International Journal of
eISSN: 2573-2838


Short Communication Volume 4 Issue 5
1Department of Analytical Chemistry, University of Havana, Cuba
2AIDS Research Laboratory (LISIDA), Cuba
3Department of Analytical Chemistry, S
Correspondence: Abel I Balbin Tamayo, Department of Analytical Chemistry, Faculty of Chemistry, University of Havana, La Habana, Cuba
Received: August 07, 2018 | Published: September 18, 2018
Citation: Tamayo AIB, Rizo LSL, Armas MB, et al. Free-label electrochemical detection of of HIV-1 proviral DNA in clinical sample.Int. J Biosen Bioelectron. 2018;4(5):217. DOI: 10.15406/ijbsbe.2018.04.00129
The detection of specific base sequences in human, viral, or bacterial DNA holds great importance in diagnosis of several diseases. Detection of infectious and inherited diseases at molecular levels provides reliable and early diagnosis.1 A epoxy conducting composite material prepared from epoxy graphite modified with graphene oxide was developed. These composite electrodes were also successfully use for the voltammetry detection of oxidation signal of guanine of oligonucleotide encoding a region of HIV-1 proviral DNA in clinical samples. The results obtained in this study showed that the composite electrode used to detected biomarkers of viral diseases such as HIV-1.
Keywords: graphite-epoxy, electrode, voltammetry, DNA, HIV-1
DNA biosensors based on carbon electrodes are under intense investigation by many research groups. Carbon electrodes are particularly attractive for sensing applications because of their low cost, wide working potential window, good electrical conductivity and relatively low background currents.1 A great deal of attention has given in the last years to the use of graphite in composite materials, taking advantage of their exceptional mechanical and electrical properties for the development of electrochemical platforms.2,3 In particular, graphite composites have high intrinsic resistance and very low relaxation time, within the time scale of the electrochemistry processes;4-6 thus, parameters and details about the percolation phenomena through the solid can be neglected.7 The graphite composite electrodes can also easily modify, allowing the incorporation of different components such as ligands, enzymes, cofactors, mediators, catalysts. Therefore, there is a special interest in using these electrodes in the development of feasible sensors for the detection of biologically-relevant molecules, such as DNA of viruses. The detection of viral diseases is usually performed by specific techniques such as immunoassay (ELISA), Polymerase Chain Reaction (PCR), among others.8 However, these techniques have several disadvantages, such as the need for expensive equipment and reagents involving laborious procedures that also requiring experienced personnel. The aim of this work was the detection of DNA in clinical sample with epoxy graphite electrodes modified as starting materials for the development of biosensors for DNA viral diseases. The composite electrode preparation included the use of epoxy graphite modified with graphene oxide, to introduce the carboxylic groups required for potential covalent binding of biologically relevant species. The final electrodes were test using a HIV-1 proviral DNA in clinical samples.
The voltammetric method was developing using a Palm Sens (Palm Instruments BV) coupled to a computer managed by Palm Sens PC software and operating in the voltammetric analysis square-wave mode. The modified graphite-epoxy with graphene oxide (EGIIog) working electrode was constructing following the work of Balbin-Tamayo et al. The Ag/AgCl(KClsat) electrode was used as the reference and a platinum wire was used as the auxiliary electrode.
Reagents
Nucleate free water SIGMA Life Science, Streptavidin, essentially salt free, lyophilized powder, ≥13 units / mg protein, EDC (1-ethyl-3- (3-dimethylaminopropyl) carbodiimide) (neat, ≥98.0%), and NHS (N-hydroxysuccinimide) (> 98.9%) were also purchased from Sigma Aldrich (USA.) Oligonucleotide for the biotinylated H1Gag1584 gag gene (sequence 5'-AAA GAT TAA TCC TGG G-3', sequence for the gag gene, identical to those of the ELI reference strain of the Los Alamos gene bank. strain ELI, Genbank accession number K03454), and complementary oligonucleotide (DNAc, sequence 5´-CCCAGGATTATCCATCTTT-3´), synthesized by (Exxtend Biotechnology Ltda) (oligonucleotide-Biotin, Brazil).
Experimental procedures
The oligonucleotide was immobilized on the surface of the modified epoxy-graphite electrode, the streptavidin was covalently linked to the surface carboxylic groups by peptide bonding.3,10 The biotinylated oligonucleotide was bound to the streptavidin bonded onto the surface of the graphene oxide-modified epoxy-graphite electrode (EGIIog) by the key-lock mechanism.11 Then the electrochemical detection of the oxidation signal of guanine of the oligonucleotide was performing. The oligonucleotide employed encodes a region of the HIV-1 H1Gag1584 gag gene, whose sequence is AAA GAT TAA TCC TGG G. This is one of the most conserved regions, which varies little between groups of HIV-1. Preparation of Clinical Samples: DNA from peripheral blood mononuclear cells was extracted from 100 μL of whole blood by column affinity chromatography with QIAmp DNA mini Kit, Qiagen, and QIAcube automated extraction kit. These samples were characterized in the Molecular Biology Laboratory of LISIDA, through their viral load through their direct detection in peripheral blood by the standard system for the in vitro diagnosis COBAS® Ampliprep / COBAS® Taqman® HIV-1 test v2.0,12 whose limit of detection is 20 copies mL-1. The samples correspond to people who were followed up clinically and serologically, as established by the algorithm for the diagnosis of HIV-1 infection in Cuba.13 Two samples with a viral load of less than 20 mL-1 copies were used as Low Positive Control (<20 mL mL-1 copies) and two samples. High viral load (≥104 copies mL-1 (mL-1 RNA) ), as High Positive Control and a sample as Negative Control (0 copies mL-1 (RNA mL-1)).
