eISSN: 2373-6372


Research Article Volume 16 Issue 5
Gastroenterology Department, Campus Teaching Hospital, Lomé, Togo
Correspondence: Laté Mawuli Lawson-Ananissoh, Gastroenterology Department, Campus Teaching Hospital, Lomé, Togo, Tel 0022890162651
Received: October 13, 2025 | Published: November 3, 2025
Citation: Lawson-Ananissoh LM, Gbolou MH, Roland-Moïse LK. Factors associated with acute kidney injury detected in cirrhotic patients 48 hours after admission to the Campus University Hospital in Lomé, Togo. Gastroenterol Hepatol Open Access. 2025;16(3):146-150. DOI: 10.15406/ghoa.2025.16.00624
Objective: to identify the factors associated with acute kidney injury in cirrhotic patients 48 hours after admission to the Campus Teaching Hospital in Lomé (Togo).
Methods: this was a descriptive and analytical cross-sectional study with prospective data collection over a 12-month period from January 2024 to December 2024. A Poisson regression model was implemented to identify factors associated with the occurrence of acute kidney injury (p < 0.20), taking into account interactions between variables with a stricter significance threshold of p < 0.05 for multivariate analysis. All statistical analyses were performed using R© statistical software version 4.4.2.
Results: during our study period, we identified 214 patients with cirrhosis, of whom 127 (59.3%) met our inclusion criteria. Forty-eight hours after hospitalization, 29 patients (22.8%) had acute kidney injury. Of the 29 patients with acute kidney injury forty-eight hours after hospitalization, 9 patients (31%) had normal absolute serum creatinine level The factors associated with acute kidney injury in our study were gastrointestinal bleeding (IRR: 2.8; 95% CI: [1.2-6.3]; p = 0.018), hepatic encephalopathy (IRR: 3.5; 95% CI: [1.4-9.0]; p = 0.008), and spontaneous ascites infection (IRR: 4.2; 95% CI: [1.1-12.7]; p = 0.018).
Conclusion: acute kidney injury is common in cirrhotic patients and is a factor associated with poor prognosis. Systematic monitoring of renal function 48 hours after hospitalization is an effective means of early detection of this complication, allowing for timely management.
Keywords: cirrhosis, acute kidney injury, 48 hours after admission, Togo
Cirrhosis is a major cause of high morbidity and mortality worldwide. In 2019, it was associated with 2.4% of deaths worldwide.1 Initially, cirrhosis is said to be compensated when no complications are found. However, it can progress to a stage of decompensation marked by the occurrence of complications such as ascites, hepatic encephalopathy, gastrointestinal bleeding due to portal hypertension, carcinomatous degeneration, kidney failure, and even hepatorenal syndrome. Kidney failure in cirrhosis is common and typically includes acute and chronic kidney failure.2 The incidence of acute kidney injury (AKI) worldwide varies from 20% to 50% in hospitalized cirrhotic patients.3,4 A study conducted in 2024 by Alioune et al in Senegal found a prevalence of 52.60%.5 In Togo, a study conducted in 2018 found a prevalence of 30%.6 None of these studies focused on early screening, particularly 48 hours after hospitalization for acute kidney injury in hospitalized cirrhotic patients. However, the definition of acute kidney injury in cirrhotic patients has undergone numerous recent updates, now taking into account dynamic variations in serum creatinine rather than its absolute value, as creatinine is a poor marker of renal function, varying according to gender, ethnicity, and weight.7 In cirrhotic patients, serum creatinine levels are often low for the following reasons: (a) decreased creatinine production due to sarcopenia, which is more common in severe cirrhosis, (b) decreased hepatic creatine synthesis due to liver failure, (c) increased distribution volume due to water and sodium retention, which dilutes creatinine, and (d) interference from hyperbilirubinemia in creatinine measurement using the colorimetric method.8 The occurrence of AKI is associated with high mortality, with an approximately 300% increased risk of death.9 This high mortality risk therefore requires early detection of this complication and better care of these patients, hence the interest of this study, the objective of which was to identify the factors associated with the occurrence of acute kidney injury in cirrhotic patients 48 hours after admission to the Campus University Hospital in Lomé.
