International Journal of
eISSN: 2577-8269


Research Article Volume 2 Issue 4
1R&D Department, Laboratori Baldacci SpA, Italy
2Department of Cardiology, Acireale Hospital, Provincial Health Centre of Catania, Italy
3Department of Cardiology, Section of Angiology, Acireale Hospital, Provincial Health Centre of Catania, Italy
Correspondence: Marco Bertini, R&D Department, Laboratori Baldacci SpA, Via San Michele degli Scalzi 73, 56124 Pisa, Italy, Tel +39 050 313271
Received: August 03, 2018 | Published: August 17, 2018
Citation: Bertini M, Costa M, Rapisarda M, et al. Microcirculation and lymphedema: the role of aminaphtone. Int J Fam Commun Med. 2018;2(4):224-227. DOI: 10.15406/ijfcm.2018.02.00084
Aims: The microcirculation, or the micro-vascular-tissue unit, represents the anatomical-functional unit where oxygen and nutrient absorption processes take place. The microcirculation plays a central role in the elimination and reabsorption of catabolites through the colloid-osmotic and hydrostatic pressure gradients equilibrium, which is modified in lymphedema. The latter is characterized by an interstitial edema of high protein concentration caused by inadequate lymphatic drainage. The effects of drug therapy with Aminaphtone in the pharmacological treatment of lymphedema have been evaluated. The management of lymphatic edema, in fact, involves the association of several treatments ranging from drug therapy, to manual and non-manual drainage techniques and compression therapy.
Materials and methods: Two main clinical studies ( study 1: De Anna and Coll: “Effects of Aminapthone therapy on chronic venous insufficiency and lymphatic stasis” Minerva Carioangiol 1989 May; 37 (5) pp 251 -254 and study 2: G. Spezzigù and Coll “Role of aminapthone in the treatment of lymphatic edema. Presentation of a case-series and proposed for a randomized-controlled study” Minerva Cardioangiol, 2012 Vol 60, Suppl 1 al N 6) and a pre-clinical study (study 3: G. Salazar and Coll “Gene expression profiling reveals novel protective effects of aminapthone on ECV304 endothelial cells” European Journal of Pharmacology 2016 782 pp 59 – 69) have been collected and revised.
Results: After combined treatment of lymphedema, aminaphthene therapy was able to significantly reduced edema in patients with stages I and II limphedema.
Conclusions: Although there were not many studies on this topic, it seems that aminapthone treatment in combination therapy could have a role in the reduction of lymphedema in the early stages of pathology.
Keywords: aminaphthone, lymphedema, microcirculation, permeability, colloidosmotic pressure, capillary filtrate, pathological condition, tissues, colloid-osmotic, hydrostatic pressure, equilibrium, chronic inflammation, fibrous tissue
The microcirculation or micro-vascular-tissue unit consists of a dense network of capillaries, composed of endothelial cells and circulating cells, and regulates the blood supply, depending on the functional moments of the individual regions, through two different mechanisms: vasomotility control (nervous control) and permeability control (endothelial control). The Micro-circulatory unit consists of: arteriole, capillary venula, AVA (Arteriovenous Anastomoses), lymphatic capillary, nerve termination and connective matrix (Figure 1). Edema is the result of the modifications in the capillary exchange with accumulations of liquid within the tissues, generally circumscribed, which originate as a result of an alteration of the microvascular wall with abnormal increase of its permeability and modification of the gradients of hydrostatic pressure within the capillary bed. The microcirculation plays a central role in the elimination and reabsorption of catabolites through the colloid-osmotic and hydrostatic pressure equilibrium which is modified in cases of lymphedema which represents a pathological condition characterized by extravasal accumulation of lymph due to excess of lymphatic production. It happened when the lymphatic system is no longer able to regularly transport the lymph. This causes the deposition in tissues of liquids with a high protein concentration. The proteins thus deposited can cause a chronic inflammation in the tissues that can turn into fibrous tissue.
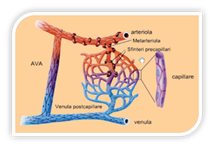
Figure 1 The Micro-circulatory unit consists of: arteriole, capillary venula, AVA, lymphatic capillary, nerve termination and connective matrix.
Lymphedema is classified as: primary (congenital or acquired), that is, a consequence of a congenital (spiegare meglio!!!) and secondary lymphatic dysplasia, that is due to a lesion of the lymphatic vessels and/or lymph nodes, in consequence of the obstruction, compression or removal, or an excessive lymphatic load (ex. anatomical obliteration following a trauma or a radical surgery, a tumoral event, an advanced venous system diseases, etc.). The course of a lymphedema, both primary and secondary, is characterized by various stages with a different progression for each subject:
Stage I: It can start with a latency period (absence of clinical symptoms and the alteration of the lymphatic system can be ascertained only with instrumental examinations) and then it manifests as a mild soft edema that regresses totally or partially during the night or with the lower limbs elevations. The typical clinical signs are the edema on the back of the foot and the accentuation of the natural skin folds at the metatarsofalangee joints (Stemmer's sign). Symptoms can be confused with other pathologies: occasional cramps, nocturnal tingling or pruritus.
