eISSN: 2378-315X


Review Article Volume 13 Issue 3
1Department of Pharmacy, Philadelphia College of Osteopathic Medicine, USA
2Department of Pharmacy, Wellstar North Fulton Medical Center, USA
3Department of Hematology/Oncology with Northwest Georgia Oncology Centers, USA
Correspondence: Zachary M Powell, Department of Pharmacy, Philadelphia College of Osteopathic Medicine, Philadelphia, Pennsylvania, USA
Received: August 09, 2024 | Published: August 30, 2024
Citation: Powell ZM, Thomas SA, Mankan N. Evaluating the use of olanzapine for prevention of chemotherapy-induced nausea and vomiting (CINV) for highly emetic chemotherapy regimens.Biom Biostat Int J. 2024;13(4):90-94. DOI: 10.15406/bbij.2024.13.00419
Background: Pharmacological management of chemotherapy-induced nausea and vomiting (CINV) includes antagonizing serotonin, neurokinin, dopamine, and histamine receptors involved in the emetic response from the vomiting center. In addition to traditional antiemetic triplet therapy with dexamethasone, neurokinin-1 receptor antagonists and serotonin antagonists recommended by the National Comprehensive Cancer Network (NCCN) for highly emetic chemotherapy (HEC), olanzapine is an atypical antipsychotic that has seen increased utilization alongside triplet therapy due to its antagonistic effects on receptors involved in the CINV signaling pathways.
Objective: The objective of this study was to evaluate the efficacy of olanzapine in reducing the severity and frequency of CINV due to HEC and provide real-world evidence of its use in clinical practice.
Methods: This study was a multicenter, retrospective, electronic chart review of patients aged 18 years and older with a confirmed cancer diagnosis who received HEC at Wellstar Northwest Georgia Oncology Centers (NGOC) between January 1, 2021, and January 1, 2023. Patient-reported episodes of CINV were compared between 94 total patients who received traditional triplet therapy plus olanzapine (study group) and those who received traditional triplet therapy alone (comparator group). Cycles administered to the study group before the initiation of olanzapine were recorded in the comparator group to mitigate potential under- or overestimation of efficacy and safety. Data collection was performed using the SlicerDicer reporting tool in the electronic health record EpicÒ.
Results: The addition of olanzapine to traditional triplet therapy significantly reduced the incidence of CINV in patients receiving HEC. Out of 159 cycles administered with olanzapine and 358 cycles without, a total of 10 (0.06 per cycle) and 242 (0.68 per cycle) episodes of CINV were documented, respectively. Additionally, HEC dose reductions secondary to CINV were significantly higher in the comparator group.
Conclusion: Limiting the need for dose modifications due to toxicities attributed to chemotherapy provide clinicians the opportunity to achieve recommended doses with evidence-based survival benefits. The reduction of CINV episodes associated with HEC in this study provides additional real-word evidence supporting the efficacy of olanzapine for managing CINV.
Keywords: olanzapine, chemotherapy, nausea, vomiting
ASCO, American society of clinical oncology; CINV, Chemotherapy-induced nausea and vomiting; EHR, Electronic health record; HEC, Highly emetic chemotherapy; NCCN, National comprehensive cancer network; NGOC, Northwest Georgia oncology centers; USD, United states dollar; VC, vomiting center
Chemotherapy-induced nausea and vomiting (CINV) involves the stimulation of serotonin (5-HT3), neurokinin (NK-1), dopamine (D1-4), and potentially several other receptors along gastrointestinal and central nervous system signaling pathways following chemotherapy administration and exposure to endogenous toxins released during cellular destruction.1,2 In turn, these signals are relayed to the vomiting center (VC) leading to the sensation of nausea and initiation of emesis.3 Variation in emetogenicity among chemotherapy agents and dosages influence the VC response and are categorized according to the frequency at which emesis has been observed.4,5 Anticancer regimens deemed highly emetic (frequency greater than 90%) often require multiple antiemetic agents and have traditionally been managed with a triple combination of neurokinin-1-receptor antagonists (NK1RA), 5-hydroxytryptamine type-3 receptor antagonists (5HT3RA), and dexamethasone to alleviate CINV.6,7
