Journal of
eISSN: 2373-633X


The changes of liver metastases under treatment with tyrosine kinase inhibitors can be monitored by MRI imaging. Evidence for a reduction of single metastasis volume or changing of the contrast medium reaction are uncertain response criteria of treatment. In the presented case, MRI native signal changes could be detected in different metastasis caused on the basis of susceptibility effect. This special treatment effect has biological reasons in the endothelial damage and membrane disruption within the metastasis, leading to ferritin deposits in the tumor tissue. This increased concentration of ferritin can be detected by MRI natively and is a possible predictor for tumor response.
Keywords: axitinib, liver metastasis, MRI
The treatment of liver metastases with cytostatics leads, depending on the primary tumor, to an inner toxic reaction of the tumor cell by interfering with the chemotherapeutic agent in the mitosis followed by cell death. A size reduction of metastasis and a changed perfusion applies, regardless of the effective mechanism, as a sign of remission.1 The different cellular changes and the morphological changes on MRI in the structure of the tumor are highly variable, even in the use of conventional liver-specific contrast agent.2,3 This case presentation shows for the first time signal changes on MRI of liver metastases from a specific, oral taken cytostatic (Axitinib), who may be regarded as morphological signs of remission of the metastasis in the native MRI.
A 68-year-old female patient with a metastatic renal cell carcinoma was treated with Axitinib for 6months (Inlyta®, 3x5mg/d). The medicine belongs to the group of low molecular weight tyrosine kinase inhibitors (sm-Kl). Immediately before initiation of the therapy and 6 months thereafter an MRI scan of the liver was done to evaluate the progress. The patient reported no significant side effects, only bilirubin increased intermittently during therapy.
The MRI of the liver was performed in a 3 Tesla high-field magnet (TIM TRIO, SIEMENS) with standard examination protocol in T1-weighted and T2-weighted images with a liver-specific contrast agent (Primovist®, from Bayer Healthcare) and showed a surprising signal change in individual metastases already in the native Gradient Echo T1-weighted images (FL 2D, TR 450ms/TE 9 and 11ms/flip 60°). Low signal differences at low different echo time are negligible. Figure 1a and b before the start of therapy multiple liver metastases of the renal cell carcinoma with a donut phenomenon can be detected. In the follow-up metastases show only a partial reduction under Inlyta® therapy, but a hypointense and central, punctiform-confluent signal reduction (arrow).
The low molecular weight kinase inhibitors (sm-Kl), to which Axitinib belongs, have been used for the treatment of liver metastases from renal cell carcinoma in 2nd line. This medication group applies as selective and highly specific for tumor cells, but leads to membrane disorders and receptor inhibition of hepatocytes as well as to hemorrhage in many other organs.2 In the product information of Axitinib the manufacturers warn against the risk of organ bleeding (nose bleeding, pulmonary hemorrhage, vaginal bleeding, menorrhagia). At the same time one of the adjuvants of the 5mg tablet in the film coating is iron (III) oxide.4 Our case presentation of a MRI of the liver after Axitinib therapy shows a treatment-related signal change in liver metastases in T1-weighted images in the sense of a susceptibility effect. This effect can occur with deposits of iron compounds in the tissue. The distribution pattern of signal interference within the metastasis with a multifocal, partly mulberry-like signal cancellation on T1-weighted images can be caused among other things by focal hemorrhages, which are not primarily visible in the peripheral zone.
Figure 2a and b shows a liver metastasis (short arrow) in the left lobe without significant signal change by Inlyta® therapy. It has come in the recording control to a size increase (PD, Recist 1.1). The metastasis (long Arrow) with a decreased signal becomes progressively smaller (PR, Recist 1.1). The correlation between signal quality and size in the adjacent liver metastases in the same patient may reflect the different biology or vascularization of the cell assembly. Standard independent image review for oncology study (RECIST) includes only the criterion of the size of the lesion. Maybe the criterion of signal reduction is an additional useful indicator for treatment response or not-response.
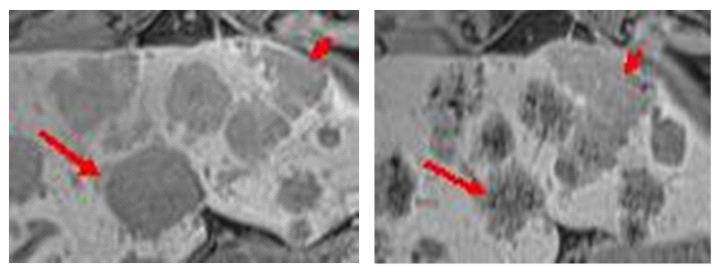
Figure 2MRI, TIM Trio, 3 Tesla (SIEMENS), Gradient Echo T1-weighted, transversal, 10 ml Primovist®, storage phase. Enlarged section from a single sectional image of untreated liver metastasis (left) and treated liver metastasis (right). The therapy with Inlyta® generates a strongly hypointense, mulberry-like signal in the metastases. These metastases are reduced in size (long arrow). Other metastases show only occasional point-like signal attenuation after therapy. These are rather increasing in size (short arrow).
A low sedimentation of the iron (III) oxide from the shell of the film-coated tablet cannot be excluded. The reason for this accumulation of ferromagnetic compounds under the effect of Inlyta® in the notion of a toxic response of tumor cells in a metastatic formation, needs to be further explored histologically. The inhibition on the Vascular Endothelial Growth Factor Receptor (VVEGFR-1, -2 and -3) by Axitinib needs to be discussed as well as the endothelial damage, bleeding and sedimentation of ferritin in the metastasis. The sedimentation of iron as a result of the bleeding seems to be the predominant effect in the present imaging.2 MRI is a highly sensitive method for the detection of Fe2 +/3 + complex, resulting in hemolysis or bleeding.
Due to the lack of histological confirmation of the single finding, we can only recommend further targeted investigations. It is important to note that other physical effects can make possible an early assessment of treatment response in addition to the uncertain criteria of image contrast enhancement in MRI. If this side-effect is a sign to a partial remission (PR) in the metastases and if hemorrhages in metastases have a positive effect on their own regression, this MRI phenomenon is perhaps significant.5
None.
Author declares there are no conflicts of interest.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.