Journal of
eISSN: 2572-8466


Research Article Volume 13 Issue 1
1Department of Parasitology, Universidad Autónoma Agraria Antonio, Calzada Antonio Narro #1923 Buenavista, Saltillo ZP 20230, Coahuila, México
2Environmental Microbiology Laboratory, Institute of Chemical- Biological Research, B3 B, University City. Universidad Michoacana de San Nicolás de Hidalgo, Fco J Mujica S/N, Col. Felicitas del Rio, ZP 58030, Morelia, Michoacán, México
Correspondence: Juan Manuel Sanchez-Yañez, Environmental Microbiology Laboratory, Institute of Chemical-Biological Research, B3 B, University City. Universidad Michoacana de San Nicolás de Hidalgo, Fco J Mujica S/N, Col. Felicitas del Rio, ZP 58030, Morelia, Michoacán, México
Received: January 14, 2026 | Published: February 2, 2026
Citation: Gallegos-Morales G, Rodriguez-Rodriguez JE, Peña-Ramos FM, et al. Aspergillus niger solubilizes phosphates with a positive effect on the healthy growth of Solanum lycopersicum and Capsicum annuum. J Appl Biotechnol Bioeng. 2026;13(1):6-11 DOI: 10.15406/jabb.2026.13.00410
The application of chemical fertilizers to agricultural soil impacts the production costs of plant crops, and in excess, deteriorates soil quality. Phosphorus as phosphates (PO4-3) is a compounds that, at contact with the soil, can react with aluminum (Al), calcium (Ca), and iron (Fe) to form insoluble phosphates. However, there are microorganisms capable of solubilizing insoluble Ca3(PO4)2 (phosphates) into soluble ones. Among these is the genus and species: Aspergillus niger, which, along with other microorganisms, promotes healthy plant growth. A. niger synthesizes organic acids in the rhizosphere of plants to solubilize phosphates, besides some phosphatases that are then uptake, thus promoting plant health growth and productivity. This process utilizes phosphates from the soil, based in this fact the aims of this research are: i) to determine the phosphate-solubilizing capacity of A. niger in liquid and solid NBRIP culture medium; ii) to analyze the effect of soluble phosphorus, spores, and mycelium of A. niger on the healthy growth of Capsicum annuum and Solanum lycopersicum iii) molecular identification of the phosphate-solubilizing A. niger isolated. For this purpose, the phosphate-solubilizing capacity was tested on NBRIP agar, where it demonstrated a solubilization index of 2.25 mm at pH 6. In liquid NBRIP, enhancing the Ca3(PO4)2 concentration increased the concentration of soluble phosphorus generated, with a positive correlation of 0.8729 between Ca3(PO4)2 and 72 h of agitation. It was also demonstrated that the application of soluble phosphorus generated by A. niger to seedling of S. lycopersicum and C. annuum promoted height, number of leaves, and stem diameter, and reduced phosphorus deficiency in both compared to the same plants without this treatment. A. niger was morphologically and molecularly identified. Therefore, it is concluded that A. niger is an ecological alternative for optimizing phosphate fertilizers applied to the soil for healthy plant growth.
Keywords: soil, phosphate solubilization, healthy plant growth, phosphate solubility index, Kps or solubility constant
In general, the soil contains reserves of insoluble phosphorus as Ca3(PO4)21 In part due to applications of phosphorus fertilizers, as well as (NH4)2HPO4 this is PO4-3not uptake by the root system of plants, because of the soil pH and the narrow solubility product constant or (Ksp).2,3 It is well known that one possible ecological solve for this agricultural problem, could be to apply some phosphate-solubilizing microorganisms, that by different solubilization mechanisms, releasing soluble phosphates, that could be uptake by plant roots, promoting plant healthy growth and productivity.4,5 Capsicum annuum and Solanum lycopersicum are two commercially valuable plant species that, in besides nitrogen fertilizer, require PO4-3.6,7 The problem of this inorganic chemical compounds is that reactions with soil cations according to pH, that increased agricultural production cost.8–10 An ecological alternative for solving this problem is to apply or to inoculate soil or plant seed with Aspergillus niger or its releasing products related with PO4-3 solubilization to increase its availability by acidified soil pH with organic acids, that it releases into the soil, as well as through chelation and the synthesis of acid and alkaline phosphatase to promote PO4-3 uptake by plant roots of C. annuum and S. lycopersicum.11–14 Therefore, the objectives of this research were: i) to determine the phosphate-solubilizing capacity of A. niger in liquid and solid NBRIP culture medium; ii) to analyze the effect of soluble PO4-3 combined with A. niger spores and mycelium on the growth of C. annuum and S. lycopersicum and iii) to morphologically and molecularly identify the phosphate-solubilizing A. niger isolated.
