Journal of
eISSN: 2373-6453


Research Article Volume 9 Issue 3
1Department of Pathology, Faculty of Dentistry, University of Los Andes, Merida, Venezuela
2Department of Biology, Faculty of Science, University of Los Andes, Merida, Venezuela
3Medical University of Los Andes Medical Clinic Rural II, Merida, Venezuela
4Vitalis Laboratory, Merida, Venezuela
Correspondence: Katiusca Villasana, Department of Pathology, Faculty of Dentistry, University of Los Andes, Merida, Venezuela, Tel +58 2742402381, +58 2742630226
Received: November 15, 2022 | Published: November 29, 2022
Citation: Villasana K, Perdomo B, Dugarte L, et al. Azole derivatives inhibit the binding of the RBD domain of SARS-Cov-2 against host ACE2 in in vitro assays. J Hum Virol Retrovirol. 2022;9(3):58-63. DOI: 10.15406/jhvrv.2022.09.00250
The reuse of medicines is a practical and rapid response when early solutions are required for sudden-onset health problems such as SARS-CoV-2, the causative agent of the COVID-19 pandemic during the years 2020 - 2021. In this study we set out to determine by in silico assays with bioinformatics tools using the Swiss Dock online server and in vitro by enzyme-linked immunosorbent assays whether azole derivatives (such as fluconazole, secnidazole, clotrimazole) and ivermectin, can inhibit the interaction between human ACE2 and the RBD domain of SARS-CoV-2 S protein. The result of the docking of azoles and ivermectin showed a significant inhibitory action against RBD of SARS-CoV- 2 S protein and the binding energy. Since the results obtained in the in vitro assays showed a significant inhibition absorbance (OD value < 0.611) of the binding of hACE2 and the SARS-CoV-2 RBD, our results suggest that these azole derivatives can be considered as potential therapeutic candidates. This may suggest that the drugs studied can be used individually or in therapeutic combinations in trials to evaluate effectiveness in patients suffering from COVID-19 both early in the infection and in late stages. Secnidazole and fluconazole are commercially available and affordable drugs.
Keywords: azoles, sars-cov-2, ivermectin, molecular docking simulation, sars-cov-2 spike protein, pharmacologic therapy
ORF; open reading frames, PD; peptidase domain, RBD; receptor binding domain, ACE2; angiotensin-converting enzyme 2, SARS-CoV-2; severe acute respiratory syndrome type 2 coronavirus, NTD; n-terminal domain
SARS-CoV-2 (Severe Acute Respiratory Syndrome Type 2 Coronavirus), the causative agent of the COVID-19 pandemic, has infected more than 179 million people according to official data, of which nearly 4 million have died.1,2 This virus belongs to the family Coronaviridae in the genus Betacoronavirus,3 has a simple chain of RNA in the positive sense of about 29.3 kb, has a nucleocapsid protein (N) that covers the genome, and is surrounded by a lipid membrane that has 3 structural proteins attached to it, the membrane protein (M) the spike protein (S) and the envelope protein (E). The viral genome has 14 open reading frames (ORF), which encode for 27 proteins, being the S protein the most characterized, in view of its importance as a therapeutic target for being the one that establishes the interaction with the host cell receptor; such interaction occurs through the receptor binding domain (RBD) of protein S with the peptidase domain (PD) of Angiotensin-converting enzyme 2 (ACE2), protein that is present in multiple cell types, thus establishing the cellular tropism of the virus.4–6
S protein is a class I homotrimeric protein made up of 1273 amino acids that allows the fusion of the viral envelope with the membrane of the host cell.7 These proteins form trimers, each monomer weighing around 180kDa; it has a shape similar to a stalk of approximately 20 nm in length. The ectodomain consists of a head, which has been extensively studied in vitro, is connected to the membrane by a thin stem, along this there are three flexible hinges that allow it to adopt various conformations, which contributes to a better scan of the receptor It has three receptor binding domains (RBD) (Arg319–Phe541 residues), which have variable conformations. In addition, S protein is densely glycosylated, which gives it a low accessibility to epitopes.8,9
S protein is divided into two parts or functional domains, a receptor-binding domain (S1) and a fusion domain (S2). The S1 domain is associated with pocket join events; to activate a host cell receptor, the RBD of S1 undergoes hinge-like conformational movements that temporarily conceal or expose the determinants of receptor binding. These two states are known as the “down” conformation and the "up” conformation, where the former corresponds to receptor inaccessibility, and is shielded by the n-terminal domain (NTD), while the up domain corresponds to the accessible state, which is believed to be less stable and unprotected. With a distance cut of 4 Å, a total of 17 residues of the RBD are in contact with 20 residues of the PD of the ACE2 (residues Ser19–Asp615), this gives it a Kd of 14.7 nM.8–11
