Journal of
eISSN: 2373-6437


Transfusion-related acute lung injury (TRALI) is a major complication post-transfusion. A consensus definition of TRALI has been recently established to improve diagnosis but the pathogenesis of TRALI is yet to be understood. Although the antibody-mediated two-hit model of TRALI is the classical narrative, increasing evidence of the probable implications of prolonged storage of blood provides novel mechanisms towards storage lesion- the potentially injurious cellular and biochemical changes that occur in stored red blood cells. Red blood cell-derived lipids and micro vesicles may have been playing an important role in the development of TRALI. This article will provide a brief overview of the current understanding of TRALI and then discuss the implications and the potential mechanisms by which stored red blood cells may lead to TRALI.
Keywords: Membrane lipids, Microvesicles, Blood storage, ARDS
Transfusion-related acute lung injury (TRALI) is a major complication characterized by rapid onset of pulmonary edema, impaired oxygenation, neutrophil infiltration and other features seen in acute respiratory distress syndrome (ARDS). Several retrospective and prospective case-control studies have shown that critically ill patients who were admitted to ICU and received transfusion of blood products exhibited prolonged hospital stay and had high risk of mortality.1‒4 Multiple protective measures to reduce the harm of transfusions have been implemented and the most prominent approach is the use of leukoreduced blood transfusion. Leukoreduction involves the removal of white blood cells from the transfused unit and thereby reduces the chance of an antibody-antigen interaction between the donor and recipient blood.5
Studies have recently paid particular attention to the relationship between storage duration of red blood cell (RBC) and subsequent transfusion complications. A recent link has been found between red blood cells stored for a prolonged period and post-transfusion mortality.6 The potentially injurious products, such as membrane lipids and reactive oxygen species that accumulate in a stored blood unit-collectively known as "storage lesion" may play an important role in the pathogenesis of TRALI.7,8 In this article, we will provide an overview of the diagnosis, incidence, pathogenesis, and risk-factors associated with TRALI and focus more on the discussion of the potential effects of RBC storage lesion, and in particular RBC-derived lipids, in the context of TRALI.
Demographics
TRALI is defined as the appearance of symptoms and physiological parameters that fulfill the classical definition of acute lung injury (ALI) within 6 hours from the time of transfusion in the absence of any other known risk factor.9,10 Nevertheless, recent insights into post-transfusion complications amongst critically-ill patients reveal that symptoms of ALI may be delayed to appear at 24-72 hours (Table 1).11,12
|
Definition |
Diagnostic Parameters |
|
TRALI |
Bi-lateral infiltrates with non-cardiogenic pulmonary edema. ARDS appearance within 6hours after transfusion. Hypoxemia: PaO2/FiO2< 300 mmHg. |
|
Potential TRALI |
Similar to TRALI diagnosis with an additional risk-factor for lung injury/ARDS. |
|
Delayed TRALI |
Diagnostic parameters characteristic of TRALI within 24-72 hours after transfusion. |
Table 1 Summary of current diagnostic criteria for TRALI
TRALI is characterized by clinical features that include, but not limited to, dyspnea, tachypnea, respiratory insufficiency, non-cardiogenic bilateral pulmonary edema, decreased pulmonary compliance and acute hypoxemia (PaO2/FiO2<300 mmHg, (Table 1).13,14
The incidence of TRALI ranges from 1/5,000-1/1,323 transfusions within the general population and it is the leading cause of transfusion-related death in the United States.15 Such large range in incidence could be attributed to the relatively unspecific diagnostics tools available for TRALI, past cases of under-diagnosis or misdiagnosis, and the elevated incidence of TRALI within vulnerable populations.14‒16 A recent study analyzing the incidence of TRALI in critically ill patients admitted to intensive care unit (ICU) observed that over 5% of all patients who received blood transfusion developed TRALI, which is 50-100 times greater occurrence than in the general hospital population.12,17
Risk factors related to recipients and donors
The risk factors of TRALI are highly diverse, among patients with cardiac surgery, sepsis, and other critical illness.6,18‒20 Vlaar et al.15 conducted a case-control study to identify risk factors associated with incidences of TRALI in a cohort of critically-ill patients. When compared to transfused control subjects, patient-related risk factors included emergency cardiac surgery, mechanical ventilation, hematologic malignancy, and high Acute Physiology and Chronic Health Evaluation II (APACHE II) score. A recent retrospective study showed substantially greater odds of TRALI for older individuals, in females than in males, white population than in non-white population, and in patients with history of post-inflammatory pulmonary fibrosis.21 The study highlights the importance of the underlying condition of the patients in particular if they underwent prior alloimmunization, such as previous pregnancies, transfusions and bone marrow or organ transplantation. Also, greater number of products transfused (more than 5) is associated with a higher incidence of TRALI.21
