eISSN: 2373-6372


Research Article Volume 17 Issue 1
Department of Internal Medicine, Wayne State University School of Medicine, USA
Correspondence: Paul Naylor, PhD Division of Gastroenterology, Department of Internal Medicine, Wayne State University School of Medicine, 6939 Hudson Murphy Building, 3990 John R, Detroit, MI 48202
Received: January 20, 2026 | Published: February 20, 2026
Citation: Atluri S, Mello M, Smadi R, et al. Racial disparity in weight loss and fibrosis of MASLD/MASH patients in a socioeconomically challenged primarily African American clinic setting . Gastroenterol Hepatol Open Access. 2026;17(1):1-5. DOI: 10.15406/ghoa.2026.17.00627
Introduction: Metabolic dysfunction-Associated Steatotic Liver Disease (MASLD) describes a group of patients with liver fat and metabolic risk factors for liver and cardiac issues. Patients who have significant liver fibrosis are classified as Metabolic dysfunction--Associated Steatohepatitis (MASH) and are at high risk for cirrhosis and hepatocellular carcinoma. This study evaluates racial disparity using a predominantly African American (AA) clinic population. Since weight loss and exercise are cornerstones of MASLD management, we evaluated non-invasive scoring systems for correlating weight changes and its impact on fibrosis.
Methodology: Patients were identified using ICD-10 billing codes and imaging for steatosis or the presence of the metabolic risk factors were confirmed using the medical records. The primary noninvasive fibrosis assessment was the NAFLD Fibrosis Score (NFS).
Results: There were 317 patients with MASLD/MASH seen between 2017 and 2023 with similar numbers for AA (177) and Non-AA (140) patients. Although AA were less likely to develop MASLD, the risk for developing MASH was similar. Regardless of race, NFS scores improved with weight loss and were worse with weight gain.
Discussion: This study confirms that AA are less likely to develop MASLD but are at similar risk for transition from MASLD to MASH. Weight loss correlated with improved fibrosis by NSF scoring regardless of the disease fibrosis or race. Weight gain led to a worsening of fibrosis.
Conclusion: Although the clinic’s population was 80% AA, MASLD/MASH rates among AA were only slightly higher compared to non-AA. However, if AA patients are diagnosed with MASLD, they are at similar risk for developing advanced fibrosis (MASH) as non-AA individuals. Given the similar risk of MASH regardless of race, the importance of referral to GI for evaluation of advanced fibrosis and using NFS as an assessment tool for fibrosis status should be emphasized.
Keywords: MASLD, MASH, Fibrosis, NFS, Steatosis, Steatohepatitis, NASLD, NASH African American
Metabolic dysfunction-Associated Steatotic Liver Disease (MASLD) describes a group of patients with liver fat and metabolic risk factors. These liver/heart risk factors include obesity (BMI≥25), type 2 diabetes (T2D), hypertension (HTN Blood Pressure ≥130/85 mm Hg), and elevated triglycerides (≥150mg/dL).1-3 While this nomenclature has replaced a previous descriptor of Non-Alcoholic Fatty Liver disease (NAFLD), the two entities are similar and can be used interchangeably when referring to earlier literature. 4 This is also true for the more advanced liver disease setting which reflects the progression of steatosis to steatohepatitis as a result of an increase in fibrosis to advanced stages. This advanced disease is designated Metabolic dysfunction--Associated Steatohepatitis (MASH) which was previously known as Non-Alcoholic Steatohepatitis (NASH).
MASLD is a highly prevalent disease due to the large population of individuals with the risk factors associated with liver steatosis and with a substantial subset of affected individuals progressing to advanced fibrosis (MASH). Patients with MASH are subsequently having an increase in liver cirrhosis and hepatocellular carcinoma development. Estimates of populations with MASLD approach 40% worldwide with higher incidences in Latin America and lowest incidence in Western Europe.
