International Journal of
eISSN: 2470-9980


Review Article Volume 1 Issue 3
1Immunotoxicology and Allergy Unit and Occupational Research Biorepository at the Center of Excellence on Aging, Italy
2Department of Medicine and Aging Sciences, Italy
Correspondence: Claudia Petrarca, O.U. Allergy and Immunotoxicology & Occupational Research Biorepository Center of Excellence on Aging (Ce.S.I.) Via Luigi Polacchi 11, 66013, Chieti, Italy
Received: November 05, 2015 | Published: December 30, 2015
Citation: Petrarca C, Carpiniello F, Di Gioacchino M (2015) Recombinant Probiotics for Allergen Immunotherapy. Int J Vaccines Vaccin 1(3): 00017. DOI: 10.15406/ijvv.2015.01.00017
Research efforts to improve the efficacy of Allergen Immunotherapy (AIT) involve the discovery of new adjuvant. Some strains of probiotic bacteria are able to induce up-regulation of co-stimulatory molecules in DCs, promote TH1 cytokine production and trigger Treg differentiation and activity. In mouse models, they have been shown to prevent or suppress the harmful TH2 response and to potentiate allergen- or allergoid-based immunotherapy of type I allergic diseases. These observations could explain the effectiveness of the prolonged administration of probiotics in the prevention of allergic disorders in infants. Consequently, the probiotics could be used as adjuvant for AIT. However, the simultaneous administration of probiotics and AIT might not come to specific responses because of degradation and not optimal presentation to APC. Moreover, the lack of specificity of the immunological response might be overcome by using probiotic bacteria expressing the sensitizing allergen. This would allow the stimulation of the intestinal immune system by both immunomodulating factors at the same site. Only a few studies noticed the development of AIT strategies based on recombinant bacteria expressing the allergen. However, most of them reported basic favorable immunomodulatory properties, rather than showing the development of immune tolerance or symptoms reduction. The allergen sequestration inside the bacterial cell may represent limits for these preventive and therapeutic approaches. Recently, our research group developed a novel AIT strategy based on a food-grade bacterium Streptococcus Thermophilus (ST) with TH1 skewing property, able to produce the allergen intracellularly and to release it at the gut-associated lymphoid tissue (GALT) level thanks to its intrinsic autolytic behavior with tolerance induction, and readdressing the pre-existing allergen-elicited TH2 response. Hence, the present knowledge on the subject will be critically considered in this review, underlying the most recent experimental achievements.
Keywords: recombinant probiotics, allergen immunotherapy, streptococcus, prevention, therapy
SIT, specific immunotherapy; ST, streptococcus thermophilus; GALT, gut-associated lymphoid tissue; Treg, T-regulatory; AIT, allergen immunotherapy; Ig, immunoglobulin; APC, antigen-expressing cells; DC, dendritic cells; MAMPs, molecular pattern associated with microorganisms; TLR, toll-like receptors
Currently, AIT represents the sole allergen-specific therapeutic approach dealing with IgE-mediated allergic disease able to control not only its symptoms, as other treatments do, but also to rebalance the immunological profile of allergic subjects. In fact, they develop detrimental immune responses towards allergens, exogenous proteins that should be otherwise harmless, entering the body from the external environment by feeding, breathing or by cutaneous absorption. The effectiveness of AIT is based on its immunological mechanisms which are complex and still not completely clarified. In this scenario, a central role seems to be played by the antigen-expressing cells (APCs), typically dendritic cells (DCs) and macrophages.1 They are induced by the AIT to mature into a phenotype able to guide differentiation of naïve T lymphocytes (TH0) into T helper type-1 (TH1) or T regulatory (Treg) cells.1 The APCs promote these latter effects by activating two alternative cytokine patterns, IL-2 and IL-12 in the first case and IL-10 in the second. In turn, TH1 cells produce IFN-γ which prompt the immunoglobulin (Ig) isotype switch towards the production of allergen-specific IgG4 (IgG2 in mice), that compete with IgE for the binding to the allergen. The cytokines produced by Treg (IL-10 and TGF-β ) exert an inhibitory activity inducing energy or apoptosis of TH2 cells. In this way, they suppress the production of their characteristic cytokine IL-4, which represents the main input for B lymphocytes to produce IgE. As a result, basophils and eosinophils cannot be activated to release