eISSN: 2373-6372


Review Article Volume 7 Issue 5
1Department of Gastroenterology, Hospital Regional Docente de Enfermedades Neopl
2Faculty of Health Science University Continental Huancayo, Peru
3San Fernando Faculty of Medicine, Major National University of San Marcos, Peru
4Department of Radiology, Hospital Regional Docente de Enfermedades Neopl
Correspondence: Luis Guerra Montero, Department of Gastroenterology, Hospital Regional Docente de Enfermedades Neoplásicas Huancayo, Jr. José Santos Chocano N: 369. Apartment 401. Los Olivos, Lima, Perú
Received: August 09, 2017 | Published: September 8, 2017
Citation: Montero LG, Canchari PG, Peña AL, Guerra EG (2017) Review Article: A Giant Hepatic Hemangiomas. Gastroenterol Hepatol Open Access 7(5): 00251. DOI: 10.15406/ghoa.2017.07.00251
Hepatic hemangiomas are the most common benign liver tumors, often they appear as a single tumor, their size varies from few millimeters until over 20cm, and the ones who measure over 10cm are called giant. Those appear rarely and are more susceptible to present symptoms and complications like circulatory shock, hemoperitoneum with spontaneous rupture, lower extremity edema, gastric obstruction, biliary ducts obstruction, heart failure, hypothyroidism and the Kasabash Merritt syndrome. The general diagnosis is with image study, and rarely use a hepatic biopsy. There are many alternatives for the treatment like surgery, arterial embolization, radiofrequency by ablation, radiotherapy and medic treatment with propranolol.
Keywords: giant hepatic hemangiomas, kasabach-merritt syndrome
The Hepatic hemangiomas (HH) are the most common benign liver tumors, often they appear as a single tumor and are common located in the right lobe, but can also appear in multiples locations, and in 40% of the cases appear in both lobes.1 Their size varies from few millimeters until over 20cm, in some studies consider giant HH, those that measure more than 4cm, but most of the authors use this definition in those that measure more than 10cm, because previous studies showed that patients with this condition (HH that measure more than 10cm) are more susceptible to present symptoms and complications.2
Epidemiology
It is the second most common tumor in the liver after metastasis and represents 73% of all benign liver tumors, although its frequency in autopsy studies is less than 0.4%,2,3 some studies indicate that its prevalence and incidence can reach 20% and 7% of the general population, respectively.3,4
Generally, these tumors affect adult women in their third and fifth decade of life, mainly multiparous women. However, it is important to mention that it can occur at any age of life.3,5 The female to male ratio ranging from 3: 1 to 5: 1 according to preview studies.2–5
Etiopathogenesis
The etiopathogenesis of HH is not yet fully understood, however, it has been postulated that they are vascular malformations or hamartomas of congenital origin that are produced secondary to an intense ectasia due to hypertrophy or vascular hyperplasia.5 There are different theories about the genetic component: Moser C et al.,6 reported the case of an Italian family, where three female members in three successive generations had symptoms related with HH and two other relatives showed asymptomatic HH on ultrasound. Likewise, Diez Redondo P et al.,7 reported a family in which six members from three successive generations presented HH, suggesting that a clear family component might exist.
Women's disease preference may be explain by the expressiveness of a presumed "HH gene”, proliferate factors are the condition of been a woman, increased production of female sex hormones (during puberty or pregnancy) or the use of oral contraceptives. In fact, there are reports of growth and rupture of these tumors during pregnancy or in menopausal women undergoing hormonal treatment.2,5
Clinical features
In most cases HH do not present symptoms, they are most likely to be discovered incidentally during imaging studies for other unrelated conditions and if they present symptoms, they are nonspecific common symptoms to many other diseases of digestive origin (nausea, pain, abdominal distention, early satiety).4 Some authors report that there is a close relationship between tumor size and the presence of symptoms, 40% of the patients present symptoms in cases that hemangiomas measure more than 4cm and 90% in cases that hemangiomas measure more than 10cm denominated giant hepatic hemangiomas (HHG). Mainly symptoms are upper abdominal pain usually mild, but that can become severe in cases of thrombosis or bleeding within the lesion, which stretches and inflames Glisson's capsule.5
Physical examination is usually normal without significant findings, only rarely the hemangioma is presented as a large abdominal mass and an arterial murmur in the upper quadrant (Figure 1). Complications will depend on the location of the tumor and is generally present in cases of giant hemangiomas (HHG). It has been described circulatory shock and hemoperitoneum with spontaneous rupture, lower extremity edema by compression of the inferior vena cava in HHGs located in the caudate lobe. Compression of adjacent structures such as the stomach that can lead to gastric obstruction, bile ducts compression can lead to jaundice and hemobilia, colon compression can lead to volvulus or torsion, high expenditure heart failure due to massive arteriovenous shunt generally in children. Also can present hypothyroidism refractory to hormone therapy due to the presence of high levels of iodothyronine, deiodinase type 3 that favors the conversion of thyroxine and triiodothyronine to biologically inactive hormones and more rarely can present Kasabash Merritt syndrome (SKM).2,4,5
Photos from the department of Gastroenterology, Hospital Regional Docente de Enfermedades Neoplásicas
SKM is a rare and severe coagulation disorder characterized by profound thrombocytopenia, microangiopathic hemolytic anemia because of the sequestration and destruction of plaques and red blood cells in large tumors and consumption coagulopathy due to existing intratumoral fibrinolysis.8,9 In the laboratory tests have showed lower hemoglobin and hematocrit, thrombocytopenia, prolonged prothrombin time (PT), activated partial thromboplastin time (APTT), elevation of D - dimer, hypofibrinogenemia and degradation of fibrin products.9,10
Diagnosis
Different imaging studies such as ultrasound, computerized axial tomography (CT) and magnetic resonance imaging (MRI) with sensitivity greater than 90% and specificity between 55%-85% are used to make the diagnosis.2,11 Less frequently scintigraphy, positron emission tomography (PET) combined with computed tomography (CT) and angiography are used.
