eISSN: 2574-9838


Case Report Volume 8 Issue 1
Department of Orthopedics, Yamamoto and maeda memorial Maeda Hospital, Japan
Correspondence: Hiroyuki Maeda, Department of Orthopedics, Yamamoto and maeda memorial Maeda Hospital, Tokyo Spinal Cord Stimulation Center, Tokyo, Japan, Tel +81424732133
Received: March 02, 2023 | Published: March 13, 2023
Citation: Maeda H. A case of post-stroke pain and gait improved by spinal cord stimulation therapy. Int Phys Med Rehab J. 2023;8(1):67-68. DOI: 10.15406/ipmrj.2023.08.00336
A female patient had residual numbness and pain in the right half of the body after intracerebral hemorrhage (capsular hemorrhage). After a 1-week trial of spinal cord stimulation (SCS), the Numerical Rating Scale (NRS) improved from 10 to 5 and the Oswestry Disability Index (ODI) improved from 76 to 52. The patient was able to lift her legs, and her 210-cm walking time improved from 24.79 s to 15.25 s, which was highly satisfactory. One month after the trial, pain flared up and gait worsened; thus, SCS implantation was performed at the request of the patient and her family. Pain and numbness have subsequently decreased and range of motion has increased. One electrode was implanted in C4 and the other electrode was implanted in Th8. This case suggests that SCS therapy may be useful for central post-stroke pain (CPSP) that involves pain and numbness that affect half of the body.
Keywords: spinal cord stimulation therapy, central post-stroke pain, intellis
ODI, oswestry disability index; SCS, spinal cord stimulation; NRS, numerical rating scale; CPSP, central post-stroke pain
Central post-stroke pain (CPSP) is central neuropathic pain that develops after cerebral stroke. CPSP may be treated with spinal cord stimulation (SCS) therapy performed with one electrode implanted in the cervical spine and another in the thoracic spine, but there are few case reports of this approach. In this study, we report a significant improvement in pain, gait, and ADL impairment in a patient with CPSP who underwent this procedure after a SCS puncture trial.
A 57-year-old woman developed a hemorrhage of approximately 30×18 mm in the left capsular region (lenticulostriate artery), and pain and numbness in the right half of the body at age 53years (Figure 1). The modified Rankin Scale (mRS) was 4. The patient was able to walk, but due to pain, numbness and allodynia, she had a spastic gait and difficulty lifting her legs. She suffered frequent pain and numbness in the upper limbs, even at rest. She had no muscle weakness in the upper and lower limbs, and could maintain motor function in daily life; however, she had complaints of slow gait, numbness, pain and decreased sensation.
Since drug treatment resulted in no improvement for nearly four years after development of cerebral bleeding, the patient requested a trial of spinal stimulation therapy. MRI of the thoracolumbar spine showed no findings of spinal stenosis. An electrode was implanted for one week for postoperative high-frequency stimulation at 1,000 Hz/90μ/1.7 mA, but there was no apparent treatment effect. A percutaneous trial was performed for one week. The patient felt more discomfort with rapid pacing of spinal stimulation therapy than that from pain and numbness caused by the paresthesia she had felt before the therapy. Thus, the stimulation setting was changed to tonic stimulation at 5Hz/300μ/2.0 mA on day 4 after surgery, and this gave a therapeutic effect. Rehabilitation was also started during hospitalization. The NRS improved from 10 to 5 with an ODI of 75 to 52, and walking time between 210-cm parallel rods improved from 24.79 s to 15.25 s. Posture also improved, the patient could bend her knees and lift her feet, and her stride length increased.
Subsequently, pain and numbness flared up again, and the symptoms returned to those before the percutaneous trial. One month after the trial, a permanent implant was placed, according to the wishes of the patient and her family members. In the supine position, a 3-cm longitudinal incision was made in the midline of the L2 spinous process, and two epidural needles were punctured from the L2 part of the lamina. The electrode lead on the right side was implanted in Th8 and placed in the midline of the spinous process. The left electrode lead was placed in the midline of the spinous process of C4 while the cervical vertebrae were bent forward and opened when passing through the neck, so that palisades would occur when the right upper limb was stimulated slightly. A 4-cm longitudinal incision was made in the seated position, and a pocket was made just above the gluteus maximus muscle. An Intellis generator (Medtronic Japan) was then implanted while paying attention to the superior cluneal nerve. The operative time was 1hour and 30minutes (Figure 2).
