eISSN: 2473-0815


Case Report Volume 10 Issue 2
1Clinic of Endocrinology and Metabolism, School of Medicine, Hospital of Clinics, Uruguay
2Pathology clinic, School of Medicine, Hospital of Clinics, Uruguay
3Pathology Clinical’ s director, School of Medicine, Hospital of Clinics, Uruguay
4Professor director’ Clinic of Endocrinology and Metabolism, School of Medicine, Hospital of Clinics, Uruguay
5Clinic of Endocrinology and Metabolism, Associate professor, School of Medicine, Hospital of Clinics, Uruguay
Correspondence: Zara Martínez, Clinic of Endocrinology and Metabolism, School of Medicine, Hospital of Clinics, UdelaR, Montevideo, Uruguay
Received: November 04, 2022 | Published: November 18, 2022
Citation: Martínez Z, Cristiani A, Centurion D, et al. Case report: combined pattern thyroid carcinoma. Endocrinol Metab Int J. 2022;10(2):54-56 DOI: 10.15406/emij.2022.10.00319
Thyroid cancer accounts for approximately 3% of all new cancer diagnoses per year globally.1 Papillary thyroid carcinoma (PTC) is the most common type, followed by follicular thyroid carcinoma (FTC).1 Recently, the simultaneous appearance of different histological variants in the same patient has been reported, being called combined pattern thyroid cancer, mixed tumors, hybrid tumors or compound tumors of the thyroid gland.2 Epidemiologically, combined pattern cancer is reported in less than 2% of cases, it presents the same proportion in men and women, and the age of presentation is between 18 and 45 years.3 Different associations are found, including mixed carcinomas: a) medullary cancer (CMT) and PTC; b) PTC and fasciitis-like stroma; c) CPT and CFT; and even combinations of CMT, CPT and CFT.4 Several studies suggest a more aggressive clinical behavior, which may be related to the coexistence of multiple patterns of aggressive variants, with early detection and rapid and aggressive treatment being of vital importance. It is based on surgical treatment, moderate-high dose radioiodine and TSH suppression (<0.1mIU/L).4 This achieves a cure rate of up to 66% and survival three years after diagnosis of 90% and 10 years of 80% to 95%.
Keywords: thyroid, cancer, thyroid cancer, combined pattern, thyroid disease
The lifetime relative risk of developing thyroid cancer is 1.2%, representing 3% of all new cancer diagnoses per year globally.1 Most present as palpable masses in the neck, which can be evaluated by ultrasound studies, fine needle aspiration (FNA), followed by cytological evaluation.1
PTC, with its many variants, and CFT are the first and second most common thyroid cancers, comprising nearly 85% and 10% of all thyroid cancers, respectively.1
In the literature, the simultaneous appearance of different histological variants of thyroid cancer in the same patient has been reported, with different presentations, among the most common the concomitant appearance of MTC and PTC, PTC with dedifferentiation to anaplastic carcinoma (ATC), PTC with fasciitis-like stroma, and PTC with PTC, the latter being an unusual event that is rarely reported.2 In this article we present a clinical cases of patient with simultaneous appearance of different thyroid cancer subtypes (Figure 1).
This is a 40-year-old woman with a history of obesity and smoking, no history of head and neck radiation or family history of thyroid cancer. Her consultation was due to a large cervical mass of one year's evolution, functional class II dyspnea on exertion, weight loss of 23 kg in one year, asthenia, and adynamia. On physical examination, an oval, fibroelastic mass with irregular edges, painless, of approximately 8cmx4cm, and various adenopathies of roughly 2cmx2cm in level III of the neck were palpated in the neck.
Basic paraclinical tests are requested, with normal results and TSH 2.06mUI/L. Neck ultrasound an hypoechoic nodule, having microcalcifications, intense central and peripheral vascularization, 50x36x35mm. There is a carotid-jugular adenopathic conglomerate to the right, with peripheral vascularization 113x45x25mm. Multiple carotid-jugular adenomegalies on the left, the largest 10x15mm. TIRADS 5.
Ultrasound-guided FNA was performed on the largest nodule in the right lobe, reporting PTC (Bethesda VI) and jugular-carotid lymphadenopathy with the presence of PTC metastases.
The patient underwent total thyroidectomy (TT), right-side lymph node dissection, and delayed left lymph node dissection.
Pathological anatomy (PA): multifocal papillary thyroid carcinoma, with a focus greater than 40mmx39mm on the right. It has multiple foci, presenting as tall cell variants on both the right and left. Foci of encapsulated CFT are observed, mostly with capsular evasion, variants of oncocytic cells with three mitoses in 10 high-power fields (HGP), and several foci of conventional histology. It has an extrathyroidal extension that contacts a bloody margin both to the right and to the left.
Right lymph node conglomerate: 54 lymph nodes, 26 show malignant neoplastic involvement. The largest metastasis corresponds to a 70mm conglomerate on the right, from group II. Left lymph node conglomerate: 65 nodes, 7 of them with neoplastic involvement, the largest of 18mm. TNM classification: T4aN1bMx.