The interest in knowing in depth the structure of S protein and its interaction with ACE 2, lies in the need to develop therapies against COVID-19. Initially, efforts are focused on the development of vaccines to stimulate the production of neutralizing antibodies against the binding of the S protein to the receptor. Some 288 vaccine candidates are currently under development, of which 104 are in the clinical phase, among them there are 13 approved for administration, being inoculated more than 2100 million doses, which mean that at least 20% of the population has received a dose.2 However, it is unknown how long immunological memory is generated.12
Azoles are a heterogeneous group of drugs that share the characteristic of a free imidazole ring bound by carbon-nitrogen bonds to other aromatic rings that confer physico-chemical properties, therapeutic effects and their respective toxicity ranges. According to the number of nitrogens in the ring, the azoles are divided into two groups: the first is triazoles, which have three nitrogens in the main ring and includes fluconazole, itraconazole, posaconazole, isavuconazole and voriconazole which has the particularity of having a group of fluoropyrimidine. The second group is imidazoles, which have two nitrogens in the structure of the main ring and includes secnidazole, miconazole, ketoconazole and clotrimazole.13
1,3-diazol and its compounds show multiple therapeutic properties, including analgesic, antifungal, antitumor, antiviral, anthelmintic, antituberculous, antihistamine, anti-inflammatory, antiallergic, anti-rheumatic, antiprotozoal, anticoagulant, antimalarial and anti-amoebic.14
Due to the long time and high costs required by the traditional production of new drugs, drug repositioning is a practical and rapid response when expeditious solutions are required for sudden-onset health problems, such as epidemics of new infectious diseases.15,16
Molecular docking is an effective method for predicting the structure of ligand-macromolecule complexes. Its use has increased over the years to become one of the most widely used tools for the discovery and development of biologically active molecules. The principle is to minimize a simplified potential energy function that measures the interaction between a ligand and a target macromolecule.17
Among the drugs proposed to test their antiviral effect against SARS-Cov-2, imidazole derivatives were selected.15 Therefore in this study we have proposed to determine by in silico and in vitro assays whether ivermectin and azole derivatives such as fluconazole, secnidazole and clotrimazole can inhibit the interaction between human ACE2 and the RBD domain of the SARS-CoV-2 S protein.
Selection of protein ligand complexes: the RBD domain of S protein and protease 3CLPRO of SARS-CoV-2 were used for the development of this research; in addition, the CYP51 protein of Candida albicans, structures taken from the Protein Data Bank (PDB), the code of each protein is observed in the Table 1.
Protein |
Code |
RBD |
6LU7 |
Sterol-14-alpha-demethylase (CYP51) |
5TZ1 |
3 CL protease |
6M2N |
Table 1 PDB code and name of receptor proteins
For the ligands, the structures of FDA-approved drugs taken from the Pubchem database (https://pubchem.ncbi.nlm.nih.gov/) was used, the structure of fluconazole, secnidazole, clotrimazole and ivermectin were used, which can be seen in the Table 2.
Ligand |
IUPAC |
PubChem Code |
Structure |
Fluconazole |
2-(2,4-difluorophenyl)- |
3365 |
|
Secnidazole |
1-(2-methyl-5-nitro-1H-imidazol-1-yl)propan-2-ol |
71815 |
|
Clotrimazole |
1-[(2-clorofenil)-difenil-metil]imidazol |
2812 |
|
Ivermectin |
(1'R,2R,4'S,5S,6R,8'R,10'E,12'S,13'S,14'E,16'E,20'R,21'R,24'S)-21',24'-dihidroxi-12'-([(2R,4S,5S,6S)-5-([(2S,4S,5S,6S)-5-hidroxi-4-metoxi-6-metiloxan-2-il]oxi)-4-metoxi-6-metiloxan-2-il]oxi)-5,11',13',22'-tetrametil-6-(propan-2-il)-3',7',19'-trioxaspiro[oxano-2,6'-tetraciclo[15.6.1.1⁴,⁸.0²⁰,²⁴]pentacosano]-10',14',16',22'-tetraen-2'-ona; (1'R,2R,4'S,5S,6R,8'R,10'E,12'S,13'S,14'E,16'E,20'R,21'R,24'S)-6-[(2R)-butan-2-il]-21',24'-dihidroxi-12'-([(2R,4S,5S,6S)-5-([(2S,4S,5S,6S)-5-hidroxi-4-metoxy-6-metiloxan-2-il]oxi)-4-metoxin-6-metiloxan-2-il]oxi)-5,11',13',22'-tetrametil-3',7',19'-trioxaspiro[oxano-2,6'-tetraciclo[15.6.1.1⁴,⁸.0²⁰,²⁴]pentacosano]-10',14',16',22'-tetraen-2'-ona |
9812710 |
Table 2 Name, PubChem code and structure of ligands
Molecular docking: molecular docking between protein-ligand complexes was performed using the Swiss Dock online server (http://www.swissdock.ch/), in which a blind docking was performed between the following protein-ligand complexes: RBD-Fluconazole, RBD-secnidazole, 3CLPro-Fluconazole, Fluconazole-CYP51. The complexes formed between Fluconazole-CYP51, and the value reported for the coupling between the RBD-ACE2, were taken as a control. The results of the coupling were displayed and analyzed using the UCSF Chimera free license software in its version 1.5.3 (https://www.cgl.ucsf.edu/chimera/1.5.3/docs/relnotes/1.5.html).