Stage II: Lymphoedema occurs as a hard swelling, it has no tendency to regression and it is invasive. The symptoms become more and more persistent. The skin surface is dry, hyperkeratotic and grayish.
Stage III: Fibrolymphedema (Lymphoedema o lymphedema? Scriviamo sempruguale!!!) With hardened subcutaneous tissue and trophic disorders. The body reacts to the persistent stagnation of high-protein liquid producing fibromatosis. Resolution is unlikely at this stage. The most severe expression of this stage is called elephantiasis.2–4
In physiological conditions the capillaries filter at the level of the arterial end (arterioles) and reabsorb into the venous (venula). 90% of the arterial filtrate is reabsorbed at the venous level and 10% (about 2 liters per day) of the interstitial fluid (with water, proteins, dead cells, viruses) is recovered by the lymphatic system, which prevents the formation of edemas by pouring it into the blood circulation. In pathological conditions, if the lymphatic system is damaged, for example due to radiotherapy or surgery with removal of the lymph nodes, the interstitial fluid cannot be completely reabsorbed by the capillaries, so it accumulates in the interstitial tissues and creates swelling. Lymphedema is therefore an edema due to an alteration of the lymphatic system. The lymphatic system consists of lymphatic capillaries. Lymphatic vessels originate between the cells of the interstitial connective tissues with very permeable capillaries covered. The lymphatic capillaries and the extracellular matrix, from which they originate, are endowed with negative pressure; this facilitates drainage of excess interstitial fluids (lymph), preventing edema and lymph nodes. The endothelium may be continuous or fenestrated, fusiform-like cells, with abluminal surface (interconnected with tissues by basal membrane, collagen, proteoglycans, heparansulphates, integrins) or luminal (interconnected with blood by Fazzy-Coat: mucopalisaccharides, glycoprotein, fibrinogen, fibrin) with elastic cytoskeleton that allows to saturate any continuous solutions. Among the endothelial functions we remember the function of selective barrier between blood and tissues. The formation of edema occurs at the microcirculation, that is in the area where the interchange of molecules takes place between the blood and the interstitial liquid and between this and the cells of all tissues (Figure 2).5,6
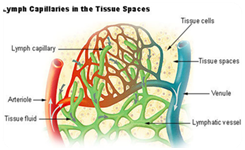
Figure 2 In pathological conditions, the district in which vascular-tissue exchanges occur is increased and extends also to arterioles and venules, as well as, of course, to capillaries.
In pathological conditions, the district in which vascular-tissue exchanges occur is increased and extends also to arterioles and venules, as well as, of course, to capillaries. The site in which the massive fluid outflow occurs, in pathological conditions, is represented by the collecting venules, and therefore not by the point of greatest filtration in physiological conditions (capillary-venular passage zone). From here, therefore, the inflammatory response and the changes in the microcirculation are activated. We assure:
The increase in vascular permeability (detectable with fluorescence videocapillaroscopy) is due to: an increase in amplitude of the "small pores" system responsible for trans-capillary diffusion to mucopolysaccharide accumulation (on which the basal membrane thickening depends) which modifies the selectivity of the filtration process. It generates alteration between the colloid-osmotic pressure of the circulating blood that tends to retain the liquid inside the vessels and the hydrostatic pressure which, instead, favors the filtration. In physiological conditions the hydrostatic pressure tends to cause the liquids escape from the capillary lumen and that the colloidosmotic pressure tends to restrain them or make them fall, note that: In the proximal portion of the microcirculation the hydrostatic pressure (35 mmHg) prevails on colloidosmotic pressure (25 mmHg) for which a filtering pressure of 10 mmHg is present. The liquid is then filtered from the plasma in the gap.
In the intermediate portion of the microcirculation the hydrostatic pressure is equal to the colloidosmotic pressure (25 mmHg-25 mmHg) for which the filtering pressure is 0 mmHg, with the consequence that the amount of liquid that diffuses from the capillary lumen in the interstitium equals that that spreads from this to that. In the terminal portion of the microcirculation the colloid-osmotic pressure (25 mmHg) prevails on the hydrostatic pressure (15 mmHg) with the consequence that it exerts a resorption force that draws liquid from the interstitium. Briefly, within 24 hours, the excess of liquid that passes through the interstitium corresponds to about 5 L which re-enter the blood through the lymphatic circulation. In pathological conditions, there is an increase in hydrostatic pressure, therefore a greater quantity of filtered liquid and a reduction in colloid-osmotic pressure, therefore a smaller quantity of retained liquid. (spiegare meglio!!!!). Therefore, an early diagnosis to reduce edema is essential to avoid the worst stages, especially in those who are familiar with lymphoedema (Figure 3).7
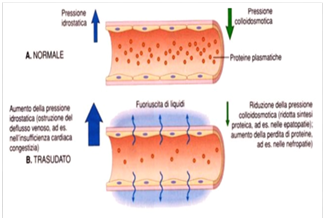
Figure 3 diagnosis to reduce edema is essential to avoid the worst stages, especially in those who are familiar with lymphoedema.
