Advances in
eISSN: 2572-8490


Review Article Volume 9 Issue 1
Correspondence: Dr. Bill Tawil, Department of Bioengineering, UCLA School of Engineering, Los Angeles, USA
Received: October 25, 2023 | Published: November 6, 2023
Citation: Zoe L, Bill T. Epilepsy: physiology, diseases, treatments, products, and market analysis. Adv Tissue Eng Regen Med Open Access. 2023;9(1):50-58. DOI: 10.15406/atroa.2023.09.00141
Epilepsy, a complex neurological disorder characterized by recurrent seizures, poses significant challenges to patients and healthcare providers alike.1 Pharmacotherapy remains a cornerstone of epilepsy treatment, and the paper explores the latest advancements in antiepileptic drugs (AEDs), highlighting novel agents and their mechanisms of action.2 Additionally, it discusses the challenges posed by drug-resistant epilepsy and explores alternative treatment options, such as neurostimulation and neuromodulation devices. Innovative techniques, such as deep brain stimulation (DBS) and vagus nerve stimulation (VNS) are reviewed in detail, emphasizing their potential as adjunctive therapies for seizure control. Surgical interventions, including resective surgery, laser interstitial thermal therapy (LITT), and functional hemispherectomy, are discussed as viable options for patients with medication-resistant epilepsy.3 This review aims to provide an encompassing exploration of types and characteristics of treatment products, and an in-depth analysis of the market, including market size, distribution, and current trends. Additionally, it will conclude by presenting an overview of emerging approaches in epilepsy treatment for potential clinical applications.
Keywords: epilepsy, seizures, anti-epileptic drugs, drug-resistant epilepsy, neuromodulation, surgery, treatment
CNS, central nervous system; AEDs, antiepileptic drugs; DBS, deep brain stimulation; VNS, vagus nerve stimulation; LITT, laser interstitial thermal therapy; MRI, magnetic resonance imaging; AE, autoimmune epilepsy
Epilepsy, a neurological disorder characterized by recurrent seizures, can affect individuals of all ages.1 While it is considered a chronic condition, approximately 70% of people can effectively manage their seizures through the use of medications and/or surgical interventions.2–4 However, it is important to acknowledge that there is still a significant population of over one million individuals in the United States alone who continue to experience uncontrolled epilepsy, necessitating continued research and the development of additional treatment options.1 Ongoing investigations into epilepsy treatment have led to the exploration of additional options, such as neuromodulation devices, which aim to disrupt abnormal brain activity before the onset of a seizure. Furthermore, there remains a significant research gap in understanding the origins of seizures, predicting their occurrence, preventing them, and elucidating the role of genetics in epilepsy.4
Comprising an intricately structured network of over 100 billion information-processing cells known as neurons, the human brain represents a remarkably sophisticated organ that governs a multitude of vital functions, including task-evoked responses, motor coordination, sensory perception, emotional processing, language, communication, cognitive processes, and memory (Figure 1A).5,6 Grey matter, constituting the predominant portion of the central nervous system (CNS), manifests as stratified ensembles of neurons.5 Within neuronal architecture, the axon fulfills the pivotal role of fostering the uninterrupted propagation of electrical impulses amidst neurons (Figure 1B).7 Glial cells undertake crucial functions that revolve around providing support and establishing an environment conducive for the optimal functioning of neurons.5 These roles encompass the maintenance of the ionic balance within neurons, governing the speed of nerve signal transmission and synaptic activity through the regulation of neurotransmitter uptake, serving as a platform for certain facets of neural development, and aiding in the recuperation process following neural damage.5

Figure 1 Anatomical depiction of the brain6 (A) and basic neuron design7 (B) illustrating the structure and organization of healthy brain tissue.
In (A), various regions of the brain are delineated, showcasing the complexity of neural connections and functional specialization. The essential components of a neuron, including the cell body, dendrites, and axon, are illustrated in (B), highlighting their structure and fundamental role in information processing and transmission within the nervous system to understand the complex architecture and functionality of the brain and its constituent neurons.