Location
This research was conducted in the Microbiology and Greenhouse Laboratory of the Parasitology Department at the Universidad Autónoma Agraria Antonio Narro, in Buenavista, Saltillo, Coahuila, Mexico 25° 22” N and 101° 02” W; 1742 m above sea level.
Origin and identification of phosphate-solubilizing A. niger
An A. niger isolated was used, recovered from the rhizosphere of Muhlenbergia macroura (grassgrass), and morphologically identified based on mycelium type and microscopic reproduction. It was found to be capable of solubilizing Ca3(PO4)2 in NBRIP (National Botanical Research Institute Phosphate Growth Medium) described in literature15–19 with the following chemical composition (g/L): Ca3(PO4)2 5, MgCl26H2O 5, MgSO4 7H2O 0.25, KCl 0.2, (NH4)2SO4 0.15, dextrose 10, agar 18, a medium specifically designed for the isolation of phosphate-solubilizing microorganisms.3,4 Species confirmation was performed by sequencing the internal transcribed regions ITS1 and ITS4 of the rDNA amplified by rtPCR12,17 LANBAMA of the Potosino Institute of Scientific and Technological Research (IPICYT). San Luis Potosi State of México.
Determination of the phosphate-solubilizing capacity of Aspergillus niger in liquid and solid NBRIP
This test was performed in Petri dishes with NBRIP agar at pH values of 4, 5, 6, and 7; each pH value was considered a treatment. A 5 mm diameter portion of culture medium containing 48 h of A. niger mycelial growth was inoculated into the center of the NBRIP. The NBRIP dishes were incubated at 28 °C with a 12:12 light/dark photoperiod.18,19 Solubilization was measured using a digital vernier caliper.20 The solubilization halo of Ca3(PO4)2 in A. niger was measured in mm every 24 h until the entire surface of the Petri dish was covered.21–24 A completely randomized design was used with four pH values and three replicates per treatment or pH value. The numerical values were subjected to an analysis of variance (ANOVA) by the statistical software SAS version 9.4. The mean values of Ca3(PO4)2 solubilization efficiency were compared and stratified according to Tukey's test (α = 0.05). At the end of the experiment (168 h), the solubilization index (SI) for each pH value was determined using the following formula: SI = microorganism diameter + solubilization halo / microorganism diameter.23–25 For the determination of phosphate solubilization in liquid NBRIP, 2000 ml baffled Erlenmeyer flasks were used, containing 700 ml of Ca3(PO4)2 with the following concentrations: T1 = 1.0 g/L, T2 = 2.0 g/L, T3 = 3.5 g/L, and T4 = 5.0 g/L. Each flask was sterilized and inoculated with a 5 mm diameter portion of A. niger mycelial culture medium with 72 h of growth. The flasks were shaken at 150 rpm for 72 h at 30°C and a 12:12 light: dark photoperiod. A completely randomized design with four treatments or concentrations was used.23–25 The determination of Ca3(PO4)2 was carried out with the Hanna Instruments H1706 colorimetric phosphorus high range kit, with a 10 ml sample of each concentration of Ca3(PO4)2: by the colorimetric reaction that determines the solubilization of Ca3(PO4)2 generated by A. niger in the culture medium with a Thermo Spectronic Spectrophotometer (Figure 1).12,16,18
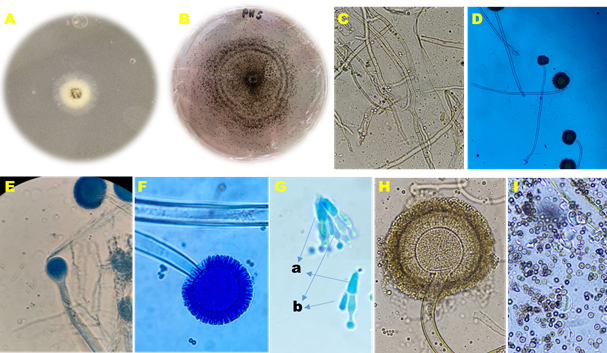
Figure 1 Morphological structures of A. niger: A) yellow mycelial growth after 48 h, B) black mycelial growth after 168 h, C) septate mycelium, D) long conidiophores. E) vesicle. F) vesicle surrounded by metulae and phialides, G) a- metulae and b- phialides, H) vesicle with metulae, phialides, and spores, I) dark brown spores.