In ultrasound, HH classically appears as a homogeneous hyperechogenic focal lesion with a relatively clear limit, in addition of color with Doppler provides better qualitative and quantitative data thus increasing the sensitivity and specificity of the test.12 In recent years, it has begun to use microbubble ultrasonography where peripheral lesions show peripheral reinforcement that expand centripetally during the portal venous phase, completely filling in the delayed images. However, all these ultrasound characteristics are not usually observed in the HHG, where thrombosis, hemorrhage, fibrosis, or calcification show a heterogeneous image of the lesion12,13 (Figure 2).
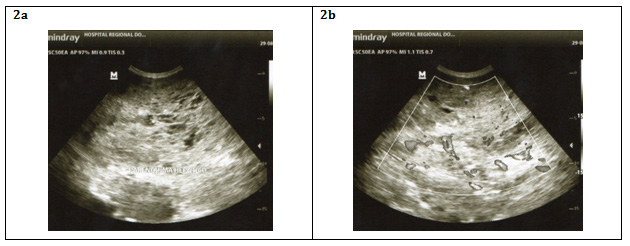
Figure 2 Big heterogeneous mass occupying both hepatic lobes with zones of hemorrhage and calcification that with the addition of Doppler can be delimited relatively.
Photos from the Department of Radiology, “Hospital Regional Docente de Enfermedades Neoplásicas”
CT with dynamic contrast (arterial, portal and late venous phases) is preferred before routine CT scan. It observes contrast enhancement around the edges of the tumor in the initial arterial phase, centripetal contrast improves in the portal phase and in the late venous phase, the contrast distributes uniformly throughout the tumor. However, when there is an intratumoral hemorrhage or areas of necrosis as in the HHG the hemangioma becomes a heterogeneous mass with high density inside it11 (Figure 3).
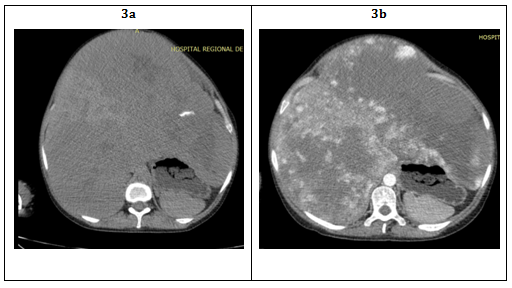

Figure 3 A: Hepatomegaly to predominance of the left hepatic lobe with multiple hypodense solid nodules, which tend to converge with each other throughout the hepatic parenchyma displacing caudal to other intra-abdominal organs.
B: After administration of contrast is evident a peripheral enhancement of the nodules in centripetal shape.
C: In the late phase the pattern of progressive centripetal filling is confirmed, as well as necrosis and calcification characteristics.
In MRI, images of hemangiomas have low signal intensity in T1 images and high signal intensity in T2 images, when uses gadolinium as a contrast agent intravenously; the images improve in a similar way to the observed in dynamic CT.14 It is important to note that HHGs may show fluid levels in CT and MRI images attributed to the separation of blood cells and serous fluid due to extremely slow blood flow through the tumor.15
Photos from the Department of Radiology, “Hospital Regional Docente de Enfermedades Neoplásicas”
Since 2001, hepatologists and surgeons are increasingly resistant to including liver biopsy as a diagnostic study in liver tumor lesions and only use it when the results of radiological studies and alpha-fetoprotein levels are inconclusive.15
It is evident that they are benign mesenchymal tumors irrigated by the hepatic artery; they can be cavernous hemangiomas, when formed by large dilated vascular ducts with abundant stroma associated in turn to intravascular thrombosis and calcification that represent 80% of the cases. More rarely can be capillary hemangiomas formed by capillaries of thin wall with little stroma3 (Figure 4).