This was a descriptive and analytical cross-sectional study, with prospective data collection over a 12-month period from January 2024 to December 2024, carried out in the Hepato-gastroenterology department of the Lomé University Hospital Campus. It included all cirrhotic patients hospitalized during the study period. All cirrhotic patients who had their creatinine levels measured upon admission and 48 hours after admission to the department were included in our study. The following patients were not included in this study: those with known chronic kidney disease; those who did not spend 48 hours in the department during their hospitalization; those who were diagnosed with acute kidney injury upon admission. Cirrhosis was diagnosed on the basis of epidemiological, clinical, biological, endoscopic and morphological criteria. Acute kidney injury was defined as an increase in serum creatinine of at least 3 mg/L (26.4 μmol/L) within 48 hours. The outcome was favorable in a patient whose renal function had improved after treatment; it was unfavorable in a patient whose renal function had not improved despite treatment, or if the patient needed to be transferred to the nephrology department, or if the patient died or was discharged without renal function improvement. Data were collected using a digitalized electronic questionnaire via the KoboToolbox platform. They were then exported as an Excel database. A Poisson regression model was implemented to identify factors associated with the occurrence of acute kidney injury (p < 0.20), taking into account interactions between variables. A stricter significance threshold of p < 0.05 was applied for significant parameters in the multivariate analysis. All statistical analyses were performed using statistical software R© version 4.4.2. The study protocol was submitted for approval to the Ethics Committee of the Faculty of Health Sciences of the University of Lomé, and authorization was obtained from the administrative officials of the Campus University Hospital of Lomé. Patients were given explanations regarding the objectives of the study so that they could give their verbal consent. When counting and collecting data, patients' names were not collected, but an identification number was assigned to preserve anonymity and confidentiality.
We identified 214 cirrhotic patients, 127 of whom met our inclusion criteria. Of the 127 patients included, 29 had AKI 48 hours after hospitalization, representing a frequency of 22.8%. The flow chart of our study population is shown in Figure 1. The mean age of patients was 49 years ± 13 with extremes of 24 and 87 years. Males were the most represented, with 100 men (78.7%), giving a sex ratio of 3.70. The complications of cirrhosis included gastrointestinal bleeding and spontaneous infection of ascites fluid in 11.0% and 3.1% of patients, respectively; 99 patients (78%) were in stage C of the Child-Pugh classification. Of the 29 patients with AKI at H48, 9 patients (31%) had normal absolute serum creatinine level. The mean age of patients was 48 years ± 14, with extremes of 27 and 79 years. There were 25 men with a sex ratio of 6.25. The mean serum creatinine level in our patients was 11.88 ± 9.18 mg/L with a mean hemoglobin level of 10.08 ± 2.27 g/L. Table 1 shows the clinical, prognostic, therapeutic, and evolutionary data for patients with AKI at H48. Table 2 shows the relationship between sociodemographic characteristics, clinical data, and complications with acute kidney injury. After univariate and multivariate analysis (Table 3), it appears that patients with gastrointestinal bleeding were 2.8 times (95% CI = [1.2-6.3] with p = 0.018) more likely to have AKI at H48. This risk was 3.5 times higher (95% CI = [1.4-9.0] with p = 0.008) in those with hepatic encephalopathy. There was a 4.2times (95% CI = [1.1-12.7] with p = 0.018) increased risk of AKI in patients with Spontaneous ascitic fluid infection (SAI).
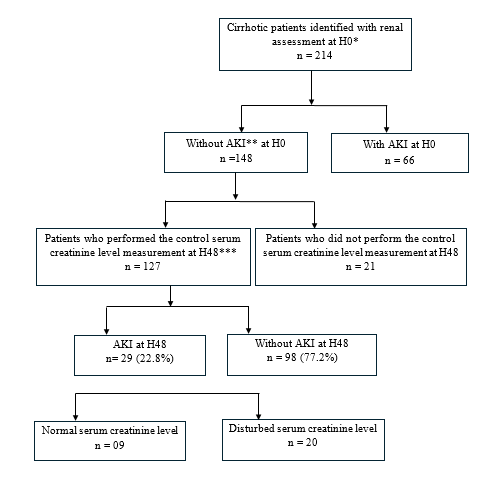
Figure 1 Flow chart of the study population.
