eISSN: 2471-0016


Case Report Volume 5 Issue 4
1Department of Pathology and Radiotherapy, India
1Department of Pathology and Radiotherapy, India
Correspondence: Kafil Akhtar, Professor, Department of Pathology, Jawaharlal Nehru Medical College, Aligarh Muslim University, Aligarh (UP), India
Received: January 01, 1971 | Published: December 6, 2017
Citation: Akhtar K, Talha M, Sherwani RK, et al. Infiltrating ductal carcinoma of breast with coexistent neurofibroma-a rare association. Int Clin Pathol J. 2017;5(4):263–265. DOI: 10.15406/icpjl.2017.05.00137
Breast cancer is the second most common cancer in the women worldwide. They exhibit a wide scope of morphological features, different immunohistochemical profiles and unique histopathological subtypes that have specific clinical course and outcome. Here we present a case of 50years old female, who presented to the surgical out patients department with complaints of painless, hard swelling in the left breast for the last 5months. On routine clinical examination, radiography and aspiration cytology a diagnosis of infiltrating ductal carcinoma was made and the patient underwent modified radical mastectomy of the left breast. Histopathogical examination showed sheets and nests of tumor cells with pleomorphism, hyperchromasia and high N/C ratio along with foci of spindle cells in fascicular pattern with wavy, serpentine nuclei of peripheral nerve and few small fibroblasts. Based on histopathology diagnosis of Infiltrating ducal carcinoma with neurofibroma was made. After 12months of follow up, the patient is doing well with no evidence of recurrent disease. This case emphasises the need for a proper evaluation of cases of breast carcinoma mastectomy specimens to look for a foci of neurofibroma.
Keywords: breast, infiltrating, ductal carcinoma, neurofibroma
WHO, world health organization; NF, neurofibromatosis
Breast cancer accounts for approximately one-quarter of all cancers in females worldwide and 27% of all cancers in developed countries with a western lifestyle.1 It’s by far, the most frequent cancer among women, with an estimated 1.67million new cases of breast cancer diagnosed in 2012.2 In India, about 145,000 new cases were diagnosed in 2012 and 70,000 deaths were attributed to it. The WHO Working Group has agreed on a more clinical follow-ups and a need of a genetic data for a better understanding of the natural history of these lesions.3 Neurofibromatosis 1 is one of the most common genetic diseases in humans, presenting with multiple neurofibromas and an increased risk of various benign and malignant tumors, including breast cancer.4 Neurofibromatosis type I (NF-1) is a complex multisystem autosomal dominant human disorder caused by the mutation of a gene on chromosome 17 which is responsible for production of a protein called neurofibromin. First report of an association between NF1 and breast cancer was published in 19725 and subsequently several clinical cases of NF1 patients with breast cancer have been reported in the literature. Neurofibromatosis type II is a genetic condition which may be inherited or arise spontaneously. It’s caused by mutations in chromosome 22, also called as Merlin gene. The main manifestation of the condition is the development of symmetric, benign brain tumors in the region of the cranial nerve VIII. Schroeder et al.5 first demonstrated an association between NF-2 and breast cancer.5
Although only a minority of patients with NF1 develops malignancy as a complication of their disorder, cancer remains an important cause of morbidity and mortality in the disorder.4 Moreover, oncologists may encounter patients with NF1 in the course of treatment for cancer, and need to be familiar with the diagnosis of the disorder and its clinical features.
A 50years old female presented to the surgical outpatients department with complaints of painless lump in the left breast for the last 5months. On inspection a swelling was seen in the upper outer quadrant. Skin overlying the swelling appeared to be normal and the nipple was slightly deviated medially. On palpation a hard, painless mass of size 4x3cms was palpated in the breast with irregular margin, fixed to the underlying tissue and attached to overlying skin. Overlying skin showed peau de orange. Left axilla showed no enlarged lymph nodes. On mammography of the left breast, the swelling showed areas of microcalcification. Routine blood investigation showed increased leucocyte count. X-ray of pelvis and spine show no evidence of distant metastasis. On fine needle aspiration cytology, sheets and nests of pleomorphic atypical ductal cells was seen, with nuclear hyperchromasia, increased nuclear cytoplasmic ratio, prominent nucleoli and moderate amount of eosinophilic cytoplasm. A foci of benign spindle cells with wavy nuclear morphology was also seen. Based on the investigations, a diagnosis of infiltrating ductal carcinoma is made and the patient underwent modified radical mastectomy. On histopathology, grossly the tumor was located in the upper outer quadrant measuring 4.5x3x1.5 cm in dimension. A subareolar extension of tumour was seen below the nipple-areola complex measuring 2x1x0.7cm in dimension (Figure 1). Cut section showed a greyish hard solid tumor with areas of haemorrhage and necrosis. Six lymph nodes were also dissected. On microscopy, sheets or whorls of tumor cells was seen with cellular and nuclear pleomorphism, hyperchromatic nucleus and high N/C ratio with prominent nucleoli and moderate amount of eosinophilic cytoplasm (Figure 2). A foci of fascicular pattern of proliferation of peripheral nerves in wavy serpentine configuration and fibroblasts was seen associated with the tumor (Figure 3). Resection margin and nipple areola complex was free from any tumor deposits. Lymph nodes showed only reactive changes. On immunohistochemistry, the foci of nerve proliferation showed diffuse positivity with S100 (Figure 4). A final diagnosis of infiltrating ductal carcinoma with neurofibroma was given. Adjuvant chemotherapy with 6 cycles of Cisplatin (50mg) was administered along with 50G of Co-60 teletherapy. After 12months of follow up, no recurrent disease was noted.

