International Journal of
eISSN: 2573-2838


Mini Review Volume 3 Issue 4
1Department of Chemical Engineering, Thapar University, India
2Department of Chemical Engineering & Technology, Nano and Micro System Design and Fabrication Lab, India
3School of Biomedical Engineering, Banaras Hindu University, India
Correspondence: Manoj Kumar, School of Biomedical Engineering, Indian Institute of Technology (Banaras Hindu University), Varanasi-221005, India, Tel +918283839972
Received: July 24, 2017 | Published: November 1, 2017
Citation: Duhan S, Ranjan S, Kumar M. Biosensor based cell-phone integrated point of care diagnostic devices: challenges. Int J Biosen Bioelectron. 2017;3(4):308–311. DOI: 10.15406/ijbsbe.2017.03.00071
Current article discuss in detail, problems in integrating the biosensing platform to the Cell-Phone based system to develop Biosensor Integrated Cell phone based Point of Care Diagnostics (BICPOCD). The aim of this article is elucidate the formidable challenges in the development of biosensing platform as well as in the integration of cell phones to make it BICPOCD. Current discussion briefly covers challenges in the area of optical biosensing platform utilizing Fluorescence Resonance Energy Transfer (FRET) as transduction mechanism as well as features available in cell phone camera.
Detection of infectious diseases is of prime concern globally. To meet this challenge continuing efforts have been invested to design easy, rapid, sensitive and selective biosensing platforms.1–14 However, efforts mostly remain confined to the diagnostic labs, because such diagnostics require use of expensive devices. Due to which diagnosis of infectious disease becomes costly and unaffordable to majority of population. Therefore, serious efforts are being put to device easy, rapid and cost effective methods affordable to all sectors of population. On the other hand, advances in cell phone technology especially in the area of camera and processor has provided good opportunity for integration with biosensing platform where simple, use and throw, inexpensive, rapid, sensitive, selective and reproducible biosensors could be designed and integrated to the cell phones. Integration of such point of care diagnostics to cell phones will allow accessibility of such systems to majority of population. For the successful design of Biosensor Integrated Point of Care Diagnostic Device (BIPOCD), following features will be desired: (i) Compatibility to maximum cell-phone platforms, (ii) minimum intervention from user, (iii) sensitive and selective enough to provide reliable and reproducible results, and (iv) affordable cost.15 To meet these requirements, seamless integration of biosensors and cell phone is highly desired which is a very complex problem and requires detailed analysis. BICPOCD constitutes of two parts- (i) Biosensing platform and (ii) Cell phone platform (Figure 1). This article will in detail cover challenges in the development of biosensor platform and to some extent cell phone platform.
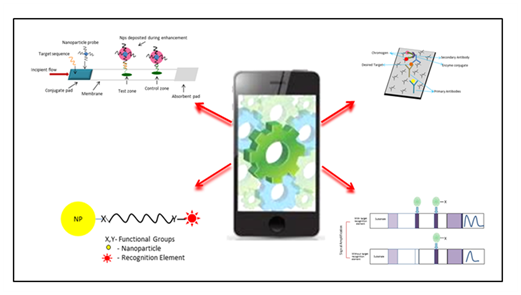
Current status & challenges
Biosensing platform houses the signal-generating unit, which consists of recognition element. Recognition element increases the concentration of desired target around the sensing element. This is achieved by binding of recognition element to the target selectively.9,16–20 Stability of the complex (recognition element-target) is indicated by the by binding constant (KD) which varies from complex to complex. For example, KD for nucleic acid duplexes.12–21 varies from μM- nM, whereas KD for antibody-antigen complex ranges from mM- nM. Further, KD value also indicates the limit of detection that can be achieved theoretically.22–24 Therefore use of different recognition elements will result in different sensitivity and hence it is important to choose the recognition element with great care. Antibodies, peptides, or aptamers are predominantly used for the detection of epitopes on the cell-surface. Whereas detection of nucleic acids such micro RNA, DNA, or RNA, is achieved by using nucleic acids as recognition elements. Therefore target to be detected, dictates the choice of recognition element and hence sensitivity and selectivity. Selectivity of the systems can be further improvised through temperature control as well as by tuning wash buffer composition. Additionally, three factors, cost, facile incorporation of recognition element and shelf-life will also dictate the success of biosensing platform. For instance nucleic acids are easy to covalently link to the biosensing platform, has low KD value and easy to store for long time as compared polyclonal antibodies.12,25–29 Polyclonal antibodies are difficult to attach to the biosensing platform, costly and have high KD value. Increase in the cost of antibodies can be attributed to their production cost. In some cases, direct linking of antibody to the surface can be avoided by utilizing sandwich format of detection. However, antibodies conjugates are quite expensive to use. On the other hand, use of polyclonal antibodies will allow one to achieve better limit of detection and selectively but only at the cost of increased expense. Therefore designing a universal biosensing platform that could house any kind of recognition element, with longer-shelf life, and easy to use, is of great challenge.