In addition to the variance in prevalence between overall ethnic country population there is also significant racial and ethnic disparities in countries such as the United States.5-10 In an early meta-analysis Hispanics had the highest burden and African Americans the lowest burden of NAFLD/MASLD, although the variance was modest. Similar results were also reported in two later analyses where the significant variability between individual studies also resulted in similar but modest racial trends. In a more recent study utilizing the Nonalcoholic Steatohepatitis Clinical Research Network observational studies, non-Hispanic African American persons in the US were found to have lower MASLD/MASH related disease rates of advanced fibrosis, cirrhosis and liver related events as compared to non-Hispanic whites. 10 Similar results were also reported from our institution using our predominant AA population.11 Thus, due to variability in the data and the lack of adequate representation of AAs in many studies, especially clinical trials, evidence regarding disease severity and racial disparities remains inconclusive. This study further utilizes an increased dataset to further evaluate racial disparity using a predominant AA clinic population and defined MASLD/MASH criteria. In addition to evaluating non-invasive scoring systems and demographics related to fibrosis development, we also focused on the relationship of weight change and liver fibrosis in AA vs non-AA patients with MASLD/MASH. This focus was important because weight loss is one of the cornerstones of MASLD management and we could use our predominant AA population to address this issue in the context of race.
Fatty liver has two billing scores which can be used to generate a set of patients who might have documented MASLD/MASH. The ICD-10 codes are K76.0 (fatty change of liver, not elsewhere classified) and K75.8 (Nonalcoholic steatohepatitis) and they were used to create a MASLD/MASH database. Individual patients with these codes were evaluated using the electronic medical records (EMR) and using the current definition of MASLD/MASH. Using this criteria, we identified 317 MASLD/MASH patients seen between 2017 and 2023. Imaging-defined steatosis was confirmed for 85% of the 317 patients with the remaining having documented at least two cardiometabolic risk factors, such as obesity, diabetes, HTN and lipidemia. Patients were defined as MASH (Metabolic disfunction Associated Steatohepatitis) if significant fibrosis was present as defined by at least one non-invasive laboratory result. NAFLD Fibrosis Score (NFS), FibroSure and FibroScan (kPa>14) were the primary criteria, although FIB-4, and APRI were also used. The complex NAFLD Fibrosis Score (NFS) was calculated in MDCalc using the formula of -1.675 + (0.037*age [years]) + (0.094*BMI [kg/m2]) + (1.13)IFG/diabetes [yes = 1, no = 0]) + (0.99*AST/ALT ratio) – (0.013*platelet count [×109/L]) – (0.66*albumin [g/dl]). The scoring range for NFS fibrosis was <-1.45 for F0-F2, >+ 0.675 for F3-F4 with indeterminate values listed as between -1.45 and +0.675. Also calculated for fibrosis were the less complex and more widely used FIB-4 and APRI.
Lab data at least 2 years apart were available for 121 patients such that a change in weight and fibrosis over time could be calculated. To have similar numbers in each of three categories prior to statistical analysis, we defined Weight Gain as >4% increase (n=28), Weight Stable as ±4% (n=38) and Weight Loss as >4% decrease (n=34). With respect to potential factors that could result in a decrease in weight, only a few patients had either weight loss surgery (10%) or received any period of GLP-1 agonist therapy (16%). Differences between numeric variables were evaluated using ANOVA and Chi Square analysis was used for character variables. Within-group changes in fibrosis scores as a function of weight change were assessed using paired t-tests.
The Wayne Health Clinic population is 80% African American (AA) but the number of MASLD/MASH patients does not reflect that demographic. In the dataset of 317 patients there were nearly similar percents of AA and non-AA patients (177 AA (56%); 140 non-AA (44%). The mean age was 51 years and 66% were female. The number of patients identified using the ICD-10 codes was similar between 2022 (Figure 1). There was a significant increase in the percentage of patients having a GI clinic visit with 2023 numbers approaching 75% of the patients identified that year.
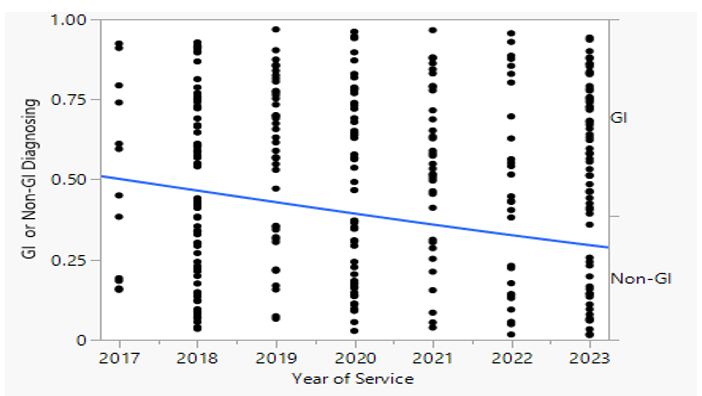
Figure 1 The number of patients seen each year was distributed between GI and non-GI visits. Percent of patients seen in GI increased each year such that 75% of MASLD/MASH patients in 2023 were seen by GI. Significance was plotted using the Logistic Fit by year of service and the increase in the % of patients seen in GI increased over time (p=0.01).