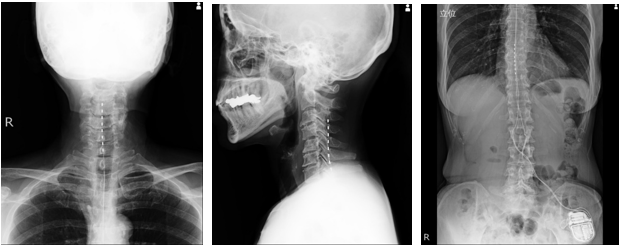
Figure 2 Radiographs after SCS implantation. Frontal and lateral views of the cervical spine (top panels). Frontal and lateral views of the thoracolumbar spine (bottom panels).
The stimulation settings were tonic stimulation at 5Hz/300 μ/0.5mA (cervical spine electrode) and 5 Hz/300 μ/2.4mA (thoracic spine electrode) at the level of sensory threshold after paresthesia mapping immediately after surgery. High-frequency stimulation of 1,000 Hz/90 μ/2.6-4.0mA (AdaptimStim™) was then used because no treatment effect was found on day 3 after surgery. After discharge, the patient was treated with higher stimulation intensity of 1,000 Hz/90 μ/2.6-4.2mA and 0.2mA at the outpatient clinic. The modified Ashworth scale improved from 2+ before permanent implant to 1+ after permanent implant. The same stimulation settings were maintained for three months after permanent implant, and improvement was observed in daily life, as seen by NRS 2 and smooth rising from a chair.
CPSP is defined as central neuropathic pain caused by a lesion in the brain parenchyma due to cerebrovascular disease, and is reported to occur in 1 to 8% of stroke patients.1 CPSP is caused by foci in the spinothalamic tract or the thalamic to cortical projection pathways, and has been attributed to abnormal nerve excitability and impairment of the descending inhibitory system, but the details are unknown. SCS may improve pain by increasing GABA release in the posterior spinal cord and ascending changes in neural activity in the pain matrix of the brain.2 CPSP is often accompanied by sensory disturbances such as sensory dullness, hypersensitivity, and allodynia, and with intractable pain and clinical problems that have no effective treatment. VAS for pain has been found to be improved by SCS performed in 166 patients with CPSP.3
Patients with CPSP may visit orthopedic surgeons or anesthesiologists if they cannot be managed by neurosurgery. Therefore, utilization of SCS therapy requires a wide range of knowledge from neurosurgery, orthopedics, and anesthesiology, as well as acquisition of the technique. It is important to perform the procedure in a short time because patients with pain and numbness often have difficulty staying in the same position while in the supine position. In spinal stimulation therapy, it is important to place a lead at an appropriate site by confirming and evaluating paresthesia using stimulation during surgery.
In patients with CPSP, the threshold of numbness is low, and electrical stimulation may be painful and uncomfortable. Initially, it is better to allow the patient to get used to the stimulation, and then gradually increase the intensity of stimulation at the outpatient clinic. It is difficult to control stimulation for CPSP in spinal stimulation therapy, and some patients may feel discomfort when pain and numbness associated with CPSP and paresthesia are increased by high electrical stimulation in postoperative spinal stimulation therapy. Since the comfortable stimulation level differs among patients and the pain threshold changes after surgery, there is often a need to adjust the stimulation level in the outpatient’s department.4
The case reported here suggests that SCS therapy may be a useful treatment option for CPSP that is intractable to other medical treatment, and that it may improve pain and gait.
All authors contributed to conception and design of the study, acquisition of data; analysis and interpretation of data; and drafting or reviewing the article, and all approve the final version to be published.
None.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.

©2023 Maeda. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.