*at the admission; **acute kidney failure; ***48 hours after admission
|
n |
% |
|
|
Precipitating factors for AKI |
||
|
Infusion |
17 |
58.6 |
|
Diuretic |
8 |
27.6 |
|
Beta blocker |
6 |
20.7 |
|
Antihypertensive |
3 |
10.3 |
|
Clinical signs |
||
|
Ascites |
25 |
86.2 |
|
Jaundice |
23 |
79.3 |
|
Cutaneous and mucosal pallor |
19 |
65.5 |
|
Low blood pressure |
14 |
48.3 |
|
Oliguria/ Anuria |
13 |
44.8 |
|
Other complications of cirrhosis |
||
|
Ascites |
25 |
86.2 |
|
Hepatic encephalopathy |
18 |
62.1 |
|
Digestive Hemorrhage |
13 |
44.8 |
|
Spontaneous ascites infection |
4 |
13.8 |
|
Prognosis |
||
|
Child-Pugh A |
2 |
6.9 |
|
Child-Pugh B |
2 |
6.9 |
|
Child-Pugh C |
25 |
86.2 |
|
Treatment |
||
|
Albumin |
3 |
10.3 |
|
Red blood cell transfusion |
4 |
13.8 |
|
Isotonic saline serum |
24 |
82.8 |
|
Evolution |
||
|
Favorable |
14 |
48.3 |
|
Unfavorable |
15 |
51.7 |
Table 1 Clinical, prognostic, therapeutic and evolutionary data of patients with acute kidney injury 48 hours after admission
|
Acute kidney injury |
p-value |
|||
|
No (n=98) |
Yes (n=29) |
Total (n=127) |
||
|
Age (years), n (%) |
0.1071 |
|||
|
<50 |
51 (71.8) |
20 (28.2) |
71 |
|
|
≥50 |
47 (83.9) |
9 (16.1) |
56 |
|
|
Gender, n (%) |
0.2631 |
|||
|
Female |
23 (85.2) |
4 (14.8) |
27 |
|
|
Male |
75 (75.0) |
25 (25.0) |
100 |
|
|
Child-Pugh Score, n (%) |
0.2221 |
|||
|
Child-Pugh A/B |
24 (85.7) |
4 (14.3) |
28 |
|
|
Child-Pugh C |
74 (74.7) |
25 (25.3) |
99 |
|
|
Precipitating factors for AKI |
|
|
|
|
|
Diuretic, n (%) |
|
|
|
<0.0012 |
|
No |
95 (81.9) |
21 (18.1) |
116 |
|
|
Yes |
3 (27.3) |
8 (72.7) |
11 |
|
|
Infusion, n (%) |
|
|
|
0.0011 |
|
No |
72 (85.7) |
12 (14.3) |
84 |
|
|
Yes |
26 (60.5) |
17 (39.5) |
43 |
|
|
Beta blocker, n (%) |
|
|
|
0.0872 |
|
No |
90 (79.6) |
23 (20.4) |
113 |
|
|
Yes |
8 (57.1) |
6 (42.9) |
14 |
|
|
Antihypertensive, n (%) |
|
|
|
0.0782 |
|
No |
96 (78.7) |
26 (21.3) |
122 |
|
|
Yes |
2 (40.0) |
3 (60.0) |
5 |
|
|
Clinical data |
|
|
|
|
|
Cutaneous and mucosal pallor, n (%) |
|
|
|
<0.0011 |
|
No |
92 (90.2) |
10 (9.8) |
102 |
|
|
Yes |
6 (24.0) |
19 (76.0) |
25 |
|
|
Oliguria/Anuria, n (%) |
<0.0012 |
|||
|
No |
98 (86.0) |
16 (14.0) |
114 |
|
|
Yes |
0 (0.0) |
13 (100.0) |
13 |
|
|
Low blood pressure, n (%) |
<0.0012 |
|||
|
No |
98 (86.7) |
15 (13.3) |
113 |
|
|
Yes |
0 (0.0) |
14 (100.0) |
14 |
|
|
Complications |
|
|
|
|
|
Digestive hemorrhage, n (%) |
<0.0012 |
|||
|
No |
97 (85.8) |
16 (14.2) |
113 |
|
|
Yes |
1 (7.1) |
13 (92.9) |
14 |
|
|
Hepatic encephalopathy, n (%) |
<0.0012 |
|||
|
No |
98 (89.9) |
11 (10.1) |
109 |
|
|
Yes |
0 (0.0) |
18 (100.0) |
18 |
|
|
Spontaneous ascites infection, n (%) |
0.0022 |
|||
|
No |
98 (79.7) |
25 (20,3) |
123 |
|
|
Yes |
0 (0.0) |
4 (100.0) |
4 |
|
Table 2 Acute kidney injury 48 hours after admission according to patient’s characteristics
|
Univariable model |
|
Initial multivariable model |
|
Final multivariable model |
|||||||||||||
|
IRR |
CI 95% |
p-value |
|
IRR |
CI 95% |
p-value |
|
IRR |
CI 95% |
p-value |
|||||||
|
Age (years) |
|
|
|||||||||||||||
|
<50 |
- |
- |
|
1.00 |
- |
|
1.00 |
- |
|||||||||
|
≥50 |
0.57 |
0.25-1.22 |
0.162 |
|
0.68 |
0.27-1.59 |
0.384 |
|
0.60 |
0.25-1.34 |
0.235 |
||||||
|
Gender |
|
|
|||||||||||||||
|
Female |
- |
- |
|
1.00 |
- |
|
1.00 |
- |
|||||||||
|
Male |
1.69 |
0.65-5.73 |
0.331 |
|
1.23 |
0.39-4.80 |
0.738 |
|
1.45 |
0.48-5.45 |
0.540 |
||||||
|
Child-Pugh Score |
|
|
|||||||||||||||
|
Child-Pugh A/B |
- |
- |
|
|
|||||||||||||
|
Child-Pugh C |
1.77 |