Irrespective of race, similar numbers of patients had liver steatosis documented by imaging (65%; figure 2). The remaining patients were defined as MASLD/MASH by the presence of metabolic risk factors. A few patients had metabolic risk factors without steatosis as defined by imaging (4%) and if imaging was not available, the dominant MASLD/MASH patients (24%) had obesity or diabetes either together or separately. When the risk factors were evaluated for all MASLD/MASH patients’ obesity was present in 90%. In the obese patients, 53 % of the population also had diabetes which was similar to that for the non-obese patients 42%. In patients diagnosed with steatotic metabolic disease an important issue is the presence or absence of fibrosis which provides for a stratification of risk for advancing to significant liver disease. The NFS score was the most likely to identify patients with advanced fibrosis (i.e. MASH) as compared to both FIB-4 and APRI (Figure 3). Conversely, FIB-4 and APRI were more likely to identify patients with minimal fibrosis as compared to NFS. There was no significant racial difference in the assessment of fibrosis by NFS or FIB-4 but APRI was more likely to result in Advanced Fibrosis scoring in non-AA as compared to AA patients.
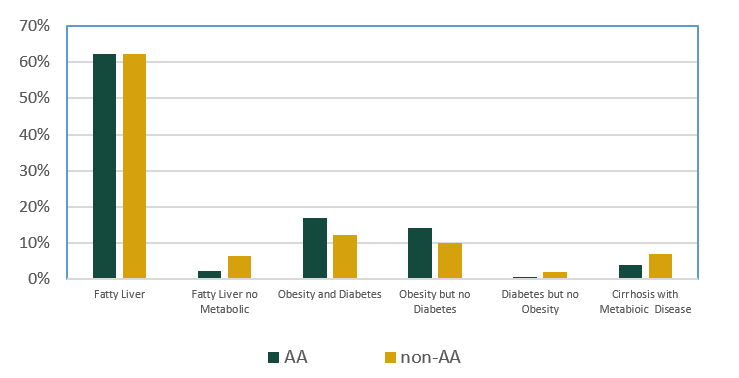
Figure 2 Criteria for MASLD/MASH in AA and non-AA patients. Most patients had documented fatty liver by imaging (AA similar to non-AA). For patients without imaging, obesity and diabetes were the primary metabolic risk factors. Few patients had imaging identifying fatty liver but no identifiable metabolic risk factors in the patients data in the EMD.
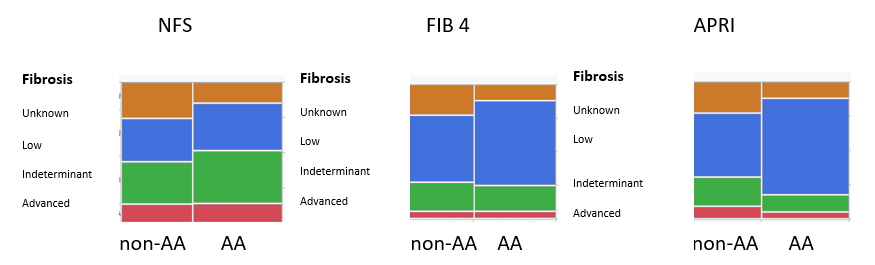
Figure 3 Racial Disparity in Assessing Fibrosis in MASLD/MASH Patients using Three Noninvasive Scoring Systems. The data is expressed as a mosaic plot with the width related to the ratio of AA to Non-AA. Fibrosis is expressed as Advanced (MASH); red), Indeterminate (green), Low Fibrosis (blue) or Unknown (brown).
With respect to the issue of advanced fibrosis (MASH) as compared to minimal fibrosis (MASLD), we utilized the definition that patients who had any one non-invasive assessment of fibrosis as sufficient to define MASH. With respect to the identification of significant fibrosis in patients who had steatotic metabolic disease, there was no racial variation in the percentage of individuals with advanced fibrosis (MASH) (Figure 4). This was both defined by AA vs non-AA and by race using subtypes in the non-AA population.