Streptavidin was covalently linked to surface carboxyl groups of the EGIIog electrode by peptide bonding. The biotinylated oligonucleotide (Biotin-H1Gag1584) was bound to streptavidin bound on the surface of the graphene oxide-modified epoxy-graphite electrode (EGIIog) by the key-lock mechanism. Then the electrochemical detection of the oxidation signal of the electrode was carried out at each step of modification and hybridization. Figure 1 shows the voltamperograms per square wave of the EGIIog electrode, EGIIog activated with NHS / EDC, EGIIog with covalently bound streptavidin, EGIIog with the biotinylated streptavidin / H1Gag1584 complex and EGIIog with the streptavidin / H1Gag1584 complex biotinylated with the complementary oligonucleotide (cDNA) in the supporting electrolyte (Buffer phosphate at pH 6.9, 20 mmolL-1). For the EGIIog electrode with the biotinylated streptavidin / H1Gag1584 complex, a potential signal of 0.94V is shown, close to the oxidation potential of guanine.14,15 Detection of proviral DNA can reduce the post-exposure window period to about 11 days and so may also be used to detect HIV-1 in individuals who have not yet mounted an immune response, or to resolve indeterminate serology results.16 The carboxyl groups of the graphene oxide-modified epoxy-graphite electrodes allowed immobilization of the biotin-H1Gag1584 oligonucleotide on the electrode by the key-lock mechanism between the biotinylated oligonucleotide and streptavidin. This immobilization avoids non-specific adsorption, allows a good spatial configuration of the oligonucleotide and improves electrode selectivity.17 This is an important element in the reliable and sensitive detection of HIV-1 proviral DNA (analyte) from hybridization. In the voltamperogram (Figure 2), a signal at 0.98 V can be observed that can be associated with the signal of oxidation of guanine of the H1Gag1584-biotinylated chain. The current intensity of the 0.98 V signal experiences a decrease when the biosensor (electrode with the oligonucleotide) contacts the positive controls under hybridization conditions.18 This behavior is related to the formation of hydrogen bonds, between complementary bases, of the biotinylated-oligonucleotide and proviral DNA, which causes a decrease in electron density by the formation of these interactions.19,20,
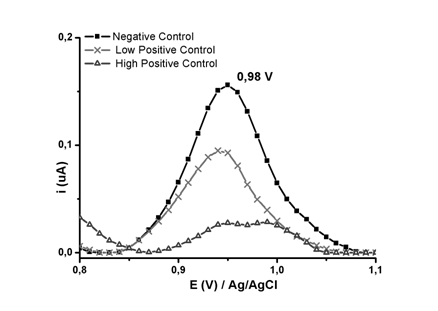
Figure 2 Square-wave voltamperogram of negative control, low positive control and high positive control, in PB at pH 6.9, 20 mmolL-1.
An initial comparison of the means of peak oxidation currents to 10 measurements, using the whisker box (Figure 3) (Table 1), shows that there are appreciable differences between the means of the negative control peak currents and the positive controls (high and low) at 0.98 V. Although a quantitative correlation between the viral load and the amount of proviral DNA is not established, it is known that more than one virion can infect the same cell, and more than one region of proviral DNA may be inserted into the chromosomal DNA. At 0.98 V, a marked decrease in peak current is observed for high positive controls (105 copies of mL-1 RNA), because there is greater hybridization than for the low positive control (less than 20 RNA mL-1 molecules). The amount of proviral DNA detectable in samples with high viral load may be conditioned by a larger number of infected cells, which may have more than one proviral DNA region integrated, which would be in agreement with the marked decrease of the signal for the high positive control. While samples with low viral load (low positive control), it is only due to proviral DNA regions in the few infected cells. Decreasing the intensity of the guanine oxidation stream from the biotin-H1Gag1484 oligonucleotide to the potential of 0.98V allowed the detection of proviral HIV-1 DNA by square-wave voltammetry in phosphate buffer pH 6.9.
|
Desviación Estándar |
Ym±r |
|
Negative control |
0,197±0.04 |
|
Low positive control |
0,119±0.02 |
|
High positive control |
0,036±0.01 |
Table 1 Standard deviation (Ym±r) for peak current at 0.98V
Square-wave voltammetry allowed the detection of HIV-1 proviral DNA, from chromosomal DNA of whole blood lymphocytes, in clinical samples by decreasing the intensity of the guanine oxidation current of the biotin-H1Gag1484 oligonucleotide to the potential of 0.98V. The results obtained in this work show the suitability of graphite-epoxy composite electrodes modified with graphene oxide for the development of electrochemical DNA sensors. These composite electrodes offer the possibility of obtaining electrochemical response at very low concentrations of single DNA in clinical sample, and the required binding groups at the electrode surface for covalent immobilization of specific oligonucleotides.
We thank the biosensor group of the Institute of Chemistry of Araraquara for their help in the development of the work presented here. CNPQ-TWAS (CNPq nº 009/2014) for fellowships and financial support.
The authors have no conflicts of interests in this work.

©2018 Tamayo, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.