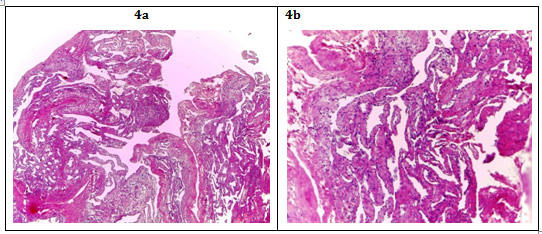
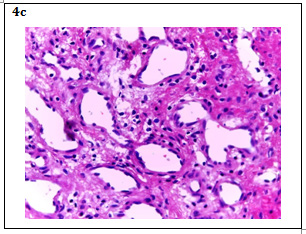
Figure 4 A: HE 4X Image: Empty spaces (channels) are observed and some with content, surrounded by scarce eosinophilic parenchyma.
B: HE 10X Image: It is observed that these channels are medium and large caliber blood vessels with endothelial lining of a single thin layer, in some of them are hematic content; The eosinophilic parenchyma corresponds to the hepatocytes that are being marginalized by vascular proliferation. Chronic inflammatory lymphocytic infiltrate is also observed around the blood vessels in response to the process of inflammation.
C: HE 40X Image: The endothelial cells that cover normal and dilated blood vessels, as well as the scarce parenchyma with some hepatocytes in degeneration and lymphocytic infiltrate, are more precisely observed.
Images from the department of Pathologic Anatomy, “Hospital Nacional Daniel Alcides Carrión”
Treatment
Only the patients who are symptomatic or complicated HHs, which are usually HHGs, should be treated, the first treatment option is surgery, since in 96% of cases, the symptoms improve, depending on the size and location of the lesion can perform open surgery or laparoscopy. Likewise, enucleation of the haemangioma, a segmental resection or complete lobectomy can possible do it.3,15
Among the other therapeutic options described in the literature, one option is arterial embolization, where the branches of the hepatic artery are embolized with polyvinyl alcohol and other substances that would cause a decrease in tumor size and relief of symptoms.16 However, rapid development of hepatic arterial collaterals decrease the final benefit of this non-extensive procedure. However, this treatment do not present serious complications such as ischemia, biliary cirrhosis, liver abscess, severe sepsis or intracavitary hemorrhage.17
Likewise percutaneous or laparoscopic radiofrequency ablation has been used successfully to improve abdominal pain in small series and in selected cases of liver transplantation justifying poor patient quality of life.3,18 The success of these therapies is largely related to the experience of the center where they are performed.
Since the 70s, several authors have proposed radiotherapy (RT) as a first-line treatment in unresectable HHG.19 Its efficacy has been well documented in medical literature based on symptom relief and growth interruption tumor, rather than its regression although in the latter aspect some recently studies have described regression of the tumor either early at 4months or late at 5years after RT.19,20
The majority of studies recommend doses of 20 and 30 Gy to obtain good dividing results in 10 to 20 fractions given in 2 to 4weeks, which in an acute phase would produce interethnic endothelial edema that is accompanied by a local increase of the colloidal matrix that is observed in the first 6months. Which would later lead to a chronic phase with late vascular fibrosis, obliteration and necrosis after 6months.19,20
Radiogenic hepatitis manifested clinically by rapidly growing hepatomegaly, ascites and sometimes jaundice and histologically by progressive fibrosis, central lobular congestion and hemorrhage is the main concern associated with the ambient temperature during HHG radiation, however it is transient and never observed with the recommended doses of 20 and 30 Gy.19,20
Complications in the gastrointestinal and renal tract are extremely rare, but possible, for which it is important to eliminate the possibility of irradiating these organs. A single case of liver cancer has been described as a late effect of RT that occurred after 20 years of the radiation of a HHG.21
In 2008, Léauté-Labrèze C et al.,22 demonstrated the usefulness of propanolol (which is a non-selective oral beta-blocker) in the treatment of proliferative HHs, which mainly acts on vasoconstriction, apoptosis and angiogenesis dysregulation. It has a high safety profile and few adverse reactions. In recent years, they have been widely used in the treatment of childhood HH with beneficial effects ranging from significant improvement to complete resolution of HH, for this reason it is reasonable to think about its use in adult HH, even some authors recommend the use of propanol in HHG.3
HHG despite not being a common disease is important to have it present in the differential diagnosis because this disease have many complications that can present if not made an appropriate treatment, even more when currently has several therapeutic options.
None.
Author declares there are no conflicts of interest.
None.

©2017 Montero, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.