Olanzapine is an atypical antipsychotic that has seen increased utilization alongside triplet therapy due to increasing evidence supporting its efficacy in the prevention and treatment of CINV.8–14 In addition to 5HT3 and D1-4, olanzapine antagonizes histamine (H1), alpha-1 adrenergic, and muscarinic (M1-5) receptors which may also contribute to the emetic response from the VC; however, further research into the pathophysiology of CINV is necessary.15 Currently, both the National Comprehensive Cancer Network (NCCN) and American Society of Clinical Oncology (ASCO) recommend the use of olanzapine in combination with triplet therapy for highly emetic chemotherapy (HEC) regimens. The recommended doses and frequency are 5-10 milligrams (mg) orally on the day of chemotherapy and subsequently on cycle days 2-4.6,7
Despite recent studies supporting its use for CINV, alternative clinical strategies may exclude olanzapine and resort to chemotherapy dose reductions and modifications that deviate from efficacious regimens with evidence-based survival benefits. Although there is potential for quality-of-life improvements, it’s reasonable to consider the use of olanzapine over such strategies to maintain chemotherapy survival benefits and the potentially lower likelihood of adverse events due to its short duration of therapy. The purpose of this study was to evaluate the efficacy of olanzapine in reducing the severity and frequency of CINV secondary to HEC regimens and provide real-world evidence of its use in clinical practice.
This was a multicenter, retrospective, electronic chart review study of patients aged >18 years with a cancer diagnosis and receiving HEC as defined by the NCCN Antiemetic Version 1.2024 Guidelines Table 1.6 Data over a two-year period between January 1, 2021, and January 1, 2023, at Wellstar Northwest Georgia Oncology Centers (NGOC) were collected utilizing the SlicerDicer reporting tool incorporated in the centers’ electronic health record (EHR) EpicÒ. Episodes of CINV experienced from cycle 2 onwards in patients who received traditional triplet therapy combined with olanzapine were compared to patients who received traditional triplet therapy alone which will serve as the comparator group. Patients that were pregnant, lacked a cancer diagnosis, had received prior chemotherapy treatment(s), received non-HEC regimens, or prescribed antipsychotic medications (e.g., olanzapine) for indications other than CINV, were excluded from this study. CINV occurring greater than 24 hours after HEC administration were also excluded. Patient demographics including age, gender, race, clinical stage, weight, BMI, and number of HEC cycles were also collected.
|
HEC Regimens (emetic risk >90%) |
|
|
Anthracycline and cyclophosphamide (AC) combination regimens |
Epirubicin >90 mg/m2 |
|
Carboplatin AUC >4 |
Fam-trastuzumab deruxtecan-nxki |
|
Carmustine >250 mg/m2 |
Ifosfamide >2 g/m2 per dose |
|
Cisplatin |
Mechlorethamine |
|
Cyclophosphamide >1500 mg/m2 |
Melphalan >140 mg/m2 |
|
Dacarbazine |
Sacituzumab govitecan-hzly |
|
Doxorubicin >60 mg/m2 |
Streptozocin |
Table 1 HEC regimens
The primary outcome was chart-documented episodes of CINV. Patients’ communications to NGOC and providers’ follow-up notes within the EHR were analyzed for documentation of nausea and vomiting with and without the use of olanzapine. HEC cycles with patient-reported episodes of CINV prior to the initiation of olanzapine in the study group were included in the comparator group to mitigate the potential over- or underestimation of olanzapine’s efficacy and safety. The secondary outcome was HEC dose reductions observed between the two groups. Descriptive statistics were used to analyze patient characteristics and both the primary and secondary outcomes.
SlicerDicerÒ identified 265 patients who received HEC (olanzapine, n=128; comparator, n=137) within the study’s designated two-year period (Figure 1). A total of 47 patients in the olanzapine group met eligibility for inclusion and were evenly matched by the comparator group (n=47) following random selection. Indications other than the management of CINV were the most common reason for study exclusion in the olanzapine group. Patient characteristics are shown in Table 2.