Selecting the best union pose: 3 criteria were taken to select the best binding poses based on the union energy of the complex, the distribution of the cluster energy and the reaction rate constant:
In vitro assays
For the in vitro assays, by Enzyme-Linked Inmune Sorbent Assay (ELISA) the GenScript® brand cPass™ SARS-CoV-2 Neutralization Antibody Detection Kit (Lot A201207-C01, Piscataway, NJ 08854 USA) was used, which uses the receptor binding domain (RBD) of the SARS-CoV-2 peroxidase-bound peroxidase (HRP) S protein, and the ACE2 cell receptor immobilized on the surface of the wells.
Preparation of reagents: All reagents must be outside the refrigerator and at room temperature (22°C in our laboratory) at the time of preparation.
Neutralization reaction: The positive and negative controls and samples of the previously indicated drugs were mixed separately with the HRP-RBD solution with a volume ratio of 1:1; and incubated at 37°C in Globe incubation stove 25 liters® (Laboratory Equipment Vitalis, Mérida-Venezuela) for 30 minutes. Subsequently, 100 μL of the positive control, negative control mixture and each of the drug samples in their respective wells were added, covered with Plate Sealer and incubated at 37°C for 15 minutes.
Then the Plate Sealer is removed and 4 washes are made with 260 μL of washing solution. The plate is struck against an absorbent towel to remove any liquid residue that may remain in the wells after washing.
Substrate reaction and absorbance measurement: 100 μL of TMB substrate solution is added to each of the wells and incubated at 20-25°C (22°C in our laboratory) for 15 minutes (time counting should be started once the substrate solution has been added to the first well). Then 50 μL of Stop solution is added to each of the wells to stop the reaction and immediately proceed to perform the absorbance measurement in spectrophotometer (Rayto 2100®, Laboratory Equipment Vitalis, Mérida-Venezuela) at a wavelength of 450nm.
Interpretation of the results: to obtain the percentage of inhibition of each of the samples studied, the optical density obtained from the samples and the negative control must be used by the following formula:
Based on the percentage y optical density (OD value) obtained from this formula, it is considered that there is inhibition against SARS-CoV-2 if it results in a percentage equal to or greater than 30% (with OD value > 1.567 negative control).
Molecular docking: the results obtained from the molecular coupling calculations are shown in Table 3. The values for the best poses obtained from the docking are presented following the three selection criteria previously indicated. It can be seen that the values obtained between the analyzed docking are above the value or very close to the value of binding energy of the controls. The highest binding energy is found between ivermectin and the RBD domain, followed by fluconazole and secnidazole.
Ligand |
Protein |
DG Union Energy (Kcal/mol) |
Cluster power |
pKi (µM) |
Fluconazole |
RBD |
-7,312617 |
-11,7181 |
3,36 |
Fluconazole |
3CLPro |
-7,1301847 |
-7,32826 |
4,60 |
Fluconazole |
CYP51 |
-7,231986 |
-11,3912 |
3,86 |
Secnidazole |
RBD |
-6,7223206 |
-16,3825 |
9,30 |
Clotrimazole |
RBD |
-6,7945123 |
40,3818 |
8,22 |
Ivermectin |
RBD |
-7,9329777 |
-2,69126 |
1,17 |
Table 3 Results of the best poses when using the selection criteria
In addition, a molecular interaction was established between the protease 3CLPro and fluconazole, to predict whether these can establish interaction and, consequently, to establish the possibility that fluconazole neutralizes not only one but two target proteins of great importance for the establishment of infection and replication of SARS-CoV-2. From this molecular interaction a binding energy was obtained with a value a little higher than the control, therefore this drug could bind favorably to neutralize this protease.