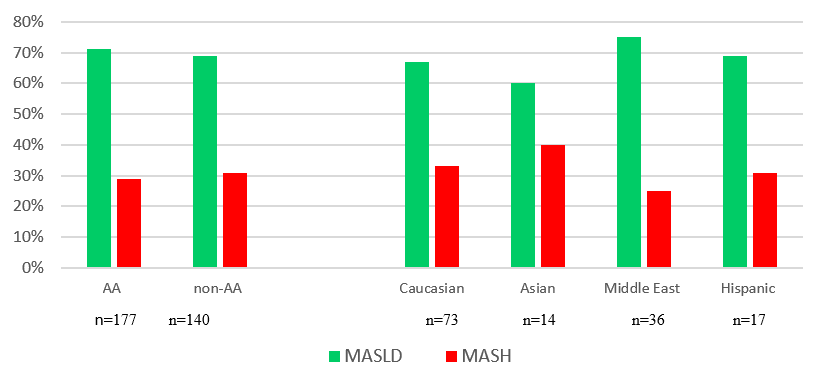
Figure 4 Racial Similarity for MASLD and MASH. Race was not a significant factor with respect to the development of advanced fibrosis (MASH). MASH was designated using a composite index with primary emphasis on NFS.
The three fibrosis assessment scoring systems were used to evaluate the effect of a change in weight on fibrosis (Figure 5; Table 1). NFS demonstrated a clear decrease in fibrosis with weight loss and an increase in fibrosis with weight gain. FIB-4 and APRI were less useful for comparing fibrosis and weight change (Table 1). Given the observation that NFS was effective in identifying patients with significant fibrosis, it was possible to use the dataset to evaluate the effect of weight change on fibrosis either by disease status (MASLD vs MASH) or race (AA vs non-AA). With respect to the influence of weight change on patients with minimal (MASLD) vs significant (MASH), patients were stratified by MASLD (F0-F2) versus MASH (F3-F4).
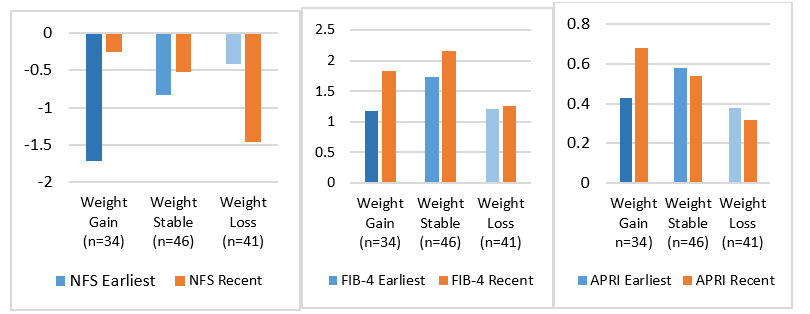
Figure 5 All MASLD/MASH patient change in NFS, FIB-4 and APRI with Weight Change. Weight loss led to improvement in fibrosis and weight gain resulted in worsening of fibrosis. Note that NFS score is reversed with lower numbers indicating more fibrosis.
|
NFS |
FIB-4 |
APRI |
|||||||
|
Earliest |
Recent |
p value* |
Earliest |
Recent |
p value* |
Earliest |
Recent |
p value* |
|
|
Weight Gain (n=34) |
-1.721 |
-0.254 |
0.0001 |
1.17 |
1.83 |
0.08 |
0.43 |
0.68 |
0.25 |
|
Weight Stable (n=46) |
-0.825 |
-0.518 |
0.06 |
1.73 |
2.16 |
0.05 |
0.58 |
0.54 |
0.56 |
|
Weight Loss (n=41) |
-0.415 |
-1.461 |
0.0008 |
1.21 |
1.26 |
0.67 |
0.38 |
0.32 |
0.01 |
Table 1 All MASLD/MASH patient weight and fibrosis by NFS, FIB-4 and APRI
*Significance defined using paired t-test for individual patients
Irrespective of whether the patients had MASLD or MASH, a loss of wight reduces fibrosis (Figure 6). Patients who had an increase in weight had an increase in fibrosis in the setting of either MASLD or MASH. Changes in FIB-4 and APRI mirrored this trend for weight gain, but the impact of weight loss on these scores was less evident.