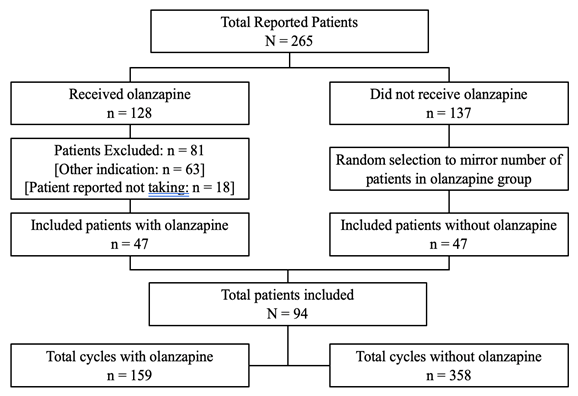
Figure 1 Patient selection.
Received olanzapine is defined as patients who received olanzapine for at least 1 cycle of their HEC regimen.
|
|
Triplet therapy + olanzapine |
Triplet therapy alone |
|
Age, y (IQR) |
58 (48-66) |
63 (53-71) |
|
Female, n (%) |
36 (76.5) |
35 (74.5) |
|
Weight, kg (IQR) |
72.7 (60-84) |
69.1 (61.8-77.7) |
|
BMI, kg/m2 (IQR) |
26.5 (21.4-32.0) |
23.7 (22.0-28.5) |
|
Race, n (%) |
||
|
Caucasian |
34 (81.0) |
32 (68.1) |
|
African American |
8 (17.0) |
11 (23.4) |
|
Other |
5 (2.0) |
4 (8.5) |
|
Clinical Stage, n (%) |
||
|
I |
10 (21.3) |
5 (10.6) |
|
II |
17 (36.2) |
9 (19.1) |
|
III |
8 (17.0) |
11 (23.4) |
|
IV |
12 (25.5) |
22 (46.9) |
|
Cycles, n (%) |
159 (57) |
238 |
|
Prior to olanzapine |
120 (43) |
- |
Table 2 Patient characteristics
The median age of the patients in the olanzapine group and comparator group was 58 and 63 years, respectively. Patients in both the olanzapine and comparator group received a median of 5 HEC cycles and were predominantly Caucasian females (81% and 68.1%, respectively). Stage II disease (36.2%) in the olanzapine group and stage IV disease (46.9%) in the comparator group were the most common clinical stages.
During this study, patients receiving HEC and traditional triplet therapy prior to the initiation of olanzapine experienced 91 CINV events over the course of 120 cycles and patients receiving traditional triplet therapy alone experienced 151 CINV events over 238 cycles, totaling 358 cycles without olanzapine (mean = 0.68 per cycle) Table 3. Following the addition of olanzapine to triplet therapy in the study group, 10 episodes of CINV were documented over the course of 159 total cycles (mean = 0.06 per cycle). The incidence of CINV is illustrated in (Figure 2). One adverse event was documented, occurring in an individual who reported experiencing syncope; however, the patient did not experience CINV but wished to discontinue therapy.
|
Outcome |
Triplet therapy + olanzapine |
Triplet therapy alone |
||
|
Prior to olanzapine |
Comparator group |
Total without olanzapine |
||
|
Total Cycles (n) |
159 |
120 |
238 |
358 |
|
CINV events |
||||
|
Total |
10 |
91 |
151 |
242 |
|
Per patient |
0.21 |
1.94 |
3.21 |
2.57 |
|
Per cycle |
0.06 |
0.76 |
0.63 |
0.68 |
Table 3 Primary outcome: patient-reported CINV
Note: Cycles omitting olanzapine in the study group were combined with cycles in the comparator group
Implemented chemotherapy dose reductions were secondary to CINV and weight-based reductions were omitted. A total of 130 dose reductions were administered to individuals receiving triplet therapy alone compared to 16 dose reductions when providers utilized olanzapine for the management of CINV. Cycles without and with olanzapine averaged 0.36 and 0.10 dose reductions respectively per cycle Table 4.
|
Outcome |
Triplet therapy + olanzapine |
Triplet therapy alone |
||
|
Prior to olanzapine |
Comparator group |
Total without olanzapine |
||
|
Total (n) |
16 |
51 |
79 |
130 |
|
Mean (%) |
10 |
43 |
33 |
36 |
Table 4 Secondary outcome: cycles with dose reductions
Note: Cycles omitting olanzapine in the study group were combined with cycles in the comparator group.