mediators of the allergic inflammation which, as it is well known, fuel and amplify pre-existing immunological response and associated symptoms. In addition, the IgE-mediated antigen presentation by APCs and the stimulation of specific TH2 clones are inhibited. All these immunological variations counteract the allergic responses and contribute to restore the non-pathological ones towards exogenous proteins. The ultimate effects are desensitization and attenuation, or even complete disappearance, of allergic symptoms. However, the AIT, which involves the administration of an allergen in native form, could trigger anaphylaxis, in particular when high doses are necessary for an improved clinical outcome. Hence, AIT could be ameliorated in terms of safety and efficacy by developing hypoallergenic antigens,2‒5 or adjuvant molecules6‒9 able to stimulate Treg and TH1 cells function. A more recent approach involves the production of carrier substances promoting the optimal presentation of the therapeutic allergen to the immune system. An example of translational research into clinical practice is represented by the development of allergoids, chemically modified allergenic proteins with reduced binding affinity for IgE, presenting lower or no risk of side effects.10
Probiotics are generally commensal and/or symbiotic gram-positive bacteria, resistant to the gastric environment and able to settle transiently the small intestine. Acting together, they create the microbiota, a complex system contributing to the intestine functional activity and favoring its normal physiology also considering its intimate association with the immune system. For this reason, this system may represent a possible target for the treatment of allergic diseases, being able to develop regulatory immune responses through IL-10 induction11 providing as well adequate stimuli for the acquisition of antigen tolerance. Probiotic bacteria could be suited to this purpose. It has been demonstrated that certain strains of probiotics regulate the immunological homeostasis of the intestinal mucosa,12‒14 and partially affect the development of allergic diseases.15‒17 Furthermore, epidemiological studies revealed the correlation between the composition of the intestinal microflora and the prevalence of atopy.18
Lactobacilli (LAB) are the main probiotic bacteria employed for the preparation of dairy products and of probiotics supplements for human use; actually, they are not pathogenic, do not damage the mucous membranes, do not own genes for antibiotic resistance and are not degraded by bile acids.10,19 Therefore, the use of LAB as adjuvant factor in AIT represents an innovative approach for desensitization. For instance, some strains are able to suppress the TH2 harmful response which characterizes allergy.20‒22 The mechanism through which LAB trigger the suppression of the TH2 harmful response is not fully clarified so far. Several in vitro and in vivo studies demonstrated that most - but not all - lactobacilli strains own immune system adjuvant properties, such as the ability to activate DC and to induce the TH1 cytokines, inhibit the TH2 cytokines and reduce the IgE production.23‒28
In murine models of allergy, it has been proved that several LAB strains are able to induce Treg cells 29,30 and their inhibitory cytokines IL-1031,32 and TGF-β,32,33 enhancing the protection from an inflammatory response of the airways.34 Behind these immunological changes there is a complex interaction between probiotics and the host cells, i.e. intestinal epithelial cells (IEC)35 and intestinal dendritic cells (DC).36 A closer approach between probiotics and the DCs inside the lamina propria may take place in the intestinal lumen. This is possible thanks to the protrusion of dendritic extensions through the IECs layer or through the lymphoid tissue "dome region" associated with GALT, where probiotics are transferred through specialized epithelial cells named M cells. The contact may occur through the interaction of molecules exposed on the surface of the different cell types involved, such as the bacterial Molecular Pattern Associated with Microorganisms (MAMPs) and the Toll-like receptors (TLRs) expressed on IECs and DCs, as well as the DC-SIGN also expressed on DCs. The link between ligand and receptor induces DCs maturation so that to produce two possible alternative types of cytokines, IL-12 or IL-10 (and TGF-β), essential for polarization of naïve CD4+T cells (TH0) towards TH137 effecter cells or T regulatory cells35‒38 (Figure 1).
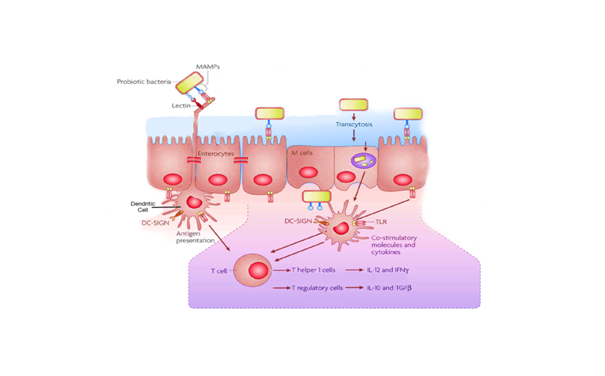
Figure 1 Cellular and molecular interactions between lactobacillus and intestinal epithelium and GALT.
