited a decrease in density as the temperature was increased. At 37°C, all solutions containing glucose are hyperbaric. In the absence of glucose, all solutions are hypobaric. There was a decrease in the concentration of bupivacaine from 0.5% to between 0.27 and 0.37%, and in the glucose concentration from 8% to between 4.3 and 5.9%.
Conclusion: The density of the mixtures obtained decreases with increasing temperature in the same way as the baricity of the final solution. Likewise, the mixture of LA and various adjuvants reduced the concentrations of bupivacaine and glucose produced by the laboratory. The ideal for spinal anesthesia safety in humans would be for each drug to be injected separately into its own syringe, with no change in the characteristics of each drug.
Keywords: Bupivacaine, fentanyl, clonidine, sufentanil, morphine, syringe, baricity, densimeter 4500
Question
What is the reason for placing local anesthetics and adjuvants in the same syringe to perform spinal anesthesia?
Meaning
One of the most important physical properties affecting the level of analgesia obtained after subarachnoid injection of a local anesthetic (LA) is its density relative to the density of cerebrospinal fluid (CSF) at 37°C. In a study carried out in 2009, with the aid of a DMA 4500 densimeter with a sensitivity of ±0.00001 g/mL, the densities of all local anesthetics on the market and the adjuvants used in this technique were evaluated. This study showed that all hyperbaric or hypobaric anesthetics are modified by the addition of adjuvants but remain hyperbaric or hypobaric. The results demonstrated that some solutions commonly called isobaric are in fact hypobaric. And that all adjuvants used in the subarachnoid space when evaluated at 37°C are all hypobaric.1
In a recent article, all the possibilities of the different types of sensory and motor blocks were demonstrated, after injections of hypobaric, isobaric and hyperbaric solutions, in the seated puncture position, in lateral decubitus and in the jack-knife position (posterior). However, the results of the three local anesthetic solutions and the different puncture positions were demonstrated throughout the article, without any association with any type of adjuvant. In this article, it was argued that the injection of LAs and adjuvants should always be in separate syringes, since this combination in the same syringe modifies the baricity of this combination, and the result may be different from when used in the same syringe.2
In an article showing the various possibilities for increasing safety in the use of spinal anesthesia, 14 steps were selected to prevent complications.3 They explain in detail the use of asepsis and antisepsis, the use of different types and calibers of needles, the non-use of off-label products, and most importantly, not mixing local anesthetics with adjuvants in the same syringe. Maintaining these procedures after completing 50 years of profession, I have never had a complication or even a process while using any type of spinal anesthesia.
In 1,330 procedures with lumbar puncture with a sensory level reaching C3 or C4 with the mixture of 0.5% hyperbaric bupivacaine (20 mg), clonidine (150 µg) and sufentanil (5 µg), it was shown to be an effective option for surgeries on somatic structures distal to the metamer of the third cervical spinal nerve lasting no longer than 4 or 5 hours.4 From this study, several anesthetists used the same mixture or several other mixtures of local anesthetic with different adjuvants, obtaining the same result.
After the disclosure of the technique called "lumbar spinal anesthesia with cervical nociceptive blockade", mainly for plastic surgeries in Brazil, nine mixtures of 0.5% hyperbaric bupivacaine and 0.5% isobaric bupivacaine with different adjuvants in the same syringe were selected and was the objective of this laboratory study. This mixture of hyperbaric anesthetic with hypobaric adjuvants and always injected in the sitting position certainly changes the total density of the mixture.
The aim of this laboratory study with a densimeter (DMA 4500) was to determine the density of solutions injected into the subarachnoid space at temperatures of 20°C, 25°C and under clinical conditions, at a temperature of 37°C, and in association with adjuvants and the mixture of local anesthetic with more than one adjuvant, according to the mixture used in the same syringe. A secondary objective was to evaluate the baricity of the solutions used by comparing the densities with the same density of the cerebrospinal fluid (CSF) used in other studies.5 The third objective was to evaluate the concentrations of LA and glucose in the mixtures found in the research.