The typical function of neurons involves generating both electrical and chemical signals that interact with other neurons, glands, and muscles, ultimately giving rise to human thoughts, emotions, and behaviors. During a seizure, numerous neurons simultaneously fire at an accelerated rate, up to 500 times per second, which is significantly faster than usual. This excessive electrical activity and temporary disruption of normal neuronal function simultaneously leads to involuntary movements, sensations, emotions, and behaviors and can result in a loss of awareness.8
The nervous system relies on the balance of chemicals and electrical gradients within its surroundings, known as the ionic milieu, to function properly. Consequently, the nervous system employs various mechanisms to regulate the ionic environment, which significantly influence its excitability. One notable example is the electrical foundation of resting membrane potential that is typically established to prevent neurons from constantly firing, yet it remains sufficiently close to the threshold to allow for the possibility of discharging when necessary, as action potential generation is vital for CNS functioning. The precise control of resting potential becomes crucial in order to prevent excessive discharge, a characteristic often associated with seizures.9
Epilepsy exhibits a multifaceted etiology that varies from person to person. While the exact causes of epilepsy may elude identification in some individuals, there are discernible factors associated with its onset. Genetic disorders can be implicated, as certain inherited traits or mutations in specific genes may contribute to an increased susceptibility to seizures. Additionally, instances of epilepsy can be linked to a history of brain trauma, such as head injuries resulting from accidents or concussions. Autoimmune diseases, characterized by an aberrant immune response against the body's own tissues, can also be correlated with the development of epilepsy. Furthermore, metabolic issues, encompassing disruptions in the body's biochemical processes, have been identified as potential triggers for seizures. Infections, both viral and bacterial in nature, have been known to provoke epileptic seizures in some individuals.10
It is noteworthy that while these various factors have been recognized as potential contributors to epilepsy, the precise interplay between these causal elements and the manifestation of the disorder can vary greatly from person to person.10
Infection stands as the prevailing cause of epilepsy on a global scale, particularly prevalent in regions of the world characterized by developing socioeconomic conditions.11
Autoimmune epilepsy (AE) arises from an alteration in the immune system's functioning. The precise frequency of autoimmune epilepsy occurrences remains elusive, though estimates suggest that approximately 5% to 35% of individuals experiencing new onset seizures may have an autoimmune etiology, equating to 1 to 7 out of 20 cases.12 In autoimmune epilepsy, seizures occur frequently and may happen multiple times a day with focal seizures being the most common type, and the presence of seizures affecting the face and arm on the same side of the body, strongly suggests autoimmune epilepsy. Seizure medications are often ineffective in controlling these seizures. Additionally, individuals with autoimmune epilepsy may experience difficulties with memory, personality changes, and fluctuations in mood or behavior, such as paranoia, depression, or hallucinations.12
Genetic causes of epilepsy are identified when seizures can be attributed to known or presumed genetic defects.13 Certain genetic pathogenic variations, involving alterations in genes, may arise spontaneously in a child without being inherited from either parent.13 Metabolic epilepsy predominantly manifests in infancy or early childhood, with seizures being a common symptom.14 These seizures are often accompanied by additional indicators, including delayed development or skill loss, abnormal movements, and potential impairments in the functioning of other organs such as the liver or kidneys.14 Structural causes of epilepsy are when there is a noticeable abnormality in the brain that significantly elevates the likelihood of seizures. These structural causes are often detectable through brain imaging techniques, particularly magnetic resonance imaging, which can reveal the presence of such abnormalities. Structural causes of epilepsy are often accompanied by additional symptoms that reflect abnormalities in specific areas of the brain that can include motor difficulties in one limb or one side of the body, speech impairments, and visual disturbances on one side.15
Epilepsy causes can also vary depending on age. Table 1 indicates common causes of epilepsy by life stage.10
|
Newborns |
Infants and children |
Children and adults |
Older Adults |
|
- Insufficient oxygen supply during the birth |
- Fever |
- Congenital abnormalities such as Down syndrome, Angelman syndrome, tuberous sclerosis, and neurofibromatosis |
- Disruption of blood supply to the brain, also known as stroke |
|
- Brain malformations altering structure |
- Infectious agents, viral or bacterial pathogens |
- Genetic predisposition |
- Degenerative neurological disorder like Alzheimer's disease |
|
- Hypoglycemia, hypocalcemia, hypomagnesemia, or other electrolyte abnormalities |
- Brain tumors and abnormal growths |
- Head trauma |
- Head trauma |
|
- Intrinsic metabolic disorders present at birth |
- Progressive neurodegenerative disorders |
||
|
- Intracranial hemorrhages |
|||
|
- Maternal drug use |
|
|
|
Table 1 Common causes of epilepsy by age,10 encompassing the distinct contributing factors observed in different age groups, including newborns, infants and children, children and adults, and older adults
Nearly all epilepsy patients will experience a comorbid condition during their treatment journey.16 Comorbid conditions with epilepsy can be medical, cognitive, or psychiatric with the five most common epilepsy comorbid conditions being migraine, stroke, depression, attention-deficit hyperactivity disorder, and osteoporosis and fractures. The frequent occurrence of comorbid health conditions among individuals with epilepsy and proposes three potential explanations for this association. Epilepsy, including its treatment, may cause the comorbid condition or the comorbid condition, along with its treatment, could lead to the development of epilepsy. The third explanation suggests that a common underlying mechanism might be responsible for both epilepsy and the comorbid condition co-occurring. It is unlikely that a single explanation can account for all comorbid conditions in epilepsy, but understanding the reasons behind the association between epilepsy and comorbid conditions is crucial for effective diagnosis and management.