0.69-6.00 |
0.29 |
|
|
||||||||||||
|
Oliguria/anuria |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
1.00 |
- |
|||||||||
|
Yes |
7.13 |
3.37-14.8 |
<0.001 |
|
2.05 |
0.76-5.78 |
0.161 |
|
2.26 |
0.86-6.27 |
0.105 |
||||||
|
Low blood pressure |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
1.00 |
- |
|||||||||
|
Yes |
7.53 |
3.60-15.7 |
<0.001 |
|
1.52 |
0.63-3.67 |
0.346 |
|
1.53 |
0.60-3.98 |
0.373 |
||||||
|
Digestive hemorrhage |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
1.00 |
- |
|||||||||
|
Yes |
6.56 |
3.10-13.6 |
<0.001 |
|
2.46 |
0.89-7.13 |
0.088 |
|
2.75 |
1.17-6.35 |
0.018 |
||||||
|
Hepatic encephalopathy |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
1.00 |
- |
|||||||||
|
Yes |
9.91 |
4.75-21.7 |
<0.001 |
|
3.47 |
1.48-8.44 |
0.005 |
|
3.50 |
1.41-9.00 |
0.008 |
||||||
|
Spontaneous ascites infection |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
1.00 |
- |
|||||||||
|
Yes |
4.92 |
1.45-12.7 |
0.003 |
|
5.02 |
1.21-17.8 |
0.016 |
|
4.18 |
1.12-12.7 |
0.018 |
||||||
|
Diuretic |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
|||||||||||
|
Yes |
4.02 |
1.67-8.73 |
<0.001 |
|
2.43 |
0.91-6.09 |
0.064 |
|
|||||||||
|
Infusion |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
|||||||||||
|
Yes |
2.77 |
1.33-5.94 |
0.007 |
|
0.96 |
0.39-2.38 |
0.937 |
|
|||||||||
|
Cutaneous and mucosal pallor |
|
|
|||||||||||||||
|
No |
- |
- |
|
1.00 |
- |
|
|||||||||||
|
Yes |
7.75 |
3.68-17.4 |
<0.001 |
|
1.26 |
0.36-4.36 |
0.713 |
|
|||||||||
Table 3 Factors associated with acute kidney injury detected in cirrhotic patients 48 hours after admission (N = 127)
CI, confidence interval; IRR, incidence rate report
The relatively small size of our sample is due to the fact that most of our cirrhotic patients did not undergo the renal assessment 48 hours after hospitalization due to lack of financial resources. Nevertheless, this study enabled us to detect acute kidney injury in some of our patients early, i.e., 48 hours after hospitalization, thereby facilitating early care of this complication. As this study is the first of its kind, it draws practitioners' attention to this serious complication of cirrhosis, which can go unnoticed if renal function tests are not carried out systematically and at an early stage in these patients. In our study, 29 (22.8%) patients had AKI. Importantly, 9 of the 29 patients, i.e. 31%, had normal absolute creatinine levels despite having AKI compared to their creatinine levels at admission. This confirms the definition of AKI in cirrhotic patients, which must now take into account creatinine kinetics rather than its absolute value, thus demonstrating that creatinine is a poor marker of renal function in cirrhotic patients.10 We did not find any studies in the literature that used the same collection method as ours, i.e., measuring serum creatinine levels 48 hours after hospitalization. However, our result is close to that found by Mohan et al in Ireland in 2024 which was 25.2%.11 Duah et al in Ghana in 2022 found a prevalence of 27.9%.12 On the other hand, Moussa et al.13 in Tchad in 2017 and Alioune et al.5 in Senegal in 2024 reported prevalences of 53% and 52.60%, respectively. These significant differences could be explained by the fact that in these studies, all cirrhotic patients who had AKI regardless of the time of hospitalization were included, whereas in our study, we only included patients who had AKI 48 hours after hospitalization. Ascites and jaundice were the most common clinical