Phosphate solubilizing capacity of A. niger on NBRIP agar
Effect of soluble phosphorus, spores, and mycelium of A. niger on the growth of C. annuum and S. lycopersicum.
Seeds of S. lycopersicum variety Floradade and C. annuum hybrid Platino were sown in 200-cell polystyrene trays with a 3:1 mix of peat-moss and perlite. Eighteen days after germination, 16 seedlings of each variety, reaching a height of 5 cm, were selected and transplanted to a 60-cell polystyrene tray containing a 1:3 mix of peat-moss and soil for the phosphate-solubilizing experiment; with a completely randomized experimental design with three treatments and an absolute control: T1= soluble phosphorus of Ca3(PO4)2 solubilized by A. niger, T2= spores of A. niger 2 x 107 x ml, T3= mycelium of A. niger, T4= absolute control or water; with four repetitions per treatment, total of 16 experimental units shown in Table 1, for both C. annuum and S. lycopersicum. Treatments 1 to 3 were applied 10 days after transplanting (DAT), and repeated at 20, 30 and 40 DAT. The following phenological response variables were evaluated: plant height, stem width, number of leaves and number of leaves with PO4-3 deficiency, using a completely randomized block design. The experimental results were subjected to ANOVA and Tukey's mean comparison test (P=0.05) with the statistical program InfoStat version 2019.1.2.0.15,16,18
|
T2 |
T1 |
T4 |
T3 |
|
T1 |
T2 |
T3 |
T4 |
|
T4 |
T3 |
T2 |
T1 |
|
T3 |
T4 |
T1 |
T2 |
Table 1 Experimental design to analyze the effect of soluble phosphorus, conidia and mycelium of A. niger on the growth of C. annuum and S. lycopersicum
In Table 2, it was registered that the solubilization of Ca3(PO4)2, was directly related to the growth of A. niger, represented by uppercase letters, while lowercase letters indicate the type of treatment. At 48 h, the numerical values showed a statistically difference. In T3, the greatest solubilization of Ca3(PO4)2, was registered with 12.43 mm. At 120 h and 168 h of incubation, there was no statistically difference. The treatment with the largest solubilization halo was A. niger T3, shown in Figure 15C with 66.56 mm, in Table 2 with an SI of 2.25 mm, followed by A. niger or T4 with a diameter of 65.48 mm registered in Table 2 with an SI of 2.24 mm observed in Table 3 and Figure 15D. In third place, A. niger or T1 with a diameter of 61.78 mm shown in Table 2 with an SI of 2.09 mm, in Table 3 and Figure 15 A and finally A. niger or T2 with a diameter of 60.87 mm registered in the table with an SI of 2.08 mm shown in Table 3 and Figure 15 B, these results were similar to those reported in literature3,7,12 with a strain of A. niger with a growth of 15 mm in diameter and a Ca3(PO4)2, solubilization halo with an SI of 4.3 mm in 14 days, unlike this research an SI of 2.25 shown in Table 2, in 7 days, of A. niger isolated from the rhizosphere of a wild grass that had a greater growth than that reported.18,22–25 The growth of A. niger and the diameter of the Ca3(PO4)2 solubilization halo were related to the genetic capacity to solubilize Ca3(PO4)2 phosphate, which depends on the synthesis of organic acids released from the sugars they use as a source of carbon and energy. Another genetically induced mechanism is the formation of acid and alkaline phosphatases that depend on the pH of the environment or the culture medium. These are produced to release Ca3(PO4)2from other substances that prevent them from using it as a source of P for energy production, as well as for the growth of the fungus, especially when inoculated in a plant culture where the soil does not allow the uptake of Ca3(PO4)2 for healthy plant growth (Figure 2).26–28
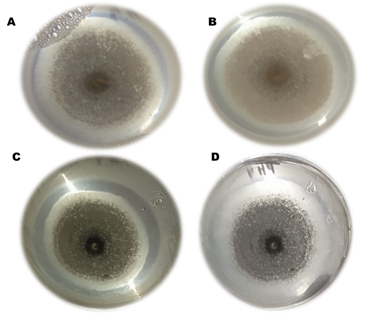
Figure 2 Solubilization test of Ca3(PO4)2 by A. niger after 168 h of incubation in NBRIP at different pH values. A) T1, NBRIP at pH 4, B) T2, NBRIP at pH 5, C) T3, NBRIP at pH 6 and D) T4, NBRIP at pH 7.