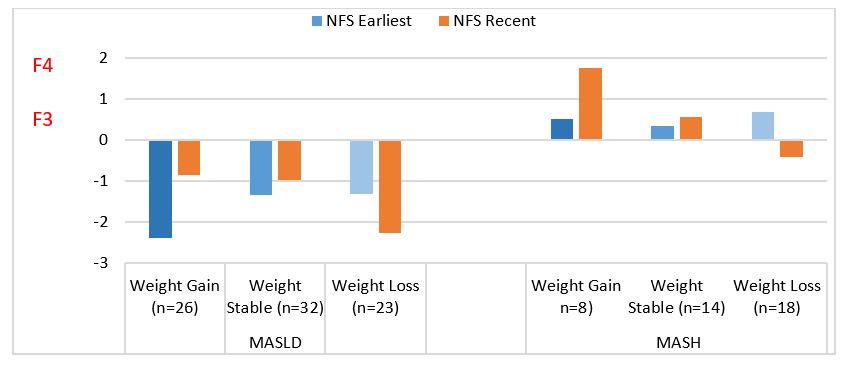
Figure 6 Change in NFS with Weight Change Stratified by MASLD vs. MASH. Weight gain led to increased fibrosis (higher number) regardless of initial status. Weight loss led to improvement in fibrosis (lower number).
Given the large population of AA patients in the dataset, it was possible to evaluate racial disparity and the effect of weight loss on fibrosis (Figure 7). Weight gain was associated with a significant increase in fibrosis (p < 0.001), while weight loss was associated with a significant reduction in fibrosis (p < 0.0008). This was consistent across both racial groups, although the magnitude of benefit from weight loss appeared greater among AA patients. Patients who gained weight had lower baseline fibrosis scores compared to those who maintained or lost weight.
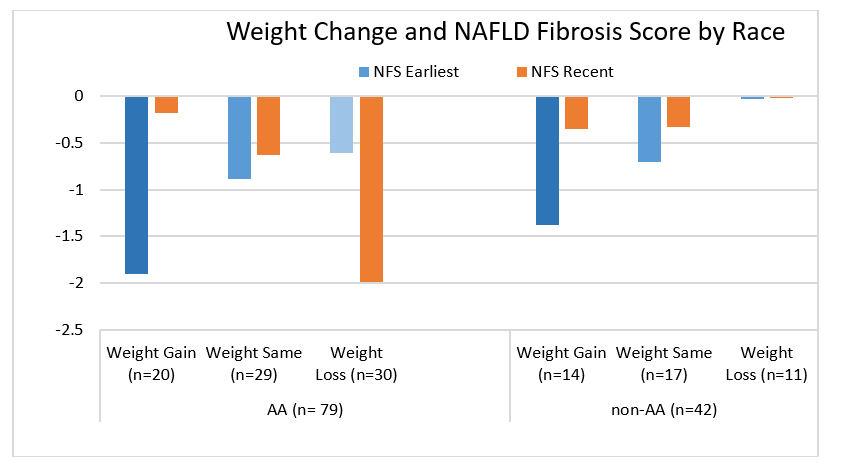
Figure 7 Weight Loss and Change in NFS by Race. The increase in NFS fibrosis score between the earliest (black bar) and in the recent (orange bar) reflects an increase in fibrosis with weight gain in both AA and non-AA patients. There was a decrease in fibrosis in AA patients with weight loss (orange bar greater than black bar.) Weight loss had minimal effect on fibrosis in non-AA patients.
The majority of MASLD patients have metabolic risk factors and most also have obesity with imaging defined steatosis.1-12 Many of the MASLD patients will develop fibrosis (MASH) that progresses from mild to severe. A portion of the MASH patients will subsequently develop advanced liver disease complications (cirrhosis and hepatocellular carcinoma). It is not yet possible to predict whether patients will progress, and, in many instances, there can be a reversal of fibrosis either naturally or by therapy.13-17 While biopsy assessment of fibrosis was originally used to track fibrosis, non-invasive assessment of fibrosis has replaced this except for therapy licensing studies where biopsy is often used in conjunction with non-invasive testing.18-22 This study utilized a predominately African American MASLD dataset to evaluate diversity with respect to disease manifestation and the utility of non-invasive serum-based assays to evaluate changes in fibrosis with weight loss.
Racial differences in the diagnosis of MASLD have been reported and this study confirms that AA patients are less likely to be diagnosed with MASLD than non-AA patients. An important observation using this large AA dataset was that although AA individuals are less likely to be identified with MASLD/MASH, patients with the diagnosis are not different from non-AA patients with respect to disease manifestation. AA and non-AA patients with MASLD/MASH were equally likely to have the disease diagnosed by ultrasound and to have similar metabolic risk factors. The patients in the clinic population are similar with respect to socioeconomic issues as defined by medical insurance which is predominantly Medicare/Medicaid and zip codes that are concentrated in the area served by the medical center. Thus, there would be no obvious reason for the variability in diagnosis of MASLD/MASH.