The first stage of the study was to verify how many mixtures are used by anesthesiologists. Since this study was carried out in a laboratory with a densimeter (Anton Paar DMA 4500 Paar Scientific Ltd., London, UK) therefore without the participation of human beings simulating the mixtures used by various anesthetists, the study therefore dispenses with the registration of the Brazil platform and the need for approval by CEP/CONEP. The densities of the various mixtures of hyperbaric local anesthetics with various adjuvants were determined at temperatures of 20°C, 25°C and 37°C, and the results were issued with five decimal places and considering the unit of measurement of g/mL. The solutions used for the study have their commercial names, active components, batch and manufacturing, all produced by the Cristália laboratory (Table 1).
|
Trade Name |
Component |
Batch |
Expiration |
|
Neocaína 0.5% ® |
Cl. bupivacaine 5 mg/mL + Glu 8% |
50024292 |
01/2027 |
|
Neocaína 0.5% ® |
Cl. bupivacaine 5 mg/mL plain |
50016142 |
06/2027 |
|
Dimorf ® |
Sulfate morphine 0.1 mg/mL |
50023006 |
12/2026 |
|
Fentanest ® |
Fentanyl 0.05 mg/mL |
50023926 |
01/2027 |
|
Fastfen ® |
Sufentanyl 5 µg/mL |
50023721 |
01/2027 |
|
Fastfen ® |
Sufentanyl 50 µ/mL |
50024222 |
01/2027 |
|
Clonidin ® |
Cl. clonidine 150 µg/mL |
50020338 |
10/2026 |
Table 1 Drugs used in the study with their components, batch and expiration date
In a consultation of several anesthesiologists who practice the technique mainly for plastic surgery, nine ways of combining anesthetics and adjuvants to obtain spinal anesthesia were found. These solutions were named with numbers from 1 to 9, to facilitate the study and to explain in detail which products were (Table 2). The first part of the study was designed to measure the density of all mixtures (nine in total) obtained through consultation with the various anesthetists who use the technique, with the drugs taken from a batch at temperatures of 20°C, 25°C and 37°C. For each mixture, a sufficient volume was obtained to make two measurements of the densities, and the average density value was obtained with two evaluations.
|
Mix (n) |
Local Anesthetic |
Bari- City |
Dose (mg) |
Clo (µg) |
Fenta (µg) |
Sufe (µg) |
Mo (µg) |
Vol (mL) |
Bupi % |
Glu % |
|
1 |
0.5% Bupi |
Hyper |
20 |
150 |
20 |
No |
No |
5.4 |
0.37 |
5.9 |
|
2 |
0.5% Bupi |
Hyper |
20 |
150 |
No |
5 |
No |
6.0 |
0.33 |
5.3 |
|
3 |
0.5% Bupi |
Hyper |
20 |
150 |
No |
No |
60 |
6.0 |
0.33 |
5.3 |
|
4 |
0.5% Bupi |
Hyper |
15 |
No |
No |
2.5 |
200 |
5.5 |
0.27 |
4.3 |
|
5 |
0.5% Bupi |
Iso |
15 |
No |
No |
2.5 |
200 |
5.5 |
0.27 |
0 |
|
6 |
0.5% Bupi |
Hyper |
20 |
75 |
No |
No |
100 |
5.5 |
0.33 |
5.8 |
|
7 |
0.5% Bupi |
Hyper |
20 |
150 |
No |
No |
100 |
6.0 |
0.30 |
5.3 |
|
8 |
0.5% Bupi |
Hyper |
20 |
150 |
No |
2.5 |
No |
5.5 |
0.36 |
5.8 |
|
9 |
0.5% Bupi |
Hyper |
20 |
150 |
No |
10 |
60 |
5.8 |
0.34 |
5.5 |
Table 2 Drugs used in each mixture, final volume and concentration of bupivacaine and glucose
Mix, Mixtures; Bupi, Bupivacaine; Bari, Baricity; Sol, Solution; Clo, Clonidine; Fenta, Fentanyl; Sufe, Sufentanil; Mo, Morphine; Vol, Volume.
