Journal of
eISSN: 2572-8466


Review Article Volume 13 Issue 1
1Department of Biology, California State University, USA
2Department of Bioengineering, University of California, USA
Correspondence: Bill Tawil, Department of Bioengineering, UCLA School of Engineering, 420 Westwood Plaza, Room 5121, Engineering V. P.O. Box: 951600, Los Angeles, CA 90095-1600, USA
Received: December 08, 2025 | Published: January 7, 2026
Citation: Vanessa M, Bill T. Controlling life at scale: the evolution and significance of bioreactors in biotechnology. J Appl Biotechnol Bioeng 2026;13(1):1-4. DOI: 10.15406/jabb.2026.13.00409
Bioreactors have been integral to the biotechnology process since at least 7000 B.C., initially employed for producing alcoholic beverages. In modern applications, bioreactors serve as critical systems in biotechnology by providing controlled environments that optimize key parameters in addition to fulfilling their primary role as containment vessels. Evaluating the effects of these parameters is essential for understanding their influence on culture production and protein activity. This review examines the principal operational parameters of bioreactors and analyzes how parameter- specific data impact the efficiency and outcomes of cell culture processes.
Keywords: bioreactors, pH, dissolved oxygen, temperature, cell culture
DO, dissolved oxygen; CHO, Chinese hamster ovarian; EPO, erythropoietin; VCD, viable cell density (); BGA, blood gas analyzer; PAT, process analytical technology
The utilization of bioreactors has been essential to a successful biotechnical process since at least 7000 B.C.1 Their use dates back to producing alcoholic drinks for analgesic, sanitary and social purposes.2 Today, bioreactors play a central role in biotechnical operations due to their ability to provide optimal conditions. These optimal conditions include parameters such as temperature, pH and substrate condition, for example, besides its basic function of containment.3 By evaluating the effects of these parameters, we are able to examine their important roles in culture production and protein activity.4 In this review article, we will provide the important parameters of a bioreactor and how their data affects the process of cell culture overall.
The history of bioreactors
Evolution
Before the biotreactor became what it is today, it was known for being a conventional fermentor.5–8 The history of bioreactors spans thousands of years, as early as 7000 BC when they were being used to ferment mixtures such as honey, rice, and fruit.9 This was considered humanity’s first attempt to harness microorganisms for food preservation and improved flavor. Similar fermentation vessels began to emerge as civilizations advanced. Ancient Iran, Mesopotamia and Egypt adapted their own technology to produce products like wine, beer, bread.10
As modern bioreactors started to develop, their advances in microbiology became more transparent. They had many milestones, marking their importance to biotechnology. In the late 1800s to early 1900s, the bioreactors transitioned from clay to metal with simple temperature control.11 By the 1940s industrial upscaling of bioreactors was driven due to the urgent need of penicillin mass production during World War II.12
It has continued to also have many impacts in the food industry. Articles mention Louis Pasteur as he was the first to popularize the idea of fermentation.6 The famous French chemist and microbiologist studied the fermentation process when French vintners were producing vinegar instead of wine.7 His work paved the way for bioreactors today.
Today, bioreactors are amongst the most sophisticated pieces of equipment we use in biotechnology; they have continued to support continuous production and efficient scale-up and scale-down strategies while also addressing sterilization and safety requirements.8 We are continuously seeing the evolution of bioreactors as we see their diversity in size and parameters.
Types