|
|
Treatments (mm) |
|
|
||||||||||||||
|
Hours |
T1 |
|
|
T2 |
|
|
T3 |
|
|
T4 |
|
|
P>F |
C.V |
|
||
|
24 |
5.73 |
ab |
G |
5.70 |
b |
G |
5.75 |
ab |
E |
7.77 |
a |
E |
0.01 |
6.19 |
|
||
|
48 |
11.39 |
ab |
F |
10.93 |
ab |
F |
12.43 |
a |
E |
9.83 |
b |
E |
0.01 |
6.08 |
|
||
|
72 |
18.76 |
b |
E |
18.92 |
ab |
E |
22.32 |
a |
D |
18.88 |
a |
D |
0.03 |
6.19 |
|
||
|
96 |
31.62 |
cb |
D |
29.88 |
c |
D |
38.77 |
a |
C |
32.33 |
b |
C |
0.001 |
2.37 |
|
||
|
120 |
43.79 |
a |
C |
45.04 |
a |
C |
47.38 |
a |
CB |
49.88 |
a |
B |
0.07 |
5.16 |
|
||
|
144 |
49.42 |
a |
B |
52.40 |
a |
B |
52.90 |
a |
B |
54.04 |
a |
B |
0.06 |
3.24 |
|
||
|
168 |
61.78 |
a |
A |
60.87 |
a |
A |
66.56 |
a |
A |
65.48 |
a |
A |
0.46 |
7.66 |
|
||
|
Pr>F |
0.001 |
|
|
0.001 |
|
|
0.001 |
|
|
0.001 |
|
|
|
|
|
||
|
C.V. |
3.19 |
|
|
4.22 |
|
|
9.25 |
|
|
4.54 |
|
|
|
|
|
||
Table 2 Phosphorus solubilization of Ca3(PO4)2by A. niger at different pH in NBRID Agar
*Means with a common letter were not statistically different (p >0.05).
|
|
Mycelial growth (mm) |
Solubility index (SI) |
|
T1 |
56.46 |
2.09 |
|
T2 |
56.04 |
2.08 |
|
T3 |
53.01 |
2.25 |
|
T4 |
52.78 |
2.24 |
Table 3 Mycelial growth and Ca3(PO4)2 solubilization index of A. niger in NBRIP after 168h of incubation
Table 3 shows that the growth of A. niger in T1 and T2 showed greater mycelial growth with 56.46 mm and 56.04 mm respectively, greater than the growth of A. niger in T3 and T4, which reached greater IS with 53.01 mm and 52.78 mm with similar phosphate solubility indices by A. niger associated with the synthesis of carboxylic acids derived from the organic carbon source, the concentration and organic or inorganic type of nitrogen source, the pH and the initial amount of Ca3(PO4)2 in the solid or liquid culture medium.3,4,12
In Figure 3 it shown standard curve of soluble phosphorus where colorimetrically was quantify the concentration of soluble phosphorus generated by A. niger when grown in NBRID with Ca3(PO4)2, a standard curve of soluble phosphorus was initially prepared using a spectrophotometer with K2HPO4 at different phosphorus concentrations, As observed in the colorimetric analysis of phosphorus, a proportionality was registered the determination, indicated by a correlation coefficient greater than 0.92, suggesting a correlation between the phosphorus concentration in solution and the color detected by the spectrophotometer at 880 nm.
Phosphate-solubilizing capacity of A. niger in NBRIP Broth
In Figure 4 is show that when A. niger was grown in NBRIP broth in shaken flasks with different concentrations of Ca3(PO4)2, the concentration of soluble phosphorus in the NBRIP broth supernatant was analyzed spectrophotometrically using the standard curve with K2HPO4 as a reference (shown in Figure 3). The soluble phosphorus concentration increased with increasing Ca3(PO4)2 concentration.8,10 The highest concentration of phosphorus solubilized by A. niger was found in the culture medium with the highest Ca3(PO4)2 concentration of 5 g/L, These results differed from those reported,7,10–12 whom observed that increasing the Ca3(PO4)2 concentration decreased the concentration of soluble phosphorus in the strain of Pseudomonas sp P2S culture medium.13,14,16 While reported a similar result to that of this investigation, with a Brevibacillus brevis that reached a phosphate solubilization diameter in solid culture medium with a correlation of 0.9987 where with the longer incubation time greater Ca3(PO4)2 solubilization in a directly proportional manner.20–23

Figure 4 Liquid NBRIP culture medium with different concentrations of Ca3(PO4)2 before and after inoculation with A. niger.