The second part of the study was to evaluate the baricity (δ drug÷ δ CSF of 37oC), which was determined by the relationship between the density of the nine mixtures at 37°C and the density of the CSF based on an article on density evaluation.5 The mixtures were considered hypobaric when the density was below the minimum limit of the confidence interval (1.00019) of the CSF density. The solution was considered hyperbaric when the density of the solution exceeded 99% of the confidence limit of the CSF density (1.00099). The solutions were considered isobaric when their densities varied between 1.00019 and 1.00099. The third part of the laboratory study was to evaluate the final concentration of bupivacaine and glucose when present.
Given the exploration nature of the present laboratory study, the small sample size (n = 9) and the presence of ties and potential outliers, nonparametric methods were used. To evaluate the effect of temperature on the density of anesthetic solutions in temperatures 20oC, 25oC and 37oC, the Friedman test was used, and Wilcoxon test with Bonferroni correction. The concentrations of bupivacaine, glucose and adjuvants, in relation to the density at 37°C, Spearman's correlation was used. A P-value ≤ 0.05 indicates a significant difference.
Consultation with several anesthesiologists revealed nine mixtures used in the various types of plastic surgeries. All nine mixtures used are shown in detail with all their components (Table 2). Of the nine mixtures, eight were made with 0.5% hyperbaric bupivacaine and only one with 0.5% isobaric bupivacaine. Of the nine mixtures, seven contained 20mg and two 15mg of 0.5% bupivacaine. Similarly, eight contained 320mg, one 240mg of glucose, and one was 0.5% plain bupivacaine (Table 2). There was a decrease in the concentration of bupivacaine from 0.5% to between 0.27% and 0.37%, and in the glucose concentration from 8% to between 4.3% and 5.9%.
|
Mixtures |
20oC (g/mL) |
25oC (g/mL) |
37oC (g/mL) |
P-Value |
|
1 |
1.02314 |
1.02180 |
1.01776 |
P < 0.001 |
|
2 |
1.02111 |
1.01988 |
1.01582 |
|
|
3 |
1.02125 |
1.01993 |
1.01591 |
|
|
4 |
1.01824 |
1.01694 |
1.01296 |
|
|
5 |
1.00450 |
1.00328 |
0.99945 |
|
|
6 |
1.02265 |
1.02127 |
1.01769 |
|
|
7 |
1.02126 |
1.01991 |
1.01589 |
|
|
8 |
1.02267 |
1.02130 |
1.01719 |
|
|
9 |
1.02173 |
1.01994 |
1.01590 |
Table 3 Evaluation mean densities of mixtures at three temperatures
Increasing the temperature decreased the densities of the 0.5% hyperbaric bupivacaine solutions, but all remained hyperbaric (Table 3). Similarly, with only one mixture, increasing the temperature decreased the densities of 0.5% isobaric bupivacaine, but remaining isobaric (Table 3). The study showed that any amount of glucose in the local anesthetic would always transform it into a hyperbaric solution. Increasing the temperature significantly decreased the density of the hyperbaric solution (eight mixtures) and the hypobaric solution (one mixture) (Figure 1).
The addition of adjuvants in the same syringe decreased the concentration of bupivacaine from 0.5% to between 0.27% and 0.37% (Table 3, Figure 2). Neocaína® 0.5% heavy is an injectable solution with 4mL of the solution and each mL contains bupivacaine hydrochloride 5mg with glucose 80mg. Therefore, the 4ml ampoule contains 20mg of the active ingredient and 320mg of glucose, and 15mg it has 240mg. Thus, the addition of the various adjuvants decreased the glucose concentration between 4.3% and 5.9% (Table 3, Figure 2). The 0.5% isobaric bupivacaine solution does not contain glucose and was hypobaric at 37°C (Table 3, Figure 2). At 37°C, all anesthetics with glucose are hyperbaric, regardless of the glucose concentration. The addition of any adjuvant to hyperbaric solutions decreases their baricity, but they remain hyperbaric. At 37°C, all isobaric anesthetics are hypobaric. The addition of any adjuvant to isobaric anesthetics further decreases the baricity of these solutions, making them hypobaric.