The reason for the evolution of bioreactors stems from their different uses; there are many types and designs of bioreactors that have different applications: continuous stirred- tanks airlift, single use bioreactors.13 The continuous stirred- tank bioreactors were designed and introduced as a way to achieve good mass transfer and mixing by utilizing mechanical stirrers.14 Airlift, bubble column, and fixed bed bioreactors on the other hand were used for fermentation applications with shear- sensitive microorganisms such as filamentous fungi; bubble culm is carried through mixing by aeration in one direction while airlift uses both the riser and down- comer section of a bioreactor.15 Waves and single use bioreactors were introduced for batch- based fermentation applications such as mammalian cell fermentation.16 The difference in the mechanisms of these bioreactors can be seen in Figure 1.17,18
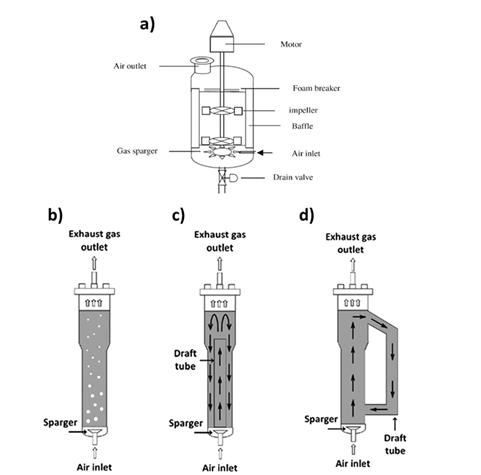
Figure 1 Common bioreactor designs. a.) continuous stirred- tank reactor. b.) bubble column reactor. c.) internal loop of airlift bioreactor. d.) external loop of airlift bioreactor.13
Parameters of a bioreactor
Bioreactors have been parameters that contribute to the overall health and living conditions of our cell culture.19 Choosing the right bioreactor parameters is very crucial due to the sensitivity of the cells.20 Some of these important parameters are: pH, temperature, and dissolved oxygen- just to name a few.2
pH
A key step in a bioprocess is monitoring and regulating the pH of the cell culture as it gives great predictions of the development of the cells.22,23 Mammalian cells specifically are viable in pH between 7.1 and 7.4, but many cell processes will fluctuate pH as they remove C02 to prevent toxic buildups.24 The pH does not fluctuate randomly, however; fluctuation of pH is caused by changes in other parameters as seen in Figure 2. As a run continues, it is important to know the optimal pH for the system to therefore monitor it in order to keep the cells healthy and under optimal conditions for optimal protein production.25
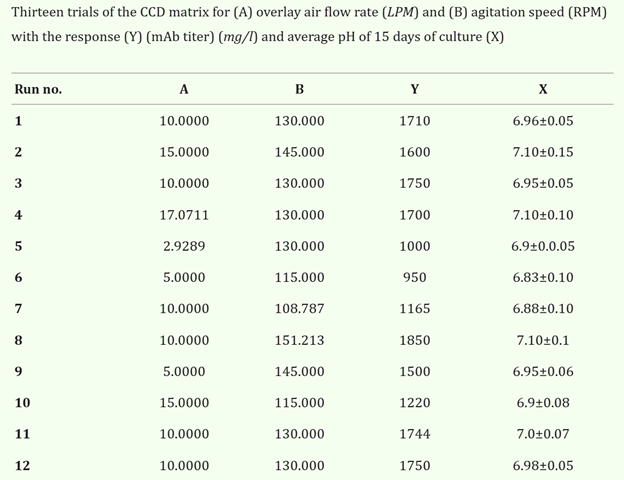
Figure 2 Results of pH as parameters such as agitation and gas flow are varied.26
Temperature
Figure 2 does not show the effects of temperature, but temperature is also an important variable when growing viable cells.21 A study in Korea showed how cell growth was optimized when the temperature was changed; according to the study, CHO were tested in multiple conditions varying in pH and temperature- an increase of 3 fold increase was seen in EPO production when the temperature was changed from 37 to 32.5°C.27 This does not mean that 32.5°C is an ideal temperature for all types of cell culture, however; the range between 37°C and temperature that grows viable cells can be as big as 37°C - 28°C depending on the cell line.28 A shown on Figure 3, different temperatures affect cell growth differently depending on the day the sample of cells was taken. Knowing these ranges is helpful because temperature is not only used to increase cell production but also suppress it if needed.29 Depending on the cell line, cells have a different doubling time; this doubling time is slowed down when the culture temperature is changed.31 This means that by using bioreactors, we are able to efficiently change the temperature in order to reach either optimal conditions or slow down cell growth.32
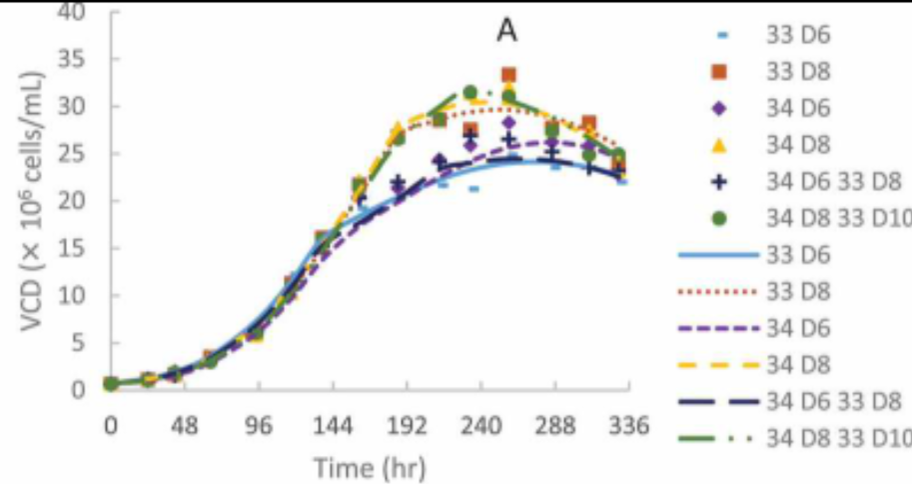
Figure 3 VCD of CHO varying in temperature on Days 6-10 of the run.30
Dissolved oxygen
Similar to pH, dissolved oxygen can also be measured through online sensors and confirmed using offline readings like BGA.33 Oxygen is a common variable measured in cell culture, so it is very important to keep levels within acceptable range.34 It’s monitored since it is a limiting substrate for cell growth owing to its low solubility in water.35 To maintain efficient levels, bioreactors will ensure efficient oxygen transfers from the gas phase to the liquid and ultimately into the cells.36 Every cell culture is different and has sensitive ranges to their dissolved oxygen intake. No matter the type of culture, however, it is important that the system housing the cells has sufficient oxygen for their growth. Figure 4 shows just how important maintaining oxygen is, as it’s essential for cellular processes. For this reason, it is important to have sufficient oxygen to ensure the culture’s viability. This further supports how bioreactors work to maintain optimization in the scale up process of fermentation to ensure quality cells.37–41