signs found in our patients. Our results are similar to those of Duah et al.12 and Mohan et al.11 in their studies conducted in Ghana and Ireland, respectively. These findings can be explained by the fact that AKI often occurs in cirrhotic patients with advanced hepatocellular insufficiency. Ascites and jaundice being the main signs of hepatocellular insufficiency. Hyponatremia was the most common biological sign found in our patients. This could be explained by the pathophysiological mechanisms of cirrhosis. Indeed, following splanchnic arterial vasodilation, there is a decrease in arterial blood volume, leading to arterial hypotension. In response to this hypotension, there is an overproduction of antidiuretic hormone, which causes free water retention, resulting in hyponatremia.14 Hepatic encephalopathy was significantly associated with acute kidney injury in patients with cirrhosis. The same observation was made by Kogiso et al.15 in Japan in 2021 and by Duah et al.12 in Ghana in 2022. These results can be explained by the fact that AKI in cirrhotic patients itself causes hepatic encephalopathy through the accumulation of ammonia, which is a neurotoxic substance.16 Gastrointestinal bleeding was associated with the occurrence of AKI in our study. This is explained by the fact that digestive hemorrhage accentuates the hypovolemia already created by a third sector existing in almost all of our cirrhotic patients due to ascitic decompensation. This hypovolemia was also recognized as impairing renal function in the study conducted by Duah et al.12 Spontaneous ascitic fluid infection (SAIS) is recognized in our study as a factor associated with the occurrence of AKI. This same finding was made by Alioune et al.5 and Duah et al.12 Bacterial infections are responsible for the production of large quantities of pro-inflammatory cytokines, leading to circulatory deterioration with reduced tissue perfusion, direct organ damage, and ultimately organ failure, including kidney failure, which characterizes what is now known as acute-on-chronic liver failure (ACLF), corresponding to acute hepatocellular insufficiency against a chronic background.17 Recent advances in research on sirtuins, which regulate cell growth, DNA repair, and stress adaptation, could provide a pathway for developing biomarkers for the early diagnosis and treatment of kidney failure related to metabolic diseases.18
AKI is common in cirrhotic patients and is a poor prognostic factor. Approximately one-third of patients with AKI had normal serum creatinine levels at admission and 48 hours after hospitalization. Systematic monitoring of renal function 48 hours after hospitalization is an effective means of early detection of this complication, enabling it to be treated more promptly in order to prevent it from worsening or leading to other complications. Factors associated with the occurrence of acute renal failure were gastrointestinal bleeding, hepatic encephalopathy, and Spontaneous ascitic fluid infection.
None.
The authors declare no conflict of interest to this work.
Conception and design of the study: LM Lawson-Ananissoh and MH Gbolou.
Acquisition of data: Pélagie Kouessi, LRM Kogoe and DV Redah.
Analysis and interpretation of data: Pélagie Kouessi, Gbolou MH and Kanake YY.
Drafting the manuscript: LRM Kogoe and LM Lawson-Ananissoh.
Approval of the version of the manuscript to be published: A Bagny, LM Lawson-Ananissoh.

©2025 Lawson-Ananissoh, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.