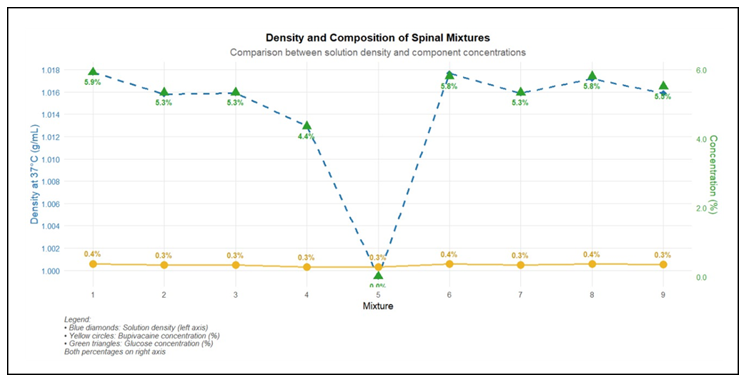
Figure 2 Showing the concentrations of bupivacaine and glucose in eight solutions and isobaric bupivacaine plain.
|
Mixtures |
δ = 37oC (g/mL) |
Baricity |
Solution |
|
1 |
1.01776 |
1.01715 |
Hyperbaric |
|
2 |
1.01582 |
1.01552 |
Hyperbaric |
|
3 |
1.01591 |
1.01531 |
Hyperbaric |
|
4 |
1.01296 |
1.01236 |
Hyperbaric |
|
5 |
0.99945 |
0.99886 |
Hypobaric |
|
6 |
1.01769 |
1.01708 |
Hyperbaric |
|
7 |
1.01589 |
1.01529 |
Hyperbaric |
|
8 |
1.01719 |
1.01659 |
Hyperbaric |
|
9 |
1.01590 |
1.01530 |
Hyperbaric |
Table 4 Evaluation of the baricity of the mixtures at 37oC compared with the average density of 1.00059.5
Test: Spearman's rank correlation coefficient (ρ)
Adjuvants are drugs which, when administered along with LAs agents, may improve the latency of onset and duration of analgesia and counteract the undesirable effects associated with large doses of LAs. The use of adjuvant drugs has the potential to improve the efficacy of central neuraxial blocks and decrease LA systemic toxicity by chiefly prolonging the duration of sensory block, enhancing motor blockade, and limiting the overall dose requirement of LAs.6 Increasing the temperature from 20°C to 25°C or 37°C corresponded to a significant decrease in the density of the nine mixtures used by the anesthesiologists. At 37°C, all solutions containing glucose are hyperbaric. In the absence of glucose, all solutions are hypobaric. There was a decrease in the concentration of bupivacaine from 0.5% to between 0.27 and 0.37%, and in the glucose concentration from 8% to between 4.3 and 5.9%. Thus, the mixture presents new components, which are different from when injected separately.
The baricity of LAs refers to the relative density of these anesthetics when compared to that of the CSF. Baricity affects the distribution and spread of the solution, influencing the characteristics of the blockade. Study in laboratory mixing local anesthetics and adjuvants in the same syringe alters the density.1 This laboratory study measured the densities of nine mixtures of 0.5% bupivacaine with and without glucose added to various adjuvants in the same 10mL syringe frequently used in spinal anesthesia, at three temperatures, using a state-of-the-art densimeter (DMA 4500) that uses a mechanical oscillation technique by resonance and its precision is ±0.00001 g/mL. The result showed that increasing the temperature decreased the density of all mixtures, also decreasing the concentration of bupivacaine and glucose when present. When compared with the CSF density, the baricity decreased with increasing temperature.