After the first surgery, 150 mCi of I131 is applied. Scintigram with SPECT/CT of the neck and chest on the seventh day after radioiodine: areas of intense radiotracer uptake in projection to the lower poles of the thyroid lodge and the upper pole of the left thyroid lodge. In addition, intense hyper-uptake is observed in a right high jugular carotid node (level IIb) of 10x12x8mm and the dorsal column at the level of the left lamina of T9. Thyroglobulin(Tg): 22.7ng/ml, Anti-thyroglobulin antibodies: 15.9 I/ml, TSH: 38.7µU/ml.
Three months later, the patient went to the medical office for control, with Tg:11.4ng/ml; Anti-thyroglobulin antibodies: 15U/ml, TSH:11µU/ml. Cataloging the outcome as an incomplete structural response, a TSH target of less than 0.1µU/ml is decided.
A combined pattern of thyroid cancer is defined as the presence of two or more well-differentiated morphological patterns in the same case, this being a highly infrequent event,3 also called “collision tumor”, related to coexisting but independent tumors which are histologically distinct.5
Epidemiologically, combined pattern cancer is reported in less than 2% of cases, it presents the same proportion in men and women, and the age of presentation is between 18 and 45 years.3 It generally occurs in carcinomas > 1cm in diameter, thus differentiating itself from microcarcinomas, which present a higher percentage of the classic variant.3
Each of the variants that compose it must be represented in at least 30% of the tumor cells. This combined pattern is made up of a predominant subtype, which corresponds to the histological variant with the highest percentage of tumor cells, and a secondary subtype, which is related to the subsequent histological variant with the following highest rate of tumor cells (Figure 2).2
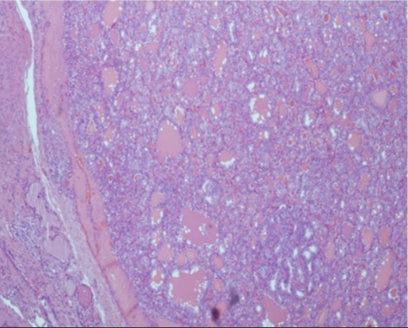
Figure 2 The largest tumor was partially encapsulated and showed follicular pattern, with conventional papillary carcinoma cytology.
Several studies suggest a more aggressive clinical behavior and may be related to the coexistence of multiple patterns of aggressive variants. This results in larger tumor size, more significant extrathyroidal extension, and risk of invasion of the thyroid capsule, lymph node metastases, and multiple distant metastases compared to single-variant tumors.4
In other words, the combined pattern seems to constitute an aggressive subtype that implies a higher risk of local recurrence. This phenomenon allows us to suppose that the progression of carcinogenesis in thyroid cells co-occurs in several clones. Another hypothesis is that other cell lines could derive from an initial clone.4 This allows us to state that the combined pattern, regardless of tumor size, should be considered a high-risk variant.3
This case is about a young woman who debuted with a large tumor mass associated with a significant right adenopathic conglomerate and general repercussion of one year of evolution, which is an unusual clinical expression of differentiated thyroid cancer. Given the characteristics of the pathological anatomy, we think that this particular form of presentation is related to the combined pattern of differentiated cancer, which gives it a more aggressive behavior.
The prognosis is determined by the presence or absence of metastases and age (older age, worse prognosis). In this case, there is a young, middle-aged patient who has lymph node metastases of large proportions and in large numbers, which persist to date.
Origin: although the synchronous coexistence of more than one type of thyroid cancer could be a coincidence, several theories have been postulated for such synchronous coexistence.7
It is a cancer type with greater aggressiveness, for which rapid and intensive therapy is promoted. The healing percentage is 66%.5,8 Therefore, it should be emphasized that the cure rate increases when there is an early uptake of the disease and, thus, early and appropriate treatment.8 Also, it is known that a complete preoperative evaluation, the application of radioiodine therapy at intermediate-high doses, and a strict follow-up after surgery, with suppression of TSH, with a goal of less than 0.1mIU/L, is essential.7,9
Abdelrahman Abdelaal et al. reported that the three-year survival rate for malignant thyroid cancer was 90%,7 therefore, supporting that the prognosis of differentiated thyroid carcinoma is favorable, with a 10-year survival rate of 80%-95%. In addition, earlier detection of small differentiated thyroid cancer with less extensive disease and standardization of treatment may contribute to decreased disease-specific mortality in such patients (Figure 4).2,3,9
Álvaro Ronco. Physician, Associate Professor of Oncology. CLAEH Faculty of Medicine. Maldonado, Uruguay. Researcher in Epidemiology of Cancer. Oncology Unit and Radiotherapy, Women's Hospital, Center Hospitable Pereira Rosell. Montevideo, Uruguay.
The authors declare no conflict of interest.

©2022 Martínez, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.