These data stimulate the studying of new lactobacilli strains, in vitro and in vivo, that can improve the therapeutic efficacy of AIT. Lactobacilli present immunomodulatory activity on T effecter cells and T regulatory cells in humans. However, the clinical application of lactobacilli for allergic diseases is still controversial. On the one hand, some clinical trials demonstrated that the administration of lactobacilli prevent the occurrence of allergic diseases in childhood39‒42 and, more recently, the co administration of a probiotic and peanut-OIT produced sustained unresponsiveness in children with peanut allergy, but the relative contribution of the probiotic to this outcome is to be clarified.43 On the other hand, other studies did not show any positive effect nor benefits were observed just in the short term.41,44 In some clinical studies on adults affected by atopic dermatitis or allergic rhinitis, lactobacilli produced a modulation of the immune response45,46 and, just in one case, a reduction of nasal symptoms.46 On the contrary, they revealed to be ineffective for allergic asthma.47 Even in patients affected by allergic rhinitis and treated with lactobacilli, it was not observed any evidence of symptoms reduction despite a clear shift of TH1-TH2 and an induction of Treg.48 Probably, the absence of a clinical remission is related to the fact that probiotics evoke immunological responses that are independent from the allergen and, thus, not specific for it.49 This might limit the clinical efficacy of the treatment in the long term, which could be achieved with an immunological tolerance established by Treg cells specific for the antigen.50,51 For this reason, it has been argued that the immunomodulatory effect of probiotics could be enhanced and made more specific thanks to a joint administration with the allergen. However, a simple “probiotic-vaccine” association may result ineffective after the enzymatic degradation of the allergen administered orally. In this case, the association of a probiotic with an allergoid, rather than with an allergen, may result more appropriate. In support of this theory, in a previous study, our research group demonstrated that the AIT effectiveness based on the allergoid Amb a 1 of Ambrosia is enhanced by the co-administration of Lactobacillus paracasei LP6.52 Recombinant probiotics may represent the proper instrument for the development of a safer and more effective anti-allergic vaccine, administrable orally, is given by recombinant probiotics producing the allergen.
Probiotic bacteria are microorganisms that can be modified in order to produce inside, or in a secreted form, exogenous proteins. These latter generate recombinant probiotics and, when expressing an allergenic protein, could be used as innovative vaccines with enhanced or upgradeable characteristics for the application in AIT protocols. Thanks to recombinant DNA technology, it is now feasible to produce large quantities of pure proteins either in native or mutated form, using the bacteria, including those probiotic, as natural "incubators" for their production. For example, by using this technology any DNA sequence could be inserted inside a bacterial expression vector introduced into the bacterium; this latter transcribe and translate it into the polypeptide chain inside its cell or secrete it in the culture medium. Furthermore, through the site-specific mutagenesis, it is possible to modify particular stretches of gene sequence in order to obtain proteins with altered activity. For the allergens, can be obtained hypoallergenic molecules able to maintain their immunogenicity.
A recombinant probiotic expressing the allergen could be employed as antiallergic vaccine, thanks to its features which are innovative not only in comparison with conventional treatments but also compared to the use of the sole probiotic or probiotic-allergen association. In the first instance, recombinant probiotics can be easily administered orally. Secondly, the probiotic is likely to protect the intracellular allergen from proteolytic degradation and to release it in situ. Moreover, by doing so, it is possible to convey the allergen in the immunological districts (mesenteric lymph nodes and Peyer's patches) where it could promote the immunological tolerance in a specific manner.
A very interesting aspect of this approach is the chance to obtain high local concentrations of allergen thanks to its accumulation inside the probiotic cell; therefore, this may results in a more effective vaccine employing lower cumulative doses of allergen with a decreased risk of anaphylactic secondary effects.