Individuals with epilepsy often experience bone health issues, such as low bone mineral density and osteoporosis. Prolonged use of AEDs, especially among women, is frequently linked to these problems.17 Notably, epilepsy patients face a higher risk of bone fractures compared to the general population, with factors including reduced bone density and an increased susceptibility to fractures due to seizures. It is also well documented that strokes can lead to epilepsy, particularly among older individuals. It is frequently observed that individuals with epilepsy often possess several risk factors associated with strokes even before experiencing the stroke itself. Among psychiatric comorbidities in epilepsy, interictal depression is the most prevalent and occurs more frequently compared to other neurological conditions and chronic non-neurological conditions. Recent empirical evidence suggests that major depression could serve as a risk factor for the development of epilepsy and there may exist a common underlying pathophysiological mechanism that forms the basis for both conditions. There also may be shared genetic and physiological mechanisms between epilepsy and certain types of migraine, including brain hyper-excitability originating from cortical depression and abnormalities in calcium signaling. Furthermore, specific subtypes of epilepsy and migraine have been associated with genetic mutations in SCN1A, CACNA1A, and ATP1A2 genes located on chromosome 17, suggesting a potential genetic susceptibility and common underlying factors contributing to the development of these conditions. Common biological mechanisms proposed for the co-occurrence of epilepsy and attention-deficit hyperactivity disorder (ADHD) include the disruption of lipid metabolism, the norepinephrine system, or the dopamine transporter system. This is supported by evidence from national surveys, population-based studies, and data from tertiary-care centers. Each patient's unique combination of seizure etiology and comorbidities requires a comprehensive understanding of their specific condition to determine the most effective treatment strategies. This emphasizes the importance of ongoing research efforts aimed at unraveling the complex mechanisms underlying seizures and associated comorbidities, ultimately leading to improved personalized treatments for individuals with epilepsy.17
Epilepsy is one of the most common neurological disorders affecting around 50 million people in the world, with 2.4 million new cases each year.18 While Sudden Unexpected Death in Epilepsy (SUDEP) is a rare occurrence, it still impacts approximately 1 in 1,000 individuals with seizures, and its precise cause remains unknown and it claims around 3,000 deaths per year in the United States.19
The global epilepsy market is primarily driven by factors such as the rising incidence of accidents, brain injuries, and the elevated risk of post-traumatic seizures among patients with brain injuries.18 Anticipated consumer demand is set to rise due to the heightened vulnerability of elderly patients with epilepsy, who exhibit a greater sensitivity to risks associated with the condition.20 Drug resistant epilepsy is expected to be valued even higher at 2.93 billion USD at the end of the forecast period.18 The global demand for epilepsy-related products and treatments is influenced by multiple factors, including the introduction of new drugs in developed regions, the prevalence of concurrent neurological disorders, and the rise in disposable income levels.20 These factors collectively contribute to the dynamics of the epilepsy market on a global scale (Figure 2).21

Figure 2 Epilepsy drugs market size by region, 2018-2030.21
This comparative assessment includes an examination of key geographical regions, namely North America, Europe, Latin America, Asia Pacific, and the Middle East & Africa. By exploring the market dynamics and trends across these diverse regions, valuable insights can be gained regarding the growth and market potential of epilepsy drugs in each specific area over the specified timeframe.