The baricity and temperature of local anesthetics are important factors affecting cranial spread in spinal anesthesia.7 The density of the human CSF is not uniform, and it can vary with age, gender, pregnancy, and several diseases; similarly, the baricity can also vary. However, this study was carried out in a laboratory so that, through the results obtained, it would be possible to know what was being injected into the subarachnoid space with all these mixtures found. This study has never been carried out before. The first spinal anesthesia’s were performed with pure cocaine, therefore considered isobaric. In 1907 glucose was added to cocaine, making hyperbaric solution the world's preferred solution for spinal anesthesia. Studying the effects of intrathecal injection of 0.5% bupivacaine in solutions containing different concentrations of glucose (0.33%, 0.83% and 8%), showed that the rate onset and the extent of blockade was significantly higher with the 8% glucose solution.8 The addition of 1% and 5% glucose to S75:R25 levobupivacaine demonstrated that high concentrations of glucose are not necessary to turn local anesthetics into hyperbaric solutions.9 Therefore, these two studies showed that any concentration of glucose added to the local anesthetic or adjuvants makes the solution hyperbaric. This laboratory study with DMA 4500 showed that the mixture decreased both the LA concentration in the hyperbaric and isobaric presentations. Similarly, there was a decrease in the glucose concentration in the mixtures with hyperbaric bupivacaine.
The practice of mixing different drugs in the same syringe is common, with the aim of reducing costs and facilitating the anesthesiologist's work, but the quality and safety of this procedure are still important issues.10 None of the anesthesiologists who use the mixtures in the same syringe know the density, baricity, LA concentration and glucose concentration. And knowledge of the density, the final concentration of LA, the glucose concentration (hyperbaric), or pure LA (isobaric or hypobaric) and without off-label adjuvants should be part of safety.3
Baricity is the relationship between the density of a solution and the density of the CSF. When the baricity of a solution is 1.0000, the solution is isotonic; > 1.0000, it is hyperbaric; and if < 1.0000, it is hypobaric. It has been suggested for some time that the solutions used in spinal blocks should be considered hypobaric when their densities are lower than the lower limit of the confidence interval of the density of the human CSF.5 Several studies already published that adding opioids or any other adjuvants (all hypobaric) to the local anesthetic solution in clinically relevant doses may affect the final density of such compositions.1,5,7,11 The result of separate intrathecal injection of fentanyl and hyperbaric bupivacaine provided better hemodynamic stability, better quality of surgical anesthesia and minimal side effects in patients undergoing cesarean section.11 However, the authors did not perform the same study comparing the two solutions in the same syringe.
The ratio of the density of LA and CSF, which is known as baricity, is one key determinant of LA distribution within the subarachnoid space. Measurements of CSF density have previously been in various articles, and relationship to various parameters. Recently, 14 steps for safe spinal anesthesia were published, avoiding neurological complications and sequelae.3 Among the various suggestions is the use of each syringe for each drug, avoiding the use of all substances in the same syringe. Likewise, avoid the use of off-label anesthetics or even those without factory sterilization. This laboratory study showed that any local anesthetic used for spinal anesthesia, when added to one or more adjuvants in the same syringe, completely alters the drugs when they are injected into separate syringes. Increasing the temperature from 20°C to 25°C or 37°C corresponded to a significant decrease in the density of the nine mixtures used by the anesthesiologists. Likewise, the mixture of LA and various adjuvants reduced the concentrations of bupivacaine and glucose produced by the laboratory. This laboratory study with nine mixtures obtained showed that there is a modification in all the parameters evaluated compared to the drugs alone. The result of the study showed that the important thing is the safety of the solutions used and not saving time or reducing costs by using all the drugs in the same syringe. This laboratory study with a DMA 4500 densimeter confirms that the pharmaceutical industry can reduce the concentrations of local anesthetic and glucose in hyperbaric solutions, for use in spinal anesthesia.
Thanks to Laboratório Cristália Produtos Quimicos e Farmacêuticos Ltda for providing the products with their batches and expiration dates, for carrying out the tests on the Anton Paar DMA 4500 densimeter.
Study carried Central Analítica, Instituto de Química Universidade Estadual de Campinas-UNICAMP, SP.
None.
None.

©2025 Imbelloni, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.