In addition to patient risk-factors, there are also donor risk-factors which relate to the source of the transfused blood unit. One such risk factor is plasma from female donors, which could be found in small residual amounts in packed red blood cells or in whole blood units even after filtration.22 Another risk factor is blood from multiparous donors (women who have had more than one child) who have higher amount of human leukocyte antigen (HLA) antibodies and have an increased likelihood for HLA allostimulation which therefore carries a greater risk for antibody-mediated TRALI.12
A recent study by the Canadian Blood Service reported that incidence rates are similar in both adult and pediatric populations. The study observed that TRALI poses significant risk for mortality and that its accurate diagnosis and extra vigilance is crucial during blood transfusions in children.23
Pathogenesis
The classical TRALI is the result of "two-hit" insults or two independent events. The first event pertains to the existing underlying diseases of the recipients. These underlying conditions will inevitably initiate a pro-inflammatory response within the host, resulting in release of inflammatory cytokines and chemokines, as well as polymorphonuclear neutrophil (PMN) infiltration into the lung. The second event consists of the transfusion of biologic response modifiers and their interaction with recipient antigens.14 Such modifiers are essentially antibodies in the donor blood and the cognate antigens for these antibodies are either (HLA) class I and class II or human neutrophil antigens (HNA) expressed on the surface of the recipient’s PMNs.14,19 HLA class II can also bind to monocytes that are found in the recipient’s circulation and express the cognate antigen and initiate the release of TNF-α and IL-1β.14
In addition, whole blood- derived platelet concentrates (WB-PLTs) and aphaeresis platelet concentrates (A-PLT) are the source of the pro-inflammatory soluble CD40 ligand (sCD40L). sCD40L can bind to the CD40 receptor on a PMN, resulting in the non-antibody mediated pro-inflammatory cytokine release and endothelial damage.24 The outcome is similar to the above mentioned antibody-mediated process.
Clinical evidence
Most clinical studies of TRALI have been conducted in operating rooms and ICU, in the form of prospective and retrospective case-control data analyses in critically ill patients in many different settings (Table 2).3‒5,17‒19,21,23,25,26 Koch et al.27 have primarily examined the effects of transfusion on cardiac patients, observing significant increase in respiratory insufficiency and necessity for mechanical ventilation after transfusion of older RBC units.3,27 Silliman and colleagues investigated the etiologic factors of TRALI prospectively in a hospital setting and observed that in addition to cardiac surgery, the age and levels of lipids found in transfused RBC units may pose risk for developing TRALI.19 In a retrospective study in critically ill patients, Vlaar reported that TRALI is common in critically ill population and that 90-day mortality in patients with TRALI was higher than control subjects who received blood transfusion or the non-transfused patients with acute lung injury (ALI).15 Janz et al.6 focused on the effect of transfusion of RBC units stored for different lengths in sepsis patients. They found that over half of the patients developed ARDS/TRALI within 96 hours post-transfusion, and thus accounting for the delayed TRALI phenomenon.6
|
Author |
Study Type |
Setting |
Underlying Disease |
Year of Study |
TRALI Incidence-Pulmonary Complications |
Mortality |
|
Silliman et al.19 |
Retrospective |
Hospital |
- |
1991-1995 |
0.08%a |
- |
|
Hebert et al.18 |
Randomized control trial |
ICU/OR |
Critically ill, cardiac surgery |
1998-2001 |
- |
10.5% |
|
Koch et al.3 |
Retrospective |
Cardiac surgery |
Coronary-artery bypass grafting orheart-valve surgery |
1998-2006 |
Respiratory insufficiencyb (9%) |
9.2%c |
|
Eikelboom et al.4 |
Prospective |
Hospital |
Cardiovascular disease |
2002-2006 |
- |
10.8% |
|
Karam et al.26 |
Prospective observational |
Paediatric ICU |
Cardiovascular disease, respiratory disorder, central nervous system, other |
2004-2005 |
Respiratory dysfunction (10%) |
5.1% |
|
Vlaar et al.15 |
Retrospective |
ICU |
Critically ill, cardiac surgery, other |
2004-2007 |
5.1% |
47%d |
|
Gajic et al.45 |
Prospective |
ICU |
Critically ill |
2005-2007 |
8.0% |
41%d |
|
Vlaar et al.16 |
Prospective |
ICU |
Cardiac surgery |
2006-2009 |
2.4% |
13%d |
|
Toy et al.22 |
Prospective |
Medical centres |
Varied |
2006-2009 |
0.02% (per transfusion) |
- |
|
Janz et al.6 |
Retrospective observational |
ICU |
Sepsis |
2008-2010 |
51% (in sepsis patients) |
- |
|
Kor et al.33 |
Randomized clinical trial |
ICU |
Mechanically ventilated medical or surgery patients |
2008-2010 |
- |
39.5% |
|
Lieberman et al.23 |
Retrospective observational |
Canadian Blood Service data |
Varied |
2001-2011 |
0.006% (per transfusion) |
Adults: 5.2% Paediatrics: 5.9% |
Table 2 Summary of clinical studies in various patient populations of TRALI
aIncidence only in product of platelet concentrate.