Recognition element introduces selectivity to the biosensing scheme. However for signal transduction an indicating element is required. This element should correlate between the physico-chemical change in the recognition element and presences of the target. This physico-chemical change in the recognition element is transduced into a readable output. This output signal could be mechanical, optical, electrochemical, magnetic or thermometric in nature. The technology pertaining to optical biosensing is highly advanced and allows development of highly sensitive, selective, robust, precise systems with very high signal to noise ratio and thus of prime concern here. Optical signal transduction may involve colorimetric absorption, fluorescence, or Fluorescence Resonance Energy Transfer (FRET) based response. To achieve better selectivity FRET based systems are highly reliable. FRET occurs due to dipole-dipole interaction between the donor and acceptor dye where donor need not to be a fluorescent. Since process is based on dipole-dipole interaction the distance of separation between donor and acceptor cannot exceed 10 nm. Donor is a fluorescent material in the excited state, which transfers its energy to the acceptor in the ground state. The condition of resonance between donor and acceptor could only be established if the excitation spectra of donor overlaps with the absorption spectra of acceptor and both are aligned and separated to each other within 10 nm range. Details of FRET are covered elsewhere and beyond the scope of current discussion. The ratiometric signal analysis in FRET allows high sensitivity and selectivity; therefore detection schemes based on FRET are of major importance.30–34 Detection using nucleic acids as recognition elements exploits three types of detection schemes: (i) sandwich, (ii) molecular beacon and (iii) ON/OFF type. In all these schemes luminescent dyes acts as donor and another dye as acceptor (fluorescent/quencher). Often in the presences of target, probes (multiple small complementary nucleic acid segments labeled with the reference dye) anneal resulting in decrease in separation distance between donor and acceptor, ensuing FRET. This results in generation of signal (ON-type) in presences of target or disappearance of fluorescence (OFF-type). Sometimes single probe with quenched emission (here acceptor is quencher), emits light in the presences of target. The above discussed schemes, when incorporated to the sensing platform serves as ON/OFF switch for generation of signal and thus completes the biosensing platform.