In this observational study, the addition of olanzapine to triplet therapy was compared to triplet therapy alone to assess its effectiveness in reducing CINV among patients receiving HEC. Our results illustrate a clinically significant reduction in the incidence of patient-reported episodes of CINV (olanzapine: 6.3%; comparator: 67.7%) and provide additional real-world evidence supporting the use of olanzapine. One adverse event was documented in the olanzapine group, involving a patient who experienced syncope. A similar retrospective study evaluating patients who failed traditional triplet therapy and subsequently were administered olanzapine observed similar findings. Slimano et al compared the frequency and severity of CINV between patients who received olanzapine plus traditional triplet therapy in the second of two observed cycles versus those who did not.16 Regimens predominantly consisted of HEC (olanzapine: 92%; comparator: 96%); however, 36% of patients included in this study had received prior chemotherapy. Olanzapine plus triplet therapy was found to significantly reduce episodes of CINV compared to triple therapy alone (48% vs. 4%, respectively; p<0.034). The p-value was shown to be statistically significant (p<0.05) which was done using the SPSS (statistical package for social sciences) program.
Chemotherapy-related toxicities pose various challenges for patients; however, employing effective supportive care strategies could substantially improve both tolerability and adherence to anticancer regimens. In turn, the need for dose modifications and/or reductions could be minimized, providing clinicians the opportunity to proceed with evidence-based regimens that demonstrate higher survival benefits. Additionally, mitigating chemotherapy complications could lower healthcare-associated costs potentially impacting patients’ quality of life. Although a combination of four antiemetics (NK1RA, 5-HT3RA, olanzapine, and dexamethasone) is the preferred treatment strategy for preventing acute and delayed CINV secondary to HEC, NCCN Antiemesis Guidelines also recommend two additional treatment options which exclude an NK1RA and olanzapine, respectively.6 As summarized by Gyawali et al, the affordability of antiemetics commonly used to prevent CINV can be financially daunting, especially in resource-limited demographics. The findings observed in their literature analysis found olanzapine reduced costs by approximately $100-500 (USD); however, costs may vary since the time of publication.17 Further, both Gyawali and colleagues and NCCN guidelines provide evidence supporting the efficacy of a three-drug regimen (olanzapine, 5-HT3RA, and dexamethasone) to combat CINV, offering a cost-saving alternative for patients unable to afford an NK1RA.
Although the financial cost of olanzapine is substantially less than an NK1RA and has evidence supporting its effectiveness in CINV, the stigma associated with the term “antipsychotic” could potentially dissuade patients from taking it. Among the 18 patients excluded in the olanzapine group, the two reasons often documented in the patients’ EHR conversations were the concerns of taking multiple antiemetic medications and refusing to take an antipsychotic. These concerns underscore the impact and value pharmacists provide to patient care. Through educating patients on the risks and benefits associated with olanzapine and clarifying its mechanisms in relation to the pathophysiology of CINV, pharmacists have the potential to reduce the stigma associated with its classification, thereby enhancing adherence and patient outcomes. Further, pharmacists can provide cost-saving strategies to help alleviate the financial burden associated with combination antiemetic therapy.
The retrospective nature of this study inherently imposes several limitations. This study had a small sample size and relied on patients notifying providers if they experienced CINV. Confounding variables that potentially influenced the incidence of CINV were unable to be assessed as patients were administered chemotherapy in outpatient infusion clinics. Additionally, regimens were documented as a binary variable (HEC/not HEC) in the inclusion criteria; however, the identification and evaluation of each regimen would have provided additional benefit to this study. The study also lack randomization which may have introduced selection bias and it was conducted in a single geographic region which limits external validity.
The addition of olanzapine to traditional triplet therapy resulted in a clinically significant reduction in the incidence of CINV versus triplet therapy alone. Although our results demonstrate a real-world scenario, the numerous limitations associated with this study hinder its external validity. Additionally, patients refusing treatment with olanzapine due to its classification of an antipsychotic, highlights the opportunity for pharmacists to provide education as a means to improve patient outcomes.
None.
The authors declare that they have no conflicts of interest.

©2024 Powell, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.
2 7