In Table 4 the amino acids and hydrogen bonds involved in the interaction between the ligand and the white protein are observed, these are responsible for the value of the binding energy, and contribute positively to the affinity of the drug for the white protein.
Coupling |
Amino acids |
Hydrogen Bridges |
Fluconazole-RBD |
Glycine -71 |
- |
|
|
|
Fluconazole-CYP51 |
Glycine-464 |
- |
|
|
|
Fluconazole-3CLPRO |
Methionine-185 |
1 |
|
|
|
Secnidazole-RBD |
Tryptophan-207 |
4 |
|
|
|
Clotrimazole-RBD |
Leucine-27 |
- |
|
|
|
Ivermectin-RBD |
Glutamate-14 |
- |
Table 4 Amino acids and hydrogen bridges involved in couplings
Figure 1 shows the best interaction poses obtained during the coupling between fluconazole and RBD of SARS-Cov-2 protein S (Figure 1A), between fluconazole and SARS-Cov-2 protease 3CLPRO (Figure 1B), between fluconazole and Candida albicans protein CYP51 (Figure 1C) , between secnidazole and SARS-Cov-2 S protein RBD (Figure 1D), between clotrimazole and SARS-Cov-2 S protein RBD (Figure 1E) and between ivermectin and SARS-Cov-2 S protein RBD (Figure 1F).

Figure 1 (A) Interaction poses obtained in the coupling between fluconazole-RBD, (B) fluconazole-3CLPRO, (C) fluconazole-CYP51, (D) secnidazole-RBD, (E) clotrimazole-RBD and (F) ivermectin-RBD.
Figure 2 shows the amino acids involved in the interaction between fluconazole and RBD of SARS-Cov-2 S protein (Figure 2A), fluconazole and SARS-CoV-2 protease 3CLPRO (Figure 2B), fluconazole and Candida albicans CYP51 protein (Figure 2C), between SARS-CoV-2 S protein RBD and Secnidazole (Figure 2D), between Clotrimazole and SARS-Cov-2 S protein RBD (Figure 2E), and between SARS-Cov-2 S protein RBD and Ivermectin (Figure 2F).
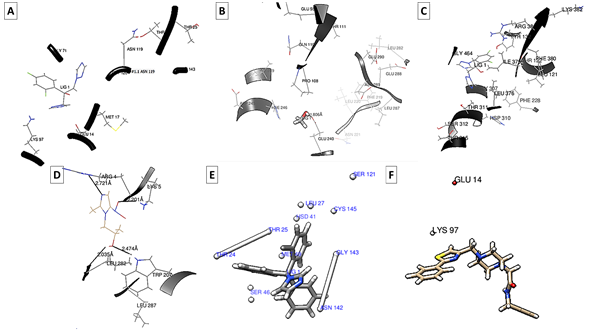
Figure 2 Amino acids involved in the interaction between fluconazole-RBD (A), fluconazole-3CLPRO (B), fluconazole-CYP51 (C), secnidazole-RBD (D), clotrimazole-RBD (E) and ivermectin-RBD (F).
Figure 3 shows the interaction of fluconazole with the hydrophobic surface of the RBD of the SARS-CoV-2 S protein, the interaction between fluconazole with the hydrophobic surface of the SARS-CoV-2 protein 3CLPRO (Figure 3B), the interaction of fluconazole with the hydrophobic surface of the CYP51 protein of Candida albicans (Figure 3C), the interaction between secnidazole and RBD of SARS-CoV-2 S protein (Figure 3D), the interaction between clotrimazole and RBD of SARS-Cov-2 S protein (Figure 3E) and the interaction between ivermectin and RBD of SARS-Cov-2 S protein (Figure 1F).
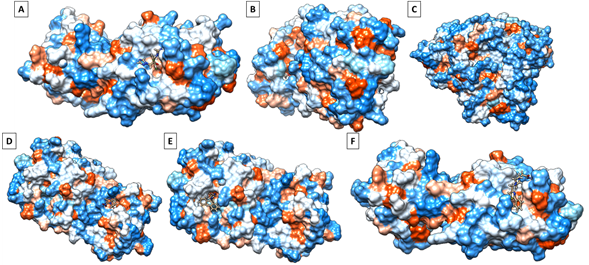
Figure 3 Interaction between the drugs and the hydrophobic domain of the proteins studied. Fluconazole-RBD (A), fluconazole-3CLPRO (B), fluconazole-CYP51 (C), secnidazole-RBD (D), clotrimazole-RBD (E), and ivermectin-RBD (F).