bAn outcome investigated along with other complications in patients receiving older blood.
cIn patients receiving older blood.
dCompared to non-transfused ALI controls.
—No data available.
Animal models of TRALI
Several animal models have been established to study TRALI (Table 3). In an ex vivo rabbit model infused with HNA-3a (5b) antibodies, investigators observed that the infusion of anti-5b antibodies in the presence of 5b-positive PMNs lead to severe lung edema within 3 to 6 hours.28 Silliman et al.13 showed that perfusion of rat lung with 42-day old purified lysophosphatidlcholine, Lyso-PC, a lipid commonly found as part of the storage lesion aggregate, led to significant pulmonary edema and PMN infiltration.29 Kelher et al.30 investigated the effects of plasma from stored packed red blood cells (PRBCs) and MHC class I antibodies-OX18 and OX27 in a two-event in-vivo rat model with lipopolysaccharide (LPS) as the first event. It was concluded that TRALI was the result of two events and that the condition is PMN-dependent as histological analysis revealed that OX18 and OX27 are localized to the surface of PMNs in the rat lung. When comparing aged RBC supernatant with aged but washed RBCs in LPS-primed animals, aged supernatant lead to increase of both inflammation and coagulopathy markers in a two-event TRALI model.31 In a "two-hit" rat model pre-treated with either saline or LPS, filtration of plasma containing 50 µg/ml of antibodies OX27 prior to infusion inhibited TRALI compared to unfiltered plasma which resulted in lung injury.32
|
Author |
Year |
Model |
Two-Event |
Outcome |
|
|
|
|
|
1st Event |
2nd Event |
|
|
Seeger et al.28 |
1990 |
Rabbit lung (ex vivo) |
Anti-5b antibodies |
- |
Severe lung edema |
|
Silliman et al.29 |
1998 |
Rat lung (ex vivo) |
LPS/Saline |
Plasma or lipids from PRBCs |
Pulmonary edema and neutrophil infiltration |
|
Kelher et al.30 |
2009 |
Rat (in vivo) |
LPS/Saline |
Plasma from PRBCs or MHC class I antibodies (OX18 and OX27) |
Acute lung injury and antibody localization to the neutrophil surface |
|
Vlaar et al.31 |
2010 |
Rat (in vivo) |
LPS |
Aged erythrocytes or supernatant of aged erythrocyte (day 14) |
Lung inflammation and coagulopathy |
|
Tung et al.39 |
2012 |
Sheep (in vivo) |
LPS/Saline |
Supernatant from day 1 or day 42 stored PRBCs |
TRALI developed predominantly in LPS-treated sheep |
|
Silliman et al.32 |
2014 |
Rat (in vivo) |
LPS/Saline |
1) Filtered or unfiltered OX27 plasma or 2) Filtered or unfiltered day 0 or day 42 RBC supernatant |
Experimental filtration inhibited TRALI in both 2nd event treatment groups |
Table 3 Summary of key animal model of TRALI
Red blood cell storage and TRALI
The quality and efficacy of RBC storage may have significant impact on the occurrence and pathogenesis of TRALI.33,34 The current standard of duration of storage of red blood cells (RBCs) by the American Association of Blood Banks is a maximum of 6 weeks (42 days) of storage.35 During this period, RBC are stored in refrigerated conditions and kept in a preservative solution (such as sodium-adenine-glucose-mannitol [SAGM]) which helps extend shelf-life.36 This 42-day limit was assigned by the Food and Drug Administration (FDA) within a criteria that requires 75% recovery of transfused blood cells 24 hours after transfusion.7,34
Storage lesion
Despite meeting storage restrictions criteria, 42-day old blood is not considered to be as effective or even as safe as blood stored for shorter durations. Reversible and irreversible changes to the function and properties of RBCs can be observed as early as 14 days after collection (Table 4).36 Stored units of blood experience progressive biochemical and morphological deterioration resulting in RBCs that have compromised viability, cell size, deformability, and lipid-membrane composition (Table 4).7,8,34,37,38 In addition, prolonged storage leads to accumulation of potentially harmful debris, most notably, aggregates of oxidized membrane lipids and proteins.7,34,39 RBC membrane and lipid loss and micro particle formation are probable causes for procoagulant, proinflammatory, and thrombogenic effects post-transfusion, and their potential role in context of acute lung injury will be examined in detail later in this paper (Figure 1).40 Collectively, the detrimental changes in RBC morphology and the accumulation of the injurious debris commonly found in stored blood are termed "storage lesion".