Figure 5 Effect of soluble phosphorus, spores, and mycelium of A. niger on growth of S. lycopersicum at seedling stage. In Table 4 and Figure 5 is shown why phosphorus as PO4-3 is essential for plant growth, nucleic acid synthesis during cell division, and ATP generation for all celular activity.2–6,23,24 In Table 4 A. niger under different concentrations of insoluble Ca3(PO4)2 caused some results statistically similar on the growth of S lycopersicum although numerically different, S. lycopersicum showed greater growth than when grown with Ca3(PO4)2 without A. niger or T4. Treatment T1 showed the greatest height of S. lycopersicum at 18.38 cm, with 30.75 leaves, similar in stem diameter to T2, that had the largest stem diameter at 3.35 mm. Meanwhile, when S. lycopersicum was treated with soluble P or T1, the lowest number of leaves with phosphorus deficiency symptoms was registered 0.50 (shown in Figure 5), statistically different from the leaves of C. annum without soluble P or A. niger or T4, used as a control, that registered the highest number of phosphorus deficiency leaves 6.50. The above is supported by the benefits of inoculation with phosphate-solubilizing microorganisms and application of soluble phosphate, as reported11,12,16,23 whom inoculated S. lycopersicum with A. niger, arbuscular mycorrhizal fungi that increased phosphorus content and improved growth compared to S. lycopersicum uninoculated with A. niger. Meanwhile reported6,18,27–29 that applying the phosphate-solubilizing genus and species of Brevibacillus brevis to Physalis peruviana increased leaf area by 3.2 cm², with a greater stem diameter, compared to P. peruviana uninoculated with soluble P10,13,20,22 reported the effect of two phosphate-solubilizing rhizobacteria, KCH3 and TSACH2, from the rhizosphere of C. annum var. habanero on S. lycopersicum.1–4 Both bacteria increased plant height to 14.29 cm and fresh weight to 128 mg with KCH3 and to 12.63 cm and fresh weight to 119 mg with TSACH24,7,15,25 compared to a control plant of S. lycopersicum uninoculated or soluble phosphorus, that had a height of 10.51 cm and a fresh weight of 90 g. Similarly reported that the use of Ca3(PO4)2 solubilizing microorganisms promotes the growth of Solanum tuboserum, resulting in a greater number of stems, 3 to 5 times larger stem diameters, and more leaves without phosphorus deficiencies, compared to uninoculated S. tuboserum.27–29

Figure 5 Seedlings of S. lycopersicum with: A) S. lycopersicum seedlings with soluble phosphorus, spores and mycelium of A. niger 40 days after sowing. B and C) S. lycopersicum leaves from the absolute control, with symptoms of phosphorus deficiency due to purple discoloration of the veins
|
Treatments |
Plant height (cm) |
Steam diameter (mm) |
Number leaves
|
Number leaves with P deficiency |
|
T1- soluble P |
18.38 ± 0.77a* |
3.16 ± 0.12a |
30.75 ± 2.04a |
0.50 ± 0.43b |
|
T2- conidia |
17.30 ± 0. 77a |
3.35 ± 0.12a |
30.50 ± 2.04a |
1.50 ± 0.43b |
|
T3- mycelium |
16.88 ± 0.77a |
2.91 ± 0.12ab |
28.75 ± 2.04a |
2.00 ± 0.43b |
|
T4- control |
16.13 ± 0.77a |
2.59 ± 0.12b |
25.25 ± 2.04a |
6.50 ± 0.43a |
Table 4 Effect of the application of conidia, mycelium of A. niger and phosphorus soluble on the growth of S. lycopersicum to seedling stage
*Means with a common letter were not statistically different (p >0.05).