So far, just a limited number of studies reported data on the development of AIT strategies based on the use of recombinant bacteria expressing the allergen53‒60 and no one of them has found application on humans. Pre-clinical studies in mouse models of allergy demonstrated that recombinant probiotics are able to counteract the allergic sensitization process with appropriate immunological variations, both in newborn and adult mice, when the treatment is administered as prophylaxis.61‒66 Peanut allergen-producing L. lactis strains modulated allergic immune responses redirecting TH2-polarized to non-allergic TH1 immune responses with involvement of sIgA and regulatory T cells.67 However, for what concerns adult mice, no data are provided regarding the induction of Treg and the local inflammatory response after airways challenge.
By contrast, the two studies on newborns detected an increase of Foxp3, the transcription factor that identifies Treg cells, and a reduction of bronchial reactivity within the offspring of mothers treated during pregnancy and lactation. In these two cases, the instauration of tolerance implies the education of the immune system during its maturation, with effects on innate immunity and on natural Treg cells; moreover, despite not being driven by the antigen, they could have a non-specific effect also on responses to a specific allergen. A very different process from the previous one must be established in adult and pre-sensitized mice, which requires the intervention of adaptive immunity and inducible Treg cells with antigen specificity. Finally, in adult sensitized mice, the use of recombinant probiotics administered according to a therapeutic setting, in two out of three studies, is associated with decreasing symptoms of allergic inflammation, even if it is not fully clarified the involvement of Treg cells in the observed effect (Table 1).
|
Protocol |
Probiotic |
Recombinant Allergen |
Site of Vaccination |
Model of Allergic Sensitization |
Immunological Effects |
References |
|
(and Symptomatological) |
||||||
|
Prophylaxis in Adult Mice |
Lactococcus lactis |
BLG |
Intragastric |
BALB/c |
↓ IgE specific |
|
|
Mice |
↑ IgG2a specific |
|||||
|
↑ IFN-γ |
||||||
|
Effects correlate with the levels of BLG expressed by L. lactis |
||||||
|
Lactobacillus plantarum; Lactococcus lactis |
Bet v 1 |
Intranasal |
BALB/c |
↓ IgE specific |
||
|
Mice |
↑ IgG2a specific |
|||||
|
↓IgG1/IgG2a |
||||||
|
↓ Eo in BALF e |
||||||
|
↓ IL-5 in BALF |
||||||
|
↑ sIgA specifiche in polmone e intestino |
||||||
|
Lactococcus lactis |
OVA |
Intragastric |
BALB/c mice (TCR specific for OVA) |
↓ local and systemic response; |
||
|
↑ lymphocytes |
||||||
|
T CD4(+)CD25(-); |
||||||
|
↑ IL-10 ↓ IFN-γ in splenocytes reactivated in vitro; |
||||||
|
↑ Foxp3 e CTLA-4 in Treg |
||||||
|
Lactobacillus plantarum |
Der p 1 |
Intranasal |
BALB/c |
↑ IgG2a specific |
||
|
mice |
↓ Eo in BALF e |
|||||
|
↓ IL-5 in BALF |
||||||
|
Prenatal and Neonatal Prophylaxis |
Lactobacillus plantarumNCIMB8826 |
Bet v 1 |
Settlement of the mothers |
BALB/c mice (mothers in pregnancy) |
Newborn |
|
|
Pre- sensitization: Th1 profile (IFN-g from splenocytes stimulated in vitro with Ag). |
||||||
|
Post sensitization: |
||||||
|
↓ IL-4 e IL-5 in SP cells and MLN reactivated in vitro with Bet v 1; |
||||||
|
↓ IgE, IgG1, IgG2a specific in serum; |
||||||
|
↑ Foxp3 mRNA in splenocytes |
||||||
|
Lactobacillus paracasei NCC 2461 |
Bet v 1 |
Oral |
BALB/c mice |
In newborns: |
||
|
(pregnant or lactating mothers) |
↓ Eo in lungs; |
|||||
|
↓ IL-5 in BAL, lungs e mediastinal lymph nodes in vitro; |
||||||
|
: IgE, IgG; |
||||||
|
↓ IL-4 e IL-5 in splenocytes in vitro reactivated in vitro with Bet v 1 or ConA; |
||||||
|
↑ Foxp3 mRNA lung; |
||||||
|
↑ TGF-β in serum; |
||||||
|
↓ peribronchial inflammation and mucus |
||||||
|
Therapy in Adult Mice |
Lactobacillus plantarum |
Der p 1 (peptide) |
Mucosal |
Mice C57BL/6 J |
↓ IFN-γ (not specific) |
|
|
↓ IL-5 (specific) |
||||||
|
Lactobacillus acidophilus |
Der p 5 |
Oral |
BALB/c |
↓ IgE specific |
||
|
Mice |
↑ IgG specific ?? |
|||||
|
↓ Eo in BALF e |
||||||
|
↓ Hyperactive response of the airways (AHR) |
||||||
|
Lactococcus lactis |
Bovine Beta-lactoglobulin (BLG) |
Intranasal |
Mice |
↓ IgG1 in BAL; |
||