As discussed in above paragraph, photons are released in the process of signal generation, which can be detected. Cell phones detectors can be integrated to the biosensing platform to detect the emitted burst of photons. The number of photons emitted from dyes may be good enough for the detection of signal, however most of them are scattered and fails to reach the small window of the cell phone detector. Increasing the number of photons produced during the process can alleviate this problem. To increase the number of emitted photons following strategies can be adopted: (i) Covalently attaching recognition elements in high density, (ii) using target amplification process, (iii) utilizing dye systems with large molar extinction coefficient and quantum yield, and (iv) reducing background noise. In the first case, mechanical and electrical steric hindrances, surface characteristics, conjugation reaction efficiency under optimized conditions, will decide the probe density that will be attached to the biosensing platform. In the second case, targets can be amplified using polymerase chain reaction (PCR) or by rolling circle amplification. The target amplification is only limited to nucleic acid based targets. Another option will be to use stabilizers such as Hydroxy Napthol Blue (HNB) that are used with intercalating dyes. It stabilizes the dye-duplex interaction and enhances signal from the dye by 30%. Further signal enhancement can be achieved through clever designing of the BICPOCD. In general, one photon-one recognition element interaction leads to generation of single photon only. Through innovative design of the BICPOCD, single photon can be allowed to interact with one transducing element multiple times hence multiple photons output from single interaction. While designing BICPOCD careful consideration towards the optical property of the sample as well as transducing dye should also be taken into consideration. Light with shorter wavelength tend to pass through thin materials (low optical density), whereas, longer wavelength light will pass through thicker material (high optical density) with ease. Thus in the first case high power excitation source may be required to excite the transducing dye which may result into introduction of non-linear signal generation, photo bleaching and photo-oxidation of sample. On the other hand, ability to generate significant photons, intrinsic properties of dyes such as molar extinction coefficient and quantum yield also plays major role. The molar extinction coefficient is a measure of photon absorption efficiency, whereas the quantum yield is measure of efficiency for the entire transition process (from absorption to emission). Therefore, dye having same absorption wavelength but higher molar extinction coefficient will absorb more number of photons than one with small value under same excitation conditions. On the contrary dye with higher quantum yield will produce more photons than one with low quantum yield, for same molar extinction coefficient and excitation conditions. Therefore to design highly sensitive BICPOCD, it is desired to have donor and acceptor dye with large molar extinction coefficient (>105 M-1Cm-1), and high quantum yield (0.99) with maximum overlap integral. Additionally, donor with near infrared excitability will be highly desirable. However till this date NIR dyes (Cyanine dye, tricarbocyanine dye, IR 1048-dye, IR 1061 dye) with high molar extinction coefficient and quantum yield are limited. Thus research in the area of developing dyes with high absorption cross-section and high quantum yield needs to be addressed. Further signal to noise ratio can be improved by reducing the noise. This can be achieved optically as well as through mathematical programs. Implementing transducing dyes with non-overlapping narrow emission and absorption spectra, noise can be reduced to considerable level. Further signal to noise ratio can be improved by introducing mathematical algorithm.35–38 However detection of photons in the BICPOCDS still remains the challenge. Thus development of materials, triggering multiple electrons on interaction with single photon still remains a formidable challenge.
On the other hand cell-phone electronics such as cameras, LED, processor, and software would also play crucial role in designing efficient and reliable BICPOCDs. The LED in the cell-phones can be used as external excitation source whereas the cameras can be utilized as detector for image acquisition39–43 or color sensing.. The CMOS (complementary metal-oxide semiconductors) technology of the cell-phone has allowed pictures acquisition in photon starved situations with the lesser power consumption and heating in comparison to the conventional CCDs (charge coupled device).44–46 Further the cell-phone cameras with good electronic resolution are available. However, their optical resolution is poor due to small numerical aperture. . Additionally, pictures acquired, are automatically post-processed by the camera software (Volks iphone RetCheck software, Sidexis 4, Basler’s Pylon software, Allied vision’s Vimba software). The small numerical aperture of the lenses of camera, least control over camera parameters and image post processing without user intervention introduces skewness in overall data. A BICPOCD can only be accepted to be reliable when the sample data obtained from BICPOCD can be compared with the lab results with accuracy and repeatability. However in the current scenario if a BICPOCD is integrated with the cell-phone camera, data for the same sample will vary from camera to camera and hence no data reliability. To overcome these problems, camera with better numerical aperture, greater control over, brightness, color balance, hue and saturation control is required. Due to the above-mentioned technological challenges, BICPOCD advancement faces major impediment and hence not many BICPOCD has been introduced into the market. Based on the challenges, research in the area of materials for making transparent biosensing platforms, sensitive photodiodes, luminescent labels, and superior electronics will be highly desired. Further, research in the area of new detection scheme ready to integrate with cell phones, simple in design, easy to use, not requiring target amplification will be highly forthcoming. Additionally, software evolution to provide noise filtering through mathematical algorithms will also support the easy development of BICPOCD. Therefore in the coming years “The International Journal of Biosensors and Bioelectronics” is going to witness large volume of publications in these areas.
None.
The author declares no conflict interest.

©2017 Duhan, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.