Epilepsy has significant costs on both individuals and the healthcare system. Based on data from 2014, it was found that epilepsy or convulsion diagnoses resulted in over 1 million visits to emergency departments and 280,000 hospital admissions. Patients with epilepsy had an average hospital stay of 3.6 days, contributing to aggregate hospital costs totaling around $2.5 billion.22 The total direct healthcare costs per person in the general epilepsy population, covering all clinically defined subgroups, exhibited a range of $10,192 to $47,862.23 However, epilepsy treatment is not created equal across all countries. In Latin America and the Caribbean, the treatment gap for epilepsy is estimated to exceed 50%, revealing that over half of the individuals affected by this disease in the region do not have access to essential healthcare services.24
The epilepsy market is estimated to be worth 590.2 million by 2030, growing at a compound annual growth rate of 7.20% from 2022 to 2030.20 The regions with the highest prevalence of epilepsy were identified as eastern, western, and southern sub-Saharan Africa, central Asia, central and Andean Latin America, and southeast Asia (Figure 3).25
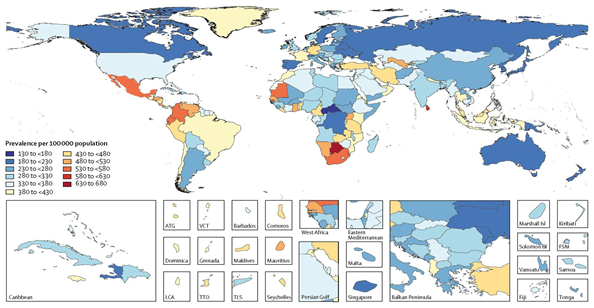
Figure 3 Age-standardized prevalence per 100,000 of idiopathic (unknown, spontaneous) epilepsy for both sexes in the year 2016,25 including specific country abbreviations such as ATG (Antigua and Barbuda), FSM (Federated States of Micronesia), Isl (Islands), LCA (Saint Lucia), TLS (Timor-Leste), TTO (Trinidad and Tobago), and VCT (Saint Vincent and the Grenadines).
The prevalence of epilepsy demonstrated an age-related increase, with peaks observed among individuals aged 5-9 years and those older than 80 years (Figure 4). However, there were no significant differences in prevalence between men and women (Figure 4).25
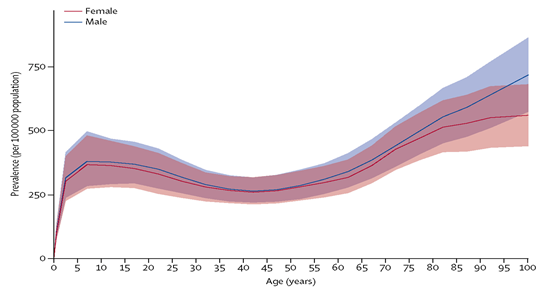
Figure 4 Global prevalence of idiopathic epilepsy by age and sex in the year 2016.25 with 95% uncertainty intervals shown in the shaded regions.
While epilepsy is not associated with high mortality rates, it is essential to acknowledge the presence of comorbidity and its significant impact. In 2017, only 38.2% of deaths among individuals with epilepsy were directly attributed to the condition itself, with the remaining 61.8% of deaths resulting from other leading causes.26 This highlights the importance of considering and addressing the broader health issues that can coexist with epilepsy and contribute to overall mortality, particularly vascular dementia and Alzheimer’s. The main factors influencing the increase in epilepsy mortality in the United States is the change in the underlying causes, number of deaths due to all neurological disorders, and increased epilepsy prevalence (Figure 5).26

Figure 5 Trends in age-adjusted mortality rates for epilepsy from 1999 to 2017, highlighting a significant increase in the slope from 2010 to 2017 compared to the period of 1999 to 2009 (based on a weighted linear regression model with piecewise linear splines, p=0.001).26
The current approach to epilepsy treatment lacks a comprehensive understanding of its underlying cause and primarily focuses on providing symptomatic relief through interventions such as medication, surgery, and the use of implanted devices.18 The limited treatment options and incomplete understanding of epilepsy create significant opportunities for development and further research in the field.18
Major companies in the global epilepsy market include LivaNova PLC, GlaxoSmithKline PLC, Eisai Co. Ltd., Pfizer Inc., Medtronic, UCB S.A., NeuroPace, Inc., Novartis AG, and Abbott Laboratories.20
AEDs are the most common form of treatment for epilepsy, helping to control seizures in about 70% of people.27 While commonly referred to as antiepileptics, it is important to note that there is currently no cure for epilepsy.2 AEDs are classified by generation to indicate when they were developed and introduced.28 The majority of AEDs regularly used are second and third generation, meaning they were developed and brought to market after 1989 (Figure 6).29

Figure 6 Global epilepsy drugs market by treatment, spanning 2013 to 2024, represented in USD millions.29
While these medications are crucial for individuals with epilepsy, it is important to note that they do not provide complete seizure control for approximately 30% of patients.27 This treatment gap has sparked interest in exploring alternative options such as surgical interventions or the use of devices to address the uncontrolled seizures. Epilepsy management extends beyond pharmaceutical interventions, as non-drug therapies have proven to be effective for drug-resistant epilepsy. These alternative treatment options encompass surgical procedures, neurostimulation devices, and dietary therapy.30
Epilepsy surgery entails the removal of a specific area of the brain where seizures originate. Given the inherent risks associated with surgery, it is typically not the initial treatment choice for individuals with epilepsy. However, it can be highly effective for those whose seizures consistently manifest in a specific area of the brain, focal epilepsy.3 Around 50-70% of people experience rare or no disabling seizures. The risks involved with invasive surgery are applicable to epilepsy surgery as well.31 These risks can include bleeding, infection, brain tissue injury, and delayed healing. Risks associated with epilepsy surgery and the removal of brain areas include memory problems, vision distortion or loss, mood problems, speech difficulties, or motor skills deficits.31