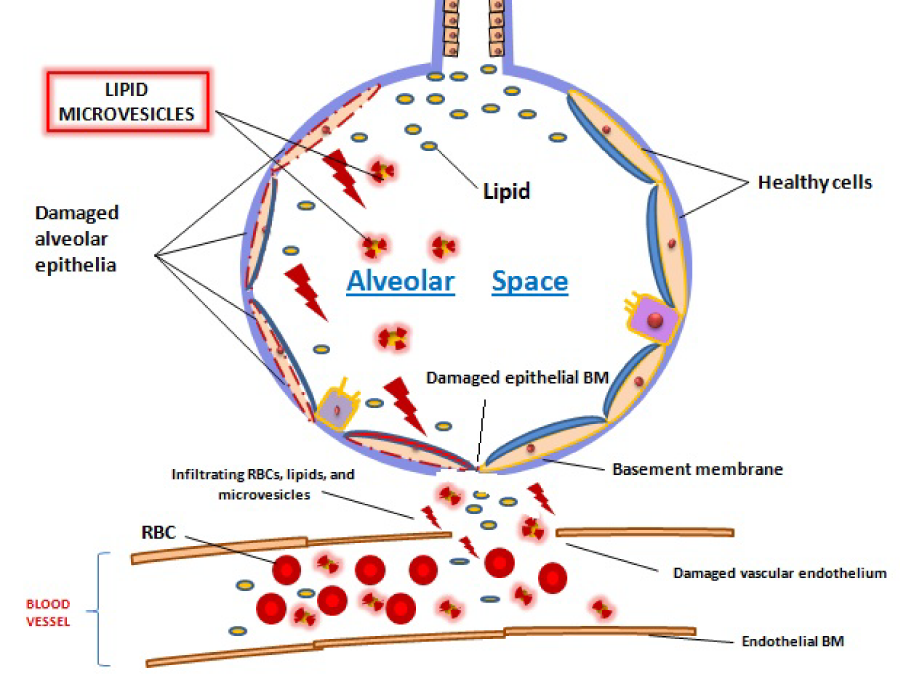
Figure 1 Possible injurious pathogenic chain of events of storage lesion components-red blood cells, microvesicles and lipids-in the context of TRALI. Leakage of blood storage components into alveolar space, secondary to inflammation and damage of the vascular endothelium, could be implicated in TRALI.