Immunoassays: Figure 4 shows of optical density value (OD450) inhibition and for each of the samples tested. Both fluconazole (OD: 0.561) and clotrimazole (OD: 0.523) had a 64% inhibition on the RBD of the SARS-CoV-2 S protein, similarly, ivermectin obtained an inhibition percentage of 61% (OD: 0.611), while secnidazole obtained the highest percentage of inhibition on the RBD of the S protein, with 73.70% (OD: 0.412). Control negative OD: 1.567.
From the beginning of the pandemic began the search for therapeutics with the ability to counteract the viral multiplication or severity of the disease; drugs that pursue this purpose have been classified into 4 types, those directed to the virus, those directed to the host, those that combine both strategies, and those drugs that act as adjuvants modulators of the disease. The first focus on inhibiting viral replication, the second group interferes with host factors, and finally the adjuvant molecules that act by modulating the immune response to reduce inflammation.18
As our understanding of the disease improves, many drugs with very promising prospects initially ended up being excluded by the FDA as a result of uninspiring results in clinical trials. These include antimalarials such as chloroquine and hydroxychloroquine and the antiparasitic ivermentin. The latter with effective antiviral results but at doses well above the safety ranges.19–21
One of the currently widely used drugs is Remdesivir, however, its high cost makes it inaccessible to populations with limited resources. It is a nucleotide analogue whose mechanism of action is to inhibit RNA-dependent viral RNA polymerase (RdRp), which prevents viral replication by showing reduction in patients' viral load; in addition, in silico studies have shown a high affinity between Remdesivir and RdRp with an ΔG of 9.34 ± 0.38 Kcal/mol.22–24
The in silico assays previously exposed demonstrate that the azole derivatives studied present a free energy binding to the RBD of the S protein of SARS-Cov-2 similar to that which they have for their therapeutic target in fungi, the enzyme CYP51.25 In addition, it was evidenced that fluconazole established a molecular interaction with the protease 3CLPro and presented a binding-free energy with a value similar to that established with the CYP51 enzyme, so it is possible that fluconazole has the ability to neutralize this SARS-Cov-2 protease.
In vitro assays show that the azole derivatives studied inhibit the binding between the RBD of the S protein and the human ACE2 with binding percentages ranging from 60% to 70%, therefore there could be significant protection against SARS-Cov-2, which makes these drugs potential therapeutic candidates against the virus of the pandemic COVID 19. In addition, the dose of 2 g/day recommended for secnidazole26 is by above the inhibition constant obtained in our in silico studies.
Likewise, although fluconazole showed a lower percentage of inhibition than secnidazole, the resulting inhibition constant is higher than the FDA approved dose (the recommended dose of 150 mg/day).27 Although clotrimazole showed a significant inhibitory effect of binding between SARS-Cov-2 RBD and human ACE2, its preferred prescribed use is topical which would limit its applicability due to the systemic nature of SARS-Cov-2 infection.28
Ivermectin showed a slightly higher percentage of inhibition than fluconazole and clotrimazole, but the inhibition constant obtained in our in silico results are close to the maximum FDA-approved dose of 12mg/day,29 making it difficult to use as a candidate for inhibition of the RBD-ACE2 junction.
Secnidazole and fluconazole are widely studied and safe drugs, and the commercial forms available on the market are economically accessible.30 The inhibition constants obtained in our in silico studies show the probability that the doses required to generate viral inhibition in vivo are lower than those currently approved, which is why there would be no risk of toxicity.
None.
None.
The author declares no conflicts of interest.

©2022 Villasana, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.
 World Malaria Day, observed annually on April 25SUP>th. The purpose of this day is to raise awareness about the global struggle to combat malaria, highlighting the need for continued research, prevention, and treatment strategies to eradicate this deadly disease. So, it is an opportunity to all the researchers to submit your interesting papers on raising the approachability on modern vaccine development, advances in disease control, prevention and eradication and the submissions received till April 25th, 2024 will be offered a best discount of 30% for publication in Journal of Human Virology & Retro virology.
World Malaria Day, observed annually on April 25SUP>th. The purpose of this day is to raise awareness about the global struggle to combat malaria, highlighting the need for continued research, prevention, and treatment strategies to eradicate this deadly disease. So, it is an opportunity to all the researchers to submit your interesting papers on raising the approachability on modern vaccine development, advances in disease control, prevention and eradication and the submissions received till April 25th, 2024 will be offered a best discount of 30% for publication in Journal of Human Virology & Retro virology.