Neurostimulation devices try to control seizures with stimulation. These do involve surgery, but it is considered less invasive because no brain area is removed.31 Examples of neurostimulation include deep brain stimulation, responsive neurostimulation, and vagus nerve stimulation. The North American VNS market size is trending upwards as a treatment method (Figure 7).32

Figure 7 Market size of Vagus Nerve Stimulation in North America.32
The ketogenic diet is an option for patients whose epilepsy does not respond to medication.33 Although the mechanisms for why the ketogenic diet works remains unknown, it has been used since the 1920’s to reduce seizures. It is believed that the low carbohydrate and high fat diet increases the ketones produced, altering brain activity and reducing the tendency for seizures.34
A summary of the market segments and trends for treatment is summarized below in Table 2.30,32–36
|
Treatment |
Method |
Advantages |
Limitations |
|
Anti-epileptic drugs |
- Altering electrical activity in neurons by affecting ion channels |
- Commonly used |
- Can have severe side effects |
|
- Altering chemical transmission between neurons by affecting neurotransmitters |
- Effective for the majority of patients |
- Does not work for 30% of people with epilepsy |
|
|
Surgery |
- Removal of the brain area where seizures are originating |
- Highly effective: 50-70% are free from seizure or only have them rarely |
- Not suitable for all epilepsy patients |
|
- Decrease seizure medications and subsequent side effects |
- General surgery risks |
||
|
- Lower cost of living with epilepsy |
- Not adjustable |
||
|
- Improved quality of life |
|||
|
Neurostimulation devices |
- Chronic electrical stimulation to specific regions of the brain |
- Shorter procedure time |
- General surgery risks |
|
- Less recovery time |
|||
|
- Decreased tissue injury |
|||
|
Ketogenic diet |
- Use of fat for energy instead of carbohydrates increases ketones |
- No age restriction |
- Not suitable for all patients |
|
- Effective, able to reduce medication |
- Requires specialized care and supervision |
||
|
- Can have other benefits such as increased alertness |
- Short and long term side effects |
||
|
|
|
|
- Can be challenging to maintain long term |
Non-invasive
Seizures manifest when there is a disruption in the delicate balance of excitatory and inhibitory circuits within the brain. This imbalance leads to abnormal and excessive electrical activity, resulting in the characteristic symptoms and manifestations of a seizure episode.36 The purpose of many anti-epileptic drugs is to suppress seizures through acting on diverse molecular targets to selectively modify the excitability of neurons so seizure-related firing is inhibited while still preserving normal neuronal activity.37 They can either increase or decrease excitation through alteration of ion channels or neurotransmitters.36 Certain antiepileptic drugs exert their therapeutic effects by interacting with a combination of ion channels or through mechanisms of action that are not yet fully understood (Figure 8).38 A summary of common AEDs and their mechanisms of action is summarized in Table 3.39–41

Figure 8 Categorization of antiepileptic drugs based on their primary mechanism of action.38
|
Drug |
Company |
Mechanism of action |
FDA approval |
|
Carbamazepine (Tegretol, Carbatrol) |
Novartis Corporation |
Inhibits sodium channels and rapid firing of brain cells |
1968 |
|
Lacosamide (Vimpat) |
Harris FRC Corporation |
Blocks sodium channels and reduces brain excitability |
2009 |
|
Lamotrigine (Lamictal) |
GlaxoSmithKline |
Lowering release of chemicals like glutamate by inhibiting voltage dependent sodium channels |
1994 |
|
Levetiracetam (Spritam) |
Aprecia Pharmaceuticals Company |
Neurotransmitter release modulation through synaptic vesicle protein SV2A binding |
2015 |
|
Phenytoin (Dilantin) |
Mylan Pharmaceuticals |
Blocks sodium channels and reduces brain excitability |
1998 |
|
Pregabalin (Lyrica) |
Pfizer Inc. |
Likely influences transport of GABA and affect calcium channels |
2004 |
|
Vigabatrin (Sabril) |
Lundbeck |
Blocks metabolism of the brain’s main inhibitory neurotransmitter GABA |
2009 |
Table 3 Commonly used anti-epileptic drugs39–41
Invasive
Epilepsy surgery is a highly invasive option usually only considered when a patient is not experiencing controlled seizures on medication. Surgery presents a viable option for reducing seizure-related mortality rates and minimizing the need for long-term use of AEDs and their associated side effects. By surgically addressing the underlying causes of epilepsy, such as removing or disconnecting the seizure focus or abnormal brain tissue, it is possible to significantly reduce the frequency and intensity of seizures. This surgical intervention offers the potential to provide long-term seizure control, leading to improved quality of life and decreased reliance on AEDs.3 The decision to pursue surgery is made after careful evaluation of individual factors and consideration of potential risks and benefits.30 Surgical interventions, when appropriate, can be instrumental in reducing seizure-related deaths and improving the overall management of epilepsy. There are many types of epilepsy surgery that depend on the location of the neurons.3