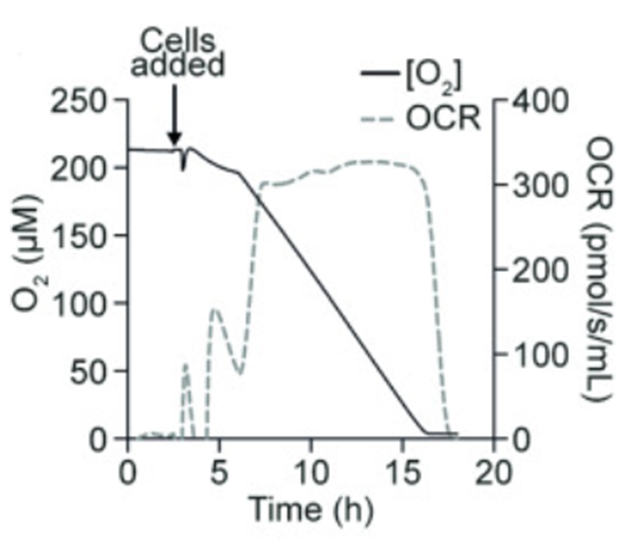
Figure 4 A drop in oxygen is shown when cells are introduced to the system.42
Accessories to bioreactors
Probes
All stable readings for these parameters can be achieved by probes. Bioreactors have great pH sensors with an accuracy of +/- 0.1 which allows for a representative reading of the cell culture inside the bioreactor.23 These probes are calibrated and then autoclaved in order to maintain sterility within the system.43 Their small size makes it very convenient and compliments the use of bioreactors.34 Online readings are very important as they give operators real-time readings on parameters. It is important to keep these readings accurate, however. For this, operators compare online readings to offline readings to ensure dependency.
BGA
An offline reading is essential to determining true pH and DO values since their readings are more accurate. Running a sample through a blood gas analyzer (BGA) will give operators insight on direct pH, partial pressure of oxygen, and partial pressure of carbon dioxide.44 The BGA allows for accurate readings that can then be used to standardize our online probes through one point standardizations. If there is a large difference between the online and offline readings, the online readings can be standardized to the offline readings in order to ensure accuracy throughout the duration of the run. The one point will allow the online probes to recognize true values and a new sample will be taken the next day to ensure the probes are still reading accurately.
The future of bioreactors
For decades, there has been a discussion about the use of stainless steel versus single use bioreactors. To this date, there is no argument between cost effectiveness, safety, or efficiency of the reactors themselves.45 There is, however, a difference in their use in the manufacturing process.
Stainless steel has been seen since the beginning of bioreactors given their customizable size.45 It is not until recently that single use bioreactors have been increasingly used in the pharmaceutical industry.46
Single use bioreactors bring many benefits that are not seen when using stainless steel. By using single use, industries are seeing lower risk of cross-contamination, suppression of the costly wash and sterilization steps between production campaigns, time savings in the building and validation of new production facilities and more flexibility in the production capacity.47
Studies also indicate that single use bioreactors can make facilities more sustainable by lowering their CO2 footprint compared to the traditional stainless steel. Research has found that single use technology requires about 50% less energy than stainless due to their reduced cleaning and sterilization at large volumes. Stainless steel also consumes far more resources like large amounts of water for their sterilization which is reduced when compared to single use bioreactors since single use utilizates disposable bags.48
Bioreactor performances play an important role in biotechnology as it is crucial to maintain optimum living conditions for cell cultures.38 Using online and offline measurements like pH, temperature, and dissolved oxygen makes it easy to ensure conditions are met within our system.39 We are continuing to see multiple additions to the traditional bioreactor like introducing PAT to see real time quality, but it is obvious the impact that bioreactors have made to the industry throughout their evolution.40
Vanessa Medina expresses appreciation to Professor Bill Tawil for overseeing the framework of this review, and for the insightful lectures and advice concerning biomaterials and tissue engineering, which contributed to the development of this paper
There is no funding to report for this study.
Authors declare that there is no conflict of interest.

©2026 Vanessa, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.