Suggestions and useful treatments are: Manual lymphatic drainage, Pressotherapy, Elastic brace (sock, bracelet or multi-layer bandage), Use of customized ergonomic insoles and postural rehabilitation, Nutrition and adequate physical activity and pharmacological therapy (phlebotonic, etc.).8
Aminapthone (2-hydroxy-3-methyl-1,4-naphthohydrochinone-2-paraminobenzoate) is a drug that has recently demonstrated to reduced the expression of VCAM and ELAM (E-Selectin), to reduced the endothelial espression of Endothelin 1 (ET-1) and to possess endothelial anti-flogistic activities by inhibits the expression of pro-inflammatory cytochine. Aminapthone could represent a first drug of a new class of product named “endothelial cell protector”. Since increase in VCAM, ET-1 and pro-infammatory molecules expression could be detrimental in lymphoedema, it seems reasonable to use this drug as add therapy in lymphedema. We have reviewed and analyzed three different published studies in which the drug has been used as add therapy in patients affected by lymphedema.9
Aminaphtone mechanism of action
a. Plebotonic Action
b. Lymphagogue action
c. Increase the micrucirculatory permeability resistances
d. Endothelial protectant action
e. Anti-sludge action
f. Endothelial antinflammatory antions.
The study 1 reports the therapeutic effects of the aminapthone in the treatment of patients affected by IVC, intended as an attempt to resolve the multiple pathogenic mechanisms operating both on macroscopic venous structures and on the capillary microcosm lymphatic. It was carried out on 114 patients with IVC, divided into two randomized and controlled groups; the first group treated with the common phlebotonic, the second group with aminaphthone at a dose of 4-6 mg /kg/day, corresponding to 4/6 capsules of 75 mg/day. The effects of the therapy were evaluated by comparing the symptomatology before and after 90 days of therapy, both with subjective methods (symptom evaluation by score) and with objective methods (eco-Doppler and measurements).10
The study 2 pointed out the add values of Aminapthone treatment in control lymphatic edema in 25 patients when added to standard of care (local decongestant trteatment). The Authors underlined the importance of a correct pathology’s stadiation at the beginning of the treatment taking into account the evidence that “aminapthone treatment seems to be useful expecially when the lymphedema was in first stage (Stage 1 lymphedema). The diagnosis of primary or secondary lymphedema does not seems to influence aminapthone efficacy. The pre-clinical study 3 pointed out the remarkable role of aminapthone in the modulation of several vasoactive factors such as endothelin 1 and vascular adhesion molecules and expecially was able to demonstrate the effectiveness of the drug in modulate and control the expression of inflammatory molecules relevant in the pathogenesis of several conditions in which the endothelial disfunction is the main player and an early event such as scleroderma. lung fibrosis and atherosclerosis. Obviously the control of pro-inflammatory cytochines by aminapthone pre-incubation in vitro, together with the control of endothelin 1 and vasular endothelial cells hyperexpression, seems to be useful also in lymphedema because during the etiopathogenesis of this disease hyperexpression of cytochines at micrucirculatory level seems to play a key role for the disease progression.
In study 1 the differences were statistically significant for subjective and objective signs and symptoms of Limphoedema in the group of patients who had taken aminaphthone. The authors have obtained excellent results also in the pathology concerning the lymphatic district having available a polyvalent drug able to acts on 3 different pathogenic fronts of the I.V.C. (venous, capillary and lymphatic). The lymphoglobin and lymphocinetic action of aminapthone, has allowed a general and lasting improvement of the treated patients, postponing the therapy over time.
The study 2 clearly demostrated the effectiveness of aminapthone in control lymphedema progression when added to standard of care (local decongestion) and ponted out the role of the drug during the first stage (Stage 1) of the disease. The study 3 conforts the rationale of use for aminapthone in lymphedema demostrating its activity in control all the relevant key-factors (endothelin 1 and adhesion molecules hyperexpression and expedially pro-inflammatory local substance’s hyperexpression) able to the progression of lymphedema.12
The superiority of the effectiveness of aminaphtone on several pathogenetic fronts has been demonstrated in more than 100 patients affected by Lymphoedema. The innovative and updated mechanism of action of aminapthone together with its role in “endothelial-protection” seems to confirm its usefulness in patients affected by Lymphoedema. Obviously, there is a need of further controlled, randomized and double-blind clinical trials to confirm its effectiveness in patients affected by Lymphoedema also if it seems that its peculiar and specific mechanism of action could completely fit with the etiopathogenesis of this pathology.
None.
The author declares there is no conflict of interest.

©2018 Bertini, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.