The most frequently performed surgical procedure for epilepsy is resective surgery, which entails the removal of a small section of the brain. This surgical approach is commonly utilized in cases where the seizure focus is localized, and it is particularly prevalent in surgeries involving the temporal lobes. Laser interstitial thermal therapy is a less invasive procedure that employs a magnetic resonance imaging (MRI)-guided laser to precisely target and destroy a specific area of brain tissue. Corpus callosotomy involves the complete or partial removal of the corpus callosum, the structure that connects the nerve fibers between the right and left hemispheres of the brain. This surgical intervention is primarily employed in cases where there is a risk of abnormal brain activity spreading from one hemisphere to the other, particularly in children with severe and medication-resistant epilepsy. By severing the connections provided by the corpus callosum, corpus callosotomy aims to restrict the propagation of seizures between the hemispheres, reducing the frequency and severity of generalized seizures. A relative severe surgical procedure is hemispherectomy is a surgical procedure that involves the removal or disconnection of one hemisphere of the brain, specifically the cerebral cortex. This procedure is selectively performed in children who experience seizures originating from multiple sites within a single hemisphere of the brain. Functional hemispherectomy is a surgical procedure performed in children that involves the disconnection of nerve connections between brain hemispheres without removing brain tissue.3
Neuroengineering is a field that interfaces with the nervous system to gain insights, restore, substitute, or augment the functionalities of neural systems.42 This burgeoning discipline has sparked considerable enthusiasm due to its untapped potential in treating neurological disorders like epilepsy.43 Deep brain stimulation (DBS) is a well-established therapeutic approach in the field of neuromodulation.44 It involves the application of chronic electrical stimulation to specific regions of the brain, with the aim of treating epilepsy and similar conditions.35 DBS includes implantation of electrodes into the brain and a pacemaker-like device in your chest to control the electrical stimulation.45 The device is permanently placed in the brain to release timed electrical signals.3
A relatively new smart device called RNS® System uses responsive neurostimulation to treat epilepsy.46 RNS™ Neurostimulator System is an implantable closed loop neurostimulator system by Neuropace Inc to prevent seizures.47 It monitors brainwaves, detects unusual activity that may lead to a seizure, and responds by sending brief pulses to disrupt this unusual activity. Patients experience significant seizure reduction and lower SUDEP rate.46
Vagus nerve stimulation (VNS) is another form of implantable device that provides electrical stimulation to the brain.48 Although it is relatively unknown how VNS therapy works, it is believed to increase brain blood flow, increase neurotransmitters, and alter EEG patterns.48 The newest generation device SenTivaTM by LivaNova provides stimulation in response to rapid increases in heart rate associated with seizures.49 Table 4 summarizes various implantable device types and products for epilepsy.46,49–57
|
|
Product |
Company |
Composition |
FDA approval |
|
Deep Brain Stimulation |
Abbott InfinityTM DBS Neurostimulation System |
Abbott Medical |
Delivers low-intensity electrical pulses to nerve centers in the brain |
2020 |
|
Percept™ PC with BrainSense™ technology |
Medtronic |
Senses and records an individual’s unique brain signals for personalized DBS and direct stimulation |
2020 |
|
|
Medtronic ActivaTM |
Medtronic |
Rechargeable and non-rechargeable battery |
2002 |
|
|
Electrical stimulation of the brain nerve centers |
||||
|
Vercise Genus™ Deep Brain Stimulation (DBS) System |
Boston Scientific |
Approved for use in MRI environment and Bluetooth rechargeable and non-rechargeable, implantable pulse generators |
2021 |
|
|
Responsive Neurostimulation |
RNS® System |
Neuropace |
Monitors brain waves, detects unusual activity, responds with electrical stimulation, records and reports data |
2013 |
|
Vagus Nerve Stimulation |
SenTivaTM VNS Therapy® |
LivaNova |
Closed loop implantable and programmable pulse generator that responds to heart rate increases and collects data |
2017 |