Effect of applying conidia, mycelium, and phosphorus solubilized by A. niger on the growth of C. annuum at the seedling level. In the Table 5 and Figure 6 is show the experiment applying conidia, mycelium, and phosphorus solubilized by A. niger to the phenology of C. annuum at the seedling level, all treatments: T1 to T3 were statistically similar but statistically different from the control or T4, that was neither treated not treated with phosphorus. However, there was a numerical difference, as shown in Table 5. Treatment 3, when C. annuum was treated with A. niger mycelium, reached the greatest height with 9.13 cm, followed by T1, that, when treated with soluble phosphorus, reached a height of 8.98 cm. Treatment T1 also showed the greatest stem diameter with 1.92 mm, the largest number of leaves 11.25, and the fewest leaves exhibiting phosphorus deficiency 0.2. While C. annuum with soluble P or T1 and T2 inoculated with A. niger conids were statistically and numerically different from C. annuum used as a control without P or A. niger elements, that showed less growth and a greater number of leaves with obvious symptoms of phosphorus deficiency,10,15,16,24 according to Figure 6 compared to applying Ca3(PO4)2 solubilizing microorganism in other vegetable crops such as C. annuum as been reported30–32 indicate that Serratia plymuthica solubilizes phosphates in C. annuum var. poblano, in that promoting an increase in plant height to 9.0 cm compared to C. annuum uninoculated with 7.8 cm; as also has reported that Burkholderia ambifaria and B. lata, solubilizing phosphate from phosphate rock in C. annum var. poblano, reduced the recommended phosphorus dose without compromising the healthy growth of the plant.33–35 In the same way it is reported that inoculating Aspergillus sp in Coffea arabica resulted in a 6.25 cm increase in plant height compared to uninoculated C. arabica with solubilizing phosphate.15,20,23,30,32

Figure 6 Effect of soluble phosphorus, spores, and mycelium of A. niger on growth of C annuum at seedling stage
T1) soluble phosphorus, T2) conidia, T3) A. niger mycelium and T4) absolute control with not growth.
|
Treatment |
Plant height cm |
Steam diameter mm |
Number of leaves |
Number leaves with P deficiency |
|
T1- soluble P |
8.98 ± 0.50a* |
1.92 ± 0.04a |
11.25 ± O.37a |
0.25 ± 0.26b |
|
T2- conidia |
8.25 ± 0.50ab |
1.86 ± 0.04a |
10.25 ± 0.37ab |
0.25 ± 0.26b |
|
T3- mycelium |
9.13 ± 0.50a |
1.91 ± 0.0a |
9.50 ± 0.37bc |
0.50 ± 0.26b |
|
T4- control |
6.38 ± 0.50b |
1.46 ± 0.04b |
8.00 ± 0.37c |
2.25 ± 0.26a |
Table 5 Effect of conidia, mycelium and phosphorus solubilized by A. niger on the growth of C. annuum at seedling stage
* Means with the same letter with no statistical difference (p >0.005).
The results registered with the C. annuum and S. lycopersicum treatment with soluble P (T1) reached the highest values in the phenology of the two plant species, with the exception of height in C. annuum, where C. annuum and S. lycopersicum with the A. niger mycelium (T3) were numerically lower than T1. This is most likely because when phosphorus is applied in soluble form, the plant uptakes and assimilates it quickly, compared to the other treatments. A. niger is adapted for the growth of compounds necessary for the solubilization of Ca3(PO4)2 through organic acids and acid and/or alkaline phosphatases. Although A. niger in the soil has the advantage of efficiently solubilizing Ca3(PO4)2 for the root system of plant crops (Cr), as in C. annuum or S. lycopersicum with A. niger mycelium or T3, that reached the greatest plant height compared to the single application. The soluble phosphorus in treatment T1 was effective because C. annuum or S. lycopersicum rapidly depleted it, while in treatment T3 with A. niger, the phosphorus was solubilized in the soil and available to C. annuum or S. lycopersicum. The importance of applying soluble phosphorus or inoculating agricultural crops with A. niger, a phosphate-solubilizing microbial agents lies in the fact that phosphorus is vital for the energy metabolism of C. annuum or S. lycopersicum. These high-energy phosphates are part of the chemical structure of nucleotides, that are fundamental for the growth and productivity of C. annuum or S. lycopersicum
To the Research Department of the Universidad Autónoma Agraria Antonio Narro, Buenavista, Saltillo, Coahuila, México, for supporting this work (2026). To the Coordinación de Investigación Científica de la Universidad Michoacana de San Nicolás de Hidalgo: “Aislamiento y selección de microorganismos endófitos promotores de crecimiento vegetal para la agricultura y biorecuperación de suelos” from the Research Project (2026), Morelia, Michoacán, México.
The authors declare no conflicts of interest.

©2026 Gallegos-Morales, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.