With respect to the issue of genetic disposition towards MASLD/MASH, a number of genes have been proposed to be suspected. Among them are the PNPLA3 gene linked to fibrosis and the anti-aging gene Sirtuin 1.23-26 With respect to PNPLA3 the G/G genotype has been strongly associated with advanced liver fibrosis and poor outcome in patients with advanced liver disease. Sirtuin 1 is critical to the prevention of liver steatosis, metabolic disease and fibrosis severity. In AA the levels of Sirtuin 1 may be lower and plasma Sirtuin 1 levels may need to be measured early to prevent and lower rates of MASLD/MASH compared with non-AA patients. It is also possible that the G/G genotype may also have a racial disparity distribution. Additional studies are clearly needed to address racial disparity and genetics in the future.
The observation that there is an increase from 2017 to 2023 in the number of patients who are being referred to and seen in the GI clinics probably reflects that increased awareness of the importance of getting fibrosis assessment to stratify the risk for progression to serious liver disease. Unfortunately the EMR does not provide sufficient data to determine the rational used by the physicians referring the patients to GI. Since the use of FibroScan has only recently been available in our clinic, our studies have focused on Fibrosis assessment by standard laboratory values. In our study, the NAFLD Fibrosis Score (NFS) yields the most patients with significant fibrosis as compared to FIB-4 and APRI. When evaluating the three standard assessments (NFS, FIB-4 and APRI) the only racial variability was in the APRI score which yields higher significant fibrosis in non-AA as compared to AA patients.
With respect to racial variability, there was also no difference in the percentage of individuals who developed significant fibrosis (MASH) regardless of whether the population was AA vs non-AA or AA vs subsets of non-AA patients. Thus, our observations are consistent with the hypothesis that after patients are diagnosed with MASLD, racial differences may not result in variability in the manifestation of the significant fibrosis in a cross-sectional study such as ours. Longitudinal studies evaluating increase in advanced fibrosis in the same patients over time may provide a different result. This is important because it confirms that the risk for progression to fibrosis is similar regardless of race and referral to GI should be regardless of race.
An area that has been somewhat understated is the role that weight loss has in the AA vs non-AA patients. Since the NFS scoring system yielded the best result for changes in weight and the effect on fibrosis, the scoring system was the primary criteria for evaluating fibrosis changes in our population. Weight change over at least 2 years was used to determine the effect of obesity on fibrosis. Weight loss had a positive effect on the improvement in fibrosis in both patients with minimal fibrosis (MASLD) and in patients with significant fibrosis (MASH). When the change in weight was evaluated based on AA vs non-AA, there was a significant increase in fibrosis when weight increased regardless of race. Unexpectedly, while a loss of weight had a significant improvement in fibrosis with weight gain for AA patients, the improvement in non-AA patients was not as significant. This observation suggests that further evaluation, perhaps in the setting of weight loss therapy in the context of GLP-1/GIP agents should be pursued. This is based on the more recent era where there is an increased use of the therapy in our Medicare/Medicaid dominant population and the approval of the use of GLP-1/GIP agents in MASLD/MASH patients.
Consistent with the literature and although the clinic’s population was 80% AA, MASLD/MASH rates among AA were only slightly higher compared to non-AA. The primary observation of this study is that if AA patients are diagnosed with MASLD (liver steatosis on imaging or steatotic metabolic disease) they are at similar risk for developing advanced fibrosis (MASH) as non-AA individuals. Given the similar risk of MASH regardless of race, the importance of referral to GI for evaluation of advanced fibrosis needs to be emphasized. Intervention to prevent or reverse significant fibrosis should be considered for both AA and non-AA patients. Regardless of race or baseline fibrosis severity, weight loss is associated with improvement in fibrosis as assessed by NAFLD Fibrosis Score (NFS). Weight gain is associated with worsening fibrosis. The benefit of weight loss appeared greater among AA patients, warranting further investigation. FIB-4 and APRI were less useful with respect to confirming that weight loss/gain were having an impact on fibrosis. The positive impact of weight loss on fibrosis was observed even in patients with more advanced disease (MASH), suggesting a potential benefit across the MASLD spectrum.
This research did not receive any specific grant from funding agencies.
Ethical approval from the Wayne State University Institutional Review Board was obtained (090616M1E)
There are no conflicts of interest.

©2026 Atluri, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.