There are currently a variety of pipeline products for epilepsy treatment with new diagnostic tools, therapeutic devices, and drugs in development.58 CODA Biotherapeutics, Inc. is using a chemogenetic platform to treat prevalent neurological disorders. Their engineered receptor and small molecule activator drug have presented promising preclinical data, demonstrating a significant reduction in focal seizure frequency in vivo.49 The mechanism underlying this approach involves the use of adeno-associated vector (AAV) to deliver a newly engineered inhibitory ion channel. This channel is designed to be activated by binding to a small molecule drug that can be taken orally and easily absorbed by the body. By harnessing the combination of AAV delivery and an orally bioavailable small molecule drug, this approach seeks to precisely regulate hyperactive brain cell firing and effectively suppress seizures, while minimizing the occurrence of adverse effects.59
For genetic epilepsy, there is currently a revolutionary gene therapy approach by Stoke Therapeutics, Inc. Their new medicine is undergoing clinical development that aims to address the underlying causes of genetic epilepsy like Dravet syndrome. They have had high success in patients that have already taken multiple standard AEDs, which has not been seen before. So far, the results have been 55% median reduction in convulsive seizure frequency, reduction in convulsive seizure frequency in 83% of patients, and over 50% reduction in two thirds of patients. This approach is unique because it could be the first disease-modifying therapy to address the genetic source of epilepsy by upregulating protein expression of functional SCN1A gene to reduce occurrence of seizures and non-seizure comorbidities.60
Although not necessarily a treatment device, Signant Health introduced today a novel electronic diary (eDiary) data collection for patients participating in epilepsy trials. This is particularly beneficial compared to traditional data collection because it has benefits of electronic data collection and standardization of measurement in the assessment of seizure events.61
Cognitive neural prosthetics (CNP) could be valuable in treatment for epilepsy due to the neural stimulation (Figure 9A). It records cognitive state, rather than strictly signals related to motor execution or sensation, which can then be used to stimulate assistive devices. Although this is not yet a clinical reality, it does hold promise for treatment of similar neurological disorders.62
Non-Invasive Vagus Nerve Stimulation (nVNS) with gammaCore VET™ is an investigational treatment that shows promise as an additional therapy for individuals with medically refractory seizures (Figure 9B). Although it has only been tested in canines so far, but this innovative approach involves the stimulation of the vagus nerve without the need for invasive procedures. The gammaCore VET™ device delivers gentle electrical impulses to the vagus nerve through the skin, targeting the nerve pathways associated with seizure activity. By modulating the activity of the vagus nerve, nVNS aims to reduce the frequency and severity of seizures in individuals who have not responded to traditional treatments alone. This non-invasive method offers a potential alternative for those who may not be suitable candidates for more invasive procedures or who seek additional options to manage their refractory seizures. Ongoing research and clinical trials are being conducted to further explore the effectiveness and safety of nVNS with gammaCore VET™ as an adjunct therapy for refractory seizures.63
Magnetic resonance-guided laser interstitial thermal therapy (MR-gLiTT) represents a pioneering and minimally invasive treatment modality for individuals with drug-resistant focal epilepsy and brain tumors. This innovative approach utilizes the power of magnetic resonance imaging (MRI) to guide a laser system that delivers controlled thermal energy to target areas in the brain. By precisely targeting and heating the affected tissue, MR-gLiTT aims to ablate or destroy the epileptic focus or tumor, thereby reducing seizure frequency or facilitating tumor shrinkage. This minimally invasive technique offers several advantages, including improved precision, reduced risk of complications, shorter hospital stays, and faster recovery times compared to traditional open surgical procedures. MR-gLiTT holds significant potential as an effective and well-tolerated treatment option for individuals facing drug-resistant focal epilepsy and brain tumors, providing renewed hope for improved seizure control and tumor management. Ongoing research and clinical studies are further exploring the efficacy and long-term outcomes of MR-gLiTT in these patient populations.64
Among noninvasive neurostimulation techniques, transcranial direct current stimulation (tDCS) is currently being investigated as a potential treatment for epilepsy.65 tDCS stands out as particularly well-suited for widespread distribution and home-based treatment due to its lightweight and portable nature, affordability, and overall favorable safety profile. Transcranial direct current stimulation (tDCS) involves the application of low-intensity electrical currents to specific regions of the brain, modulating neural activity and potentially offering therapeutic benefits. The portability and affordability of tDCS devices enable broader access to treatment, potentially reaching a larger population in need. Numerous studies on tDCS have consistently shown the technique to be well tolerated and safe. The application of direct electrical currents for stimulation is currently FDA-approved for extracranial use, and ongoing FDA applications are underway for the utilization of tDCS in the management of mood disorders and chronic pain. Ongoing research is exploring the efficacy and safety of tDCS as an adjunctive therapy for seizure control in individuals with epilepsy. The ability of tDCS to directly target and modulate cortical excitability holds promise for improving the management of epilepsy and providing an alternative or complementary treatment option to existing approaches.65