|
↓ IL-4 ↑ IFN-γ in splenocytes reactivated in vitro; |
||||||
|
↓ local response to nasal challenge |
||||||
|
Lactococcus lactis |
Ara h 2 |
Oral |
Mice |
↓ TH2, ↑ TH1 |
||
|
↑SIgA, Treg (local response) |
||||||
Table 1 Overview of studies focusing on recombinant probiotics in experimental allergy
Table abbreviations: BAL: Bronchoalveolar Lavage; Der p 5: Dermatophagoides Pteronyssinus Group-5 Allergen; BLG: Bovine Beta-Lactoglobulin; OVA: Ovalbumin; Bet v 1: Major Allergen of Birch Pollen; ConA: Concanavalin A; TCR: T-cell Receptor; Cry j 1: Major Allergen of Japanese Cedar Pollen; Der p 1: Immunodominant T Epitope of D. Pteronyssinus
The preclinical studies described so far confirm the adjuvant effect of recombinant probiotics in the prevention and treatment of allergies, but do not clarify whether they are vaccinal tools able to stimulate long-lasting immunological tolerance mediated by Treg towards the allergen. For this reason, our group has recently conducted a research in vivo using a probiotic strain with peculiar characteristics, evaluating the Treg and pulmonary response.51
In this study, it was generated a strain of Streptococcus Thermophilus (ST) expressing Bet v 1, the major allergen of Betula verrucosa, in order to verify its possible adjuvant or therapeutic effect in BALB/c mice made IgE-responsive to this same allergen.51 The choice of this strain stems from different considerations. First, it has been taken into account the large presence of peptidoglycan on the cellular wall and of (lipo)teichoic acids, owning immunomodulatory activities. Secondly, it has been considered its autolytic character provided by the presence of lysogenic bacteriophage expressing an enzyme able to degrade the bacterial cell wall; finally, it represents a prokaryotic expression system suitable for integrating and expressing permanently foreign genes.
In particular, the impossibility of such recombinant probiotic strain to colonize stably the intestine due to its autolytic character is a rather captivating feature for human application because the risk of transferring the antibiotic resistance is null.
The immunomodulation level and therapeutic efficacy of the ST recombinant probiotic was assessed and compared to the effects of only ST and ST+rBet v 1 association by measuring various immunological and histopathological parameters (cytokines in serum and produced by immune cells in vitro, Treg cells, inflammatory cells and cytokines in the lung tissue). This study showed that the therapeutic treatment of pre-sensitized mice with the recombinant probiotic expressing the allergen produces, upon recall airway challenge, specific local and systemic anti-inflammatory and inhibitory response along with clearance of lung eosinophilia,51 which characterizes the inflammatory response of allergic asthma in mice.64 Our findings suggest that the recombinant probiotic releases the allergen in an immunologically active form into the intestine and stimulates the resident immune cells determining the observed allergen-specific and favorable immunomodulant effects.
In the present review, we have shown the most recent experimental findings supporting the use of recombinant probiotics in the prevention of allergic disorders, suggesting as well their potential usability for the allergen immunotherapy (AIT). We showed that the probiotic lactobacillus Streptococcus Thermophilus expressing the major allergen of Betula verrucosa administered orally is able to restrict the TH2 allergic inflammatory reaction in sensitized mice, with a shift towards TH1 and Treg allergen-specific immune responses.51 Our study confirmed the immunomodulating properties of probiotics and showed that allergen-expressing probiotics can represent vaccines to induce tolerance within an experimental allergen immunotherapy setting (Figure 2). The contribution of our approach, which highlights how recombinant probiotics may act, at once, as adjuvant and antigenic stimuli, contributes to figure them not only as valid players in preventive protocols, as most studies over the past years demonstrated, but also as tools for specific immunotherapy applications.
None.
Author declares there are no conflicts of interest.
None.

©2015 Petrarca, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.