Functional changes in stored red blood cells |
Aggregates in red blood cell supernatant |
↑ RBC rigidity |
↑ Free haemoglobin |
↑ RBC lysis |
↑ Potassium |
↓ ATP production |
↑ Lactate |
↓ 2,3-diphosphoglycerate (DPG) |
↑ Sodium |
↓ pH |
↑ IL-1β |
↑ Adhesion to endothelial cells |
↑ Lipid microvesicles |
↑ Membrane vesiculation |
↑ Oxidized proteins |
↓ Oxygen affinity |
↑ Peroxidised lipids |
Table 4 Morphological, functional and biochemical changes taking place in stored red blood cell unit over a period of 42 days (based on Cohen and Matot34)
Clinical relevance
Although the relationship between storage duration of RBCs and potential incidence of TRALI was not always found15,19,33,41 a meta-analysis examining 21 studies (primarily in the field of cardiac surgery and trauma) demonstrated that mortality increased with transfusion of older RBC units compared to units stored for shorter periods (a time period closer to 21 days was set as a cut-off point between fresh and older blood, unless mentioned otherwise).2 In trauma patients, the risk for development of ARDS was significantly increased in patients who received transfusion of leukoreduced RBCs stored for longer than 14 days, and transfusion of red cells older that 28 days has also been associated with deep vein thrombosis and multiple organ.19,25 Patients who received blood units stored for longer than 14 days there was a significantly greater incidence of multiple organ dysfunction syndrome (MODS), with respiratory dysfunction observed in 10% of the cases.26 In a retrospective observational study, post-transfusion ALI/ARDS has been assessed in critically ill patients with sepsis. Over half of all the patients that met the criteria have developed ALI/ARDS within 96 hours after transfusion. Most importantly, the age of transfused blood in patients who developed ALI was significantly greater than in those who did not.6
Despite these numerous studies which suggest that the use of fresher blood for transfusions could be potentially beneficial, a randomized control trial did not find a difference in outcome between the use of a single unit of fresh blood and a single unit of stored blood when assessing pulmonary, inflammatory and coagulation markers.33
Animal models
In a healthy rat model, transfusion of aged (day 14) but not fresh RBCs contributed to increased pulmonary inflammation, while in lipopolysaccharide LPS-primed rats, aged RBCs lead to increased levels of coagulopathy.31 In sheep that were pre-treated with either saline (healthy sheep) or LPS, TRALI developed in as much as 80% of the LPS-treated sheep after transfusion of 42-day old RBCs but not in healthy sheep transfused with either day 1 or day 42 RBCs.39 Callan et al.42 demonstrated significant inflammatory responses in healthy dogs after receiving transfusion of 28-day old but not 7-day old RBCs.
RBC-derived lipids
A significant component of the accumulated storage lesion consists of primarily lipid-rich micro vesicles, free polar and non-polar lipids (either from the plasma or from the RBC membrane), reactive oxygen species, and other biological mediators. Changes throughout storage can directly affect membrane phospholipids composition and integrity of RBCs, both of which are integral to the maintenance of red cell function.43,44 Although the exact mechanism by which the biologically active lipids can become injurious within the transfusion recipient is yet to be illuminated, lipids were, nevertheless, assessed as potential transfusion-related risk factor in clinical studies. Assessment of patient blood post-transfusion neutral lipids and Lyso-PC revealed higher priming ability after transfusion compared to pre-transfusion.19 In a case-control study where the amount of Lyso-PCs in transfused was assessed, the concentration of Lyso-PC in blood transfused into patients who developed TRALI was significantly higher than in units given to control subjects.45
Silliman et al.46 have carried out multiple studies to illuminate the effect of stored RBC-derived lipids. In a rat model, results showed that perfusion of rat lung with 42-day old purified lysophosphatidlcholine led to significant pulmonary edema and lung injury in LPS-primed rats. The investigators further demonstrated that infusion of a mixture of four non-polar lipids purified from 42-day old leukoreduced RBC from healthy volunteers induced lung injury in LPS-treated rats, but 1-day lipid solution did not.47
Microvesicles
The oxidative environment within the stored unit of RBCs leads to protein and phospholipids break down which ultimately results in the shedding and accumulation of bioactive micro vesicles (MVs). These RBC membrane-derived MVs have more recently been shown to have physiologic and pathophysiologic significance by playing a role in inflammation and vascular dysfunction.47 Cardo et al.48 assessed the neutrophil-priming ability of washed and unwashed RBCs stored for different durations and specifically the role of micro vesicles in priming neutrophils in vitro. They observed that after washing and filtration of RBC, the expression of CD11b, a surface marker of neutrophils, was reduced suggesting that micro vesicles could recruit neutrophils. Almizraq et al.43 have recently shown that the MV released from RBC membranes is rich in polyunsaturated phospholipids.44 In a comparative analysis of the lipid composition of MV and RBC membranes during storage showed that the glycerophospherine (PS) profile of stored RBC membranes did not resemble the PS profile of fresh RBCs, and the MV showed greater PS abundance.44 This finding suggests that the loss of PS from the RBC membrane could be an important contributor to the process of vesicle-formation during storage. The same group of investigators recently revealed a near doubling in the absolute number of RBC-derived MV 42 days of storage.43 Nevertheless, the role of MVs in TRALI development is yet to be elucidated.
Recently, great attention has been paid to storage lesion rather than the established antibody mechanisms in the pathogenesis of TRALI. The mechanisms of RBC membrane derived lipids and micro vesicles contributing to TRALI are currently under investigation. The potential for preventing storage lesion and the associated formation of lipids and micro vesicles could be a novel approach to prevent and reduce the incidence of TRALI.

© . This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.