To advance the development of epilepsy treatments, it is essential to gain a comprehensive understanding of the underlying mechanisms of the condition. This area of research, known as epileptogenesis, focuses on unraveling the processes and factors that contribute to the initiation, progression, and maintenance of epilepsy. Areas of epileptogenesis research include inflammatory cytokines, mTOR pathway, proteins crucial for generating electrical impulses, breakdown of the blood brain barrier, impairment of glial cells, and the immune system. Ongoing advancements in the identification of genetic factors underlying epilepsy play a crucial role in guiding the medical care and management of affected individuals. By uncovering the genetic causes of the condition, healthcare providers can offer more personalized and targeted approaches to treatment. In cases involving heritable mutations, this knowledge not only aids in understanding the risks for affected families but also facilitates informed decision-making and genetic counseling. The continued progress in genetic research holds the potential to greatly enhance the care and support provided to individuals with epilepsy and their families.66
The significant progress in the field of genetics has been largely driven by the emergence of next-generation sequencing. These high-throughput methods of genetic sequencing have enabled faster and more comprehensive analysis of DNA and next-generation sequencing has facilitated the identification of genetic factors underlying conditions like epilepsy. This breakthrough technology has opened up new avenues for research, offering valuable insights into the genetic underpinnings of diseases and paving the way for more precise and personalized approaches to diagnosis, treatment, and management.66
There are also advancements being made to improve already effective treatments such as surgery. The introduction of novel imaging technologies represents a significant advancement in localizing the precise effects of surgical interventions and reducing the occurrence of adverse events. These cutting-edge imaging techniques provide healthcare professionals with enhanced visualization capabilities, enabling them to accurately identify and precisely target specific areas during surgery. By improving the accuracy and precision of surgical procedures, these advancements contribute to minimizing potential risks and optimizing patient outcomes. Researchers are also investigating a combination of imaging techniques such as diffusion tensor imaging (DTI), functional magnetic resonance imaging (fMRI), and magnetoencephalography (MEG) to improve preservation of language zones. High-frequency oscillations (HFOs) detected in the neocortex and temporal lobe have emerged as potential biomarkers for epileptic networks and can help surgical mapping and outcome prediction of epilepsy surgery.66
The therapeutic application of electrical brain stimulation continues to be a strategy of significant interest. By delivering controlled electrical impulses to specific regions of the brain, this approach holds potential for modulating neural activity and treating various neurological conditions. Ongoing research and advancements in this field aim to further refine and expand the therapeutic applications of electrical brain stimulation, offering new hope for individuals with neurological conditions.66
Despite the numerous challenges posed by epilepsy, there is reason for optimism regarding the potential for improved treatment options for patients. Ongoing research, advancements in technology, and a better understanding of the underlying mechanisms of epilepsy are paving the way for innovative approaches to managing the condition. From novel antiepileptic drugs and neurostimulation techniques to surgical interventions and non-pharmacological therapies, there is a growing arsenal of treatment modalities being explored. Additionally, advancements in personalized medicine and precision therapies offer the potential for tailored and more effective treatment strategies. With a focus on improving seizure control, reducing side effects, and enhancing the overall quality of life for individuals with epilepsy, the medical community is actively working towards advancements in epilepsy treatment. While challenges remain, the collective efforts of researchers, healthcare professionals, and individuals affected by epilepsy provide hope for a brighter future with improved outcomes and better quality of life for patients.
None.
The authors declare that there are no conflicts of interest.
None.

©2023 Zoe, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.