Figure 1 Grossly the tumor was located in the upper outer quadrant measuring 4.5x3x1.5cm in dimension with a subareolar extension below the nipple-areola complex measuring 2x1x0.7cm in dimension.
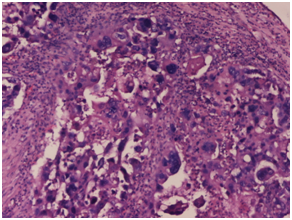
Figure 2 On microscopy, sheets or whorls of tumor cells was seen with cellular and nuclear pleomorphism, hyper chromatic nucleus and high N/C ratio with prominent nucleoli and moderate amount of eosinophilic cytoplasm. Haematoxylin and Eosin x 40X.
Neurofibromatosis 1 is one of the most common genetic diseases in humans, with a prevalence of one case in 3,000births. The disease is caused by mutations in the NF gene, which is considered a classical tumor suppressor. Neurofibromatosis 1 is an autosomal dominant condition with complete penetrance but an extremely variable phenotype. Multiple neurofibromas, café-au-lait spots, “freckling” in the inguinal and axillary regions and Lisch nodules develop in most affected individuals.6 The first report of an association between neurofibromatosis 1 and breast cancer was published in 1972.5 Several other clinical cases of neurofibromatosis 1 with breast cancer have been subsequently reported. Because breast cancer is a common tumor in the general female population, the exact relationship between neurofibromatosis and breast cancer is still not clear. Uusitalo et al.7 conduct a study on 1404 neurofibromatosis 1patients and diagnosed breast cancer in 32patients during follow up. They concluded that women with NF1 had an estimated lifetime risk of 18.0% for breast cancer.7 Da silva et al.8 emphasized the importance of using more intensive clinical and radiological approach like ultrasound and magnetic resonance imaging in early detection of breast cancer in neurofibromatosis 1 patients, as routine mammography is difficult to interpret in these patients due to cutaneous lesions.8 Schroeder et al conduct a study to found an association between neurofibromatosis 2 and breast cancer and found that one to two percent of breast cancers patients have NF2 mutations. A decrease in merlin gene expression correlates with increase in tumor grade and associated with poor prognosis.5
NF1 can obscure or delay the identification of breast lesions not only because skin neurofibromas can mask the signs of a malignant lesion, but also because patients and physicians may mistakenly consider a breast mass to be a manifestation of the primary disease.9 To date, the study with the largest cohort of NF1 individuals (n=448) that investigated the prevalence of breast cancer, as well as other types of cancer, showed that the risk of breast cancer was significantly higher in NF1 patients younger than 50years of age than in the general population.10 Sharif et al identified 14cases of breast cancer within a cohort of 304 NF1 women older than 20years, which represented a 3.5-fold risk of breast cancer in association with NF1.4 The same study calculated a 4.9-fold risk of developing breast cancer up to age of 50, representing an 8.4% cumulative risk of developing breast cancer compared with the risk in the general population of 2%. The most common histopathological type of breast cancer in NF1, as well as in the general population, is infiltrating ductal carcinoma, as observed in our case.11 In our case, there was no evidence of diffuse neurofibromatosis, with no significant family history of neurofibromatosis. The mastectomy specimen showed a foci of neurofibroma with benign clusters of peripheral nerves admixed with fibroblasts, which showed diffuse positivity for S100. To the best of our knowledge, Campos et al were the only group to report a family with individuals with diagnosis of NF1 and breast cancer who were carriers of both BRCA1 and NF1 mutations.12 They concluded that the concurrence of NF1 and breast cancer was probably due to the simultaneous existence of two cancer-predisposing conditions. Most of the cases of breast cancer in NF1 individuals are diagnosed at an advanced stage with a T score greater than 2.12,13 Therefore, the worse prognosis of breast cancer in NF1 may not be a characteristic of the disease itself, but may result from late-stage diagnosis due to the presence of skin neurofibromas, which hinder its identification, or due to the delay in seeking medical care by patients who think the breast mass is a neurofibroma.14,15 Our case of infiltrating ductal carcinoma showed an incidental finding of associated neurofibroma.
Breast neurofibromas usually appear as probably benign on mammography and ultrasound.15,16 Breast neurofibromas tend to be non-calcified, oval lesions with smooth and partially obscured margins. Neurofibroma breast masses can cause lobulations of cutaneous tissue and asymmetrical breast enlargement as well, particularly when they occur in multiples. Ultrasound images of a breast neurofibroma will tend to show well-defined hypoechoic oval lesions, with mild posterior acoustic enhancement.16 Sometimes a breast neurofibroma can show distal acoustic enhancement, which can resemble a breast cyst and result in misdiagnosis. Breast neurofibromas vary in size and shape to a degree but most are quite small, between 1cm and 2cm. Although no evidence of neurofibromatosis was found in our patient, but regular follow up should be done to diagnose benign and malignant neoplasms such as gliomas, malignant peripheral nerve sheath tumors (MPNSTs), juvenile chronic myelomonocytic leukemia, rhabdomyosarcoma and pheochromocytoma because of close association between diffuse neurofibromas and occurrence of these neoplasm in the same patient.9–11
Occurrence of a solitary foci of neurofibroma in a case of infiltrating ductal carcinoma is extremely rare. Moreover absence of neurofibromatosis in our patient make this case exceptional. If any such foci is found, then possibility of all the benign and malignant neoplasms associated with neurofibromatosis should be kept in mind.
None.
The author declares no conflict